Advanced Synthesis Of Azabicyclo Alkanone Dipeptide Mimetics For Commercial Drug Development
The pharmaceutical industry continuously seeks robust scaffolds that can mimic the biological activity of peptides while overcoming their inherent pharmacokinetic limitations. Patent CN111560017B introduces a groundbreaking class of dipeptide mimetics based on an azabicyclo [X, Y, 0] alkanone framework, offering a compelling solution for modern drug discovery programs. These novel compounds are engineered to possess superior metabolic stability, high bioavailability, and enhanced membrane permeability, addressing the critical challenges associated with traditional polypeptide therapeutics. The structural rigidity provided by the bicyclic core allows for precise conformational control, enabling the simulation of specific peptide turns such as beta-turns, which is essential for targeting protein-protein interactions. As a leading manufacturer, we recognize the immense potential of this technology in developing next-generation inhibitors for enzymes like ACE, NEP, and caspases, as well as viral proteases. This report provides a deep technical analysis of the synthesis methodology and its commercial implications for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of conformationally constrained peptidomimetics has been plagued by lengthy synthetic routes and poor overall yields, which significantly hinder their progression from lead compounds to clinical candidates. Traditional methods often require multiple protection and deprotection steps, chiral resolutions, and harsh reaction conditions that are incompatible with sensitive functional groups. Furthermore, the lack of modularity in older synthetic strategies makes it difficult to rapidly generate diverse libraries of analogs for structure-activity relationship (SAR) studies. The reliance on stoichiometric reagents and inefficient cyclization techniques often results in substantial waste generation and high production costs, making these compounds less attractive for large-scale commercial manufacturing. Consequently, many promising peptide-based drug candidates fail to reach the market due to insurmountable economic and technical barriers associated with their production processes.
The Novel Approach
The methodology disclosed in CN111560017B represents a paradigm shift by employing a concise and highly efficient synthetic strategy centered around Ring-Closing Metathesis (RCM) and transition metal-catalyzed cross-coupling reactions. This approach drastically reduces the number of synthetic steps required to construct the complex azabicyclo alkanone skeleton, thereby improving the overall atom economy and process mass intensity. By utilizing a diene precursor that undergoes intramolecular cyclization, the core bicyclic structure is formed with high stereocontrol and minimal byproduct formation. The subsequent functionalization via palladium-catalyzed coupling allows for the late-stage introduction of diverse aryl, heteroaryl, and alkyl groups, providing unparalleled flexibility in molecular design. This modularity not only accelerates the drug discovery timeline but also simplifies the manufacturing process, making it ideally suited for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and RCM
The core of this innovative synthesis lies in the strategic application of Ring-Closing Metathesis to form the seven or eight-membered rings fused to the pyrrolidine core. The process begins with a diene substrate, typically an N-acryloyl allylpyrrolidine derivative, which is subjected to a ruthenium-based carbene catalyst, such as Grubbs' second-generation catalyst, under reflux conditions in anhydrous toluene. This catalytic cycle facilitates the cleavage and reformation of carbon-carbon double bonds, driving the equilibrium towards the formation of the cyclic alkene product while releasing ethylene gas. The efficiency of this step is critical, as it establishes the stereochemical foundation for the entire molecule, ensuring the correct spatial orientation of the substituents required for biological activity. The robustness of the RCM reaction allows it to tolerate various functional groups, making it a versatile tool for constructing complex heterocyclic systems found in high-purity OLED material and pharmaceutical contexts alike.
Following the cyclization, the resulting unsaturated lactam undergoes a regioselective halogenation and elimination sequence to generate a vinyl halide intermediate, which serves as a pivotal handle for further diversification. This intermediate is then subjected to transition metal-catalyzed cross-coupling reactions, such as Suzuki-Miyaura or Buchwald-Hartwig couplings, to introduce the R1 substituent at the C6 position of the bicyclic framework. The use of palladium catalysts in conjunction with phosphine ligands ensures high turnover numbers and excellent selectivity, minimizing the formation of homocoupling byproducts. Finally, catalytic hydrogenation of the remaining double bond yields the saturated azabicyclo alkanone scaffold with high diastereoselectivity. This comprehensive mechanistic pathway ensures that impurities are kept to a minimum, satisfying the rigorous purity requirements demanded by regulatory agencies for reliable agrochemical intermediate supplier standards.
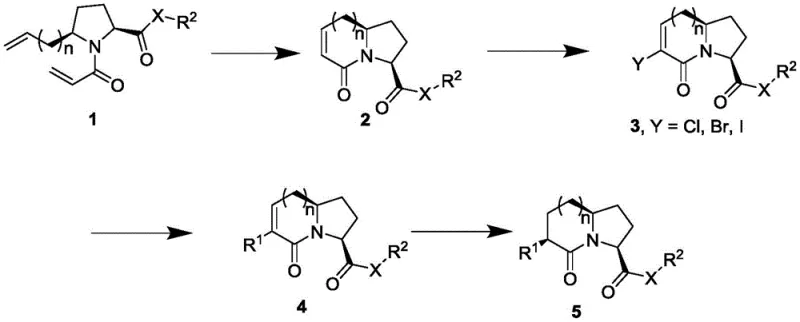
How to Synthesize Azabicyclo Alkanone Efficiently
The practical implementation of this synthesis requires careful control of reaction parameters to maximize yield and purity at every stage. The initial RCM step demands strictly anhydrous conditions and an inert atmosphere to prevent catalyst decomposition, while the subsequent coupling reactions require optimized base and solvent systems to facilitate oxidative addition and reductive elimination cycles. Detailed standard operating procedures for each transformation are essential to ensure reproducibility across different batches and scales. For research and development teams looking to replicate or adapt this chemistry, understanding the nuances of catalyst loading, temperature profiles, and workup procedures is paramount. The detailed standardized synthesis steps are outlined in the guide below to assist technical teams in process validation.
- Perform Ring-Closing Metathesis (RCM) on the diene precursor using a Grubbs catalyst to form the core bicyclic alkene structure.
- Conduct halogenation followed by elimination to generate the vinyl halide intermediate suitable for cross-coupling reactions.
- Execute transition metal-catalyzed coupling to introduce diverse functional groups, followed by catalytic hydrogenation to finalize the saturated scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers transformative benefits for procurement managers and supply chain directors seeking to optimize their sourcing strategies for complex pharmaceutical intermediates. The streamlined nature of the process directly translates to reduced operational expenditures, as fewer unit operations mean lower labor costs, reduced energy consumption, and decreased solvent usage. The elimination of cumbersome protection group manipulations and chromatographic purifications at intermediate stages further contributes to significant cost savings in fine chemical manufacturing. Moreover, the use of commercially available starting materials and robust catalysts mitigates the risk of supply disruptions, ensuring a stable and continuous flow of raw materials. This reliability is crucial for maintaining production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive chiral auxiliaries and reduces the total number of isolation steps, which dramatically lowers the cost of goods sold. By avoiding the use of stoichiometric amounts of heavy metal reagents and minimizing waste generation, the process aligns with green chemistry principles, reducing disposal costs and environmental compliance burdens. The high efficiency of the coupling reactions ensures that expensive boronic acids or amines are utilized effectively, maximizing the return on investment for raw materials. Overall, the process economics are significantly improved compared to traditional linear peptide synthesis methods.
- Enhanced Supply Chain Reliability: The reliance on well-established chemical transformations such as hydrogenation and cross-coupling ensures that the manufacturing process is not dependent on niche or proprietary reagents that may face supply constraints. The versatility of the route allows for the sourcing of alternative starting materials if necessary, providing a buffer against market volatility. Furthermore, the robustness of the chemistry supports production in multiple geographic locations, diversifying the supply base and reducing the risk of single-point failures. This resilience is vital for securing the long-term availability of critical drug substances.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The absence of hazardous reagents and the generation of benign byproducts simplify waste treatment protocols, ensuring compliance with stringent environmental regulations. The ability to produce large quantities of the intermediate without compromising quality supports the commercial scale-up of complex polymer additives and pharmaceutical ingredients. This scalability ensures that the supply chain can respond flexibly to fluctuations in market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dipeptide mimetics. The answers are derived directly from the patent specifications and our internal technical assessments to provide accurate guidance for stakeholders. Understanding these aspects is crucial for evaluating the feasibility of integrating this technology into existing drug development pipelines. We encourage partners to review these insights to fully appreciate the value proposition of this advanced synthetic platform.
Q: What are the metabolic advantages of azabicyclo alkanone dipeptide mimetics?
A: These compounds feature a rigid bicyclic structure that significantly enhances metabolic stability and bioavailability compared to linear peptides, while maintaining strong membrane permeability for effective drug delivery.
Q: How does the new synthesis route improve manufacturing efficiency?
A: The patented method utilizes a convergent strategy involving Ring-Closing Metathesis and palladium-catalyzed coupling, which shortens the overall synthetic route and allows for the versatile introduction of various substituents.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process relies on robust chemical transformations such as RCM and standard hydrogenation, which are well-established in industrial settings, ensuring reliable commercial scale-up and consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azabicyclo Alkanone Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure required to bring this sophisticated chemistry from the laboratory to commercial reality. Our team of experienced chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of dipeptide mimetic meets the highest international standards. Our commitment to quality and consistency makes us the preferred partner for multinational corporations seeking a reliable source for high-value pharmaceutical intermediates.
We invite you to collaborate with us to explore the full potential of this technology for your specific therapeutic applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating how our manufacturing capabilities can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us support your journey from discovery to market with our world-class CDMO services.
