Revolutionizing Pharmaceutical Intermediate Synthesis with Novel Lewis Acid Catalysis
Revolutionizing Pharmaceutical Intermediate Synthesis with Novel Lewis Acid Catalysis
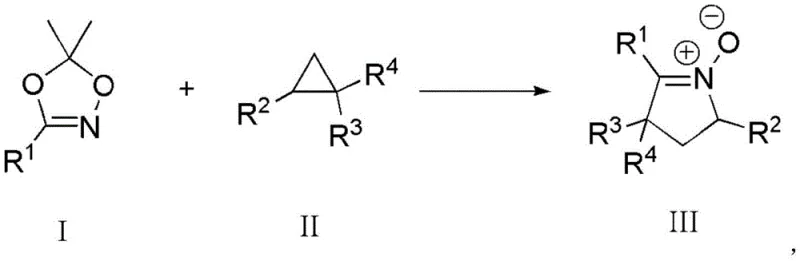
The pharmaceutical industry constantly seeks robust synthetic routes for complex heterocyclic scaffolds, and the recent disclosure in patent CN109020862B presents a transformative approach for constructing 3,4-dihydro-1-pyrroline-N-oxide derivatives. This specific class of compounds serves as a critical 1,3-dipolar precursor for building pyrrolo[1,2-b]isoxazole structural units, which are ubiquitous in modern medicinal chemistry and natural product synthesis. The innovation lies in the utilization of a Lewis acid-promoted reaction between substituted dioxazoles and substituted cyclopropanes, bypassing the need for hazardous oxidants. This technical breakthrough not only simplifies the operational workflow but also dramatically improves the diastereoselectivity of the final product. For R&D directors and process chemists, this represents a significant opportunity to streamline the synthesis of high-value intermediates while maintaining stringent purity standards required for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dihydro-1-pyrroline-N-oxides has relied heavily on two primary strategies, both of which present substantial drawbacks for large-scale manufacturing. The first conventional method involves the oxidation of cyclic secondary amines or hydroxylamines using strong oxidizing agents such as m-CPBA or Oxone. While chemically feasible, this route is plagued by severe reaction conditions that often lead to poor functional group tolerance, limiting the diversity of substrates that can be successfully processed. Furthermore, these oxidative processes frequently suffer from mediocre yields and inadequate regioselectivity, resulting in complex purification challenges and increased waste generation. The second traditional approach utilizes intramolecular condensation between hydroxylamines and carbonyl compounds, which, although milder, is often synthetically cumbersome when aiming for structural diversity. These legacy methods create bottlenecks in supply chains due to their sensitivity and the high cost associated with handling hazardous reagents.
The Novel Approach
In stark contrast, the methodology outlined in patent CN109020862B introduces a streamlined pathway that leverages the reactivity of substituted dioxazoles and cyclopropanes under Lewis acid promotion. This novel strategy operates under significantly milder conditions, eliminating the need for aggressive oxidants and thereby enhancing the safety profile of the manufacturing process. The reaction demonstrates exceptional substrate applicability, accommodating a wide range of electron-withdrawing groups and aryl substituents without compromising efficiency. By shifting the mechanistic paradigm from oxidation to Lewis acid-catalyzed ring construction, this method achieves superior diastereoselectivity, which is crucial for reducing the burden on downstream chiral separation processes. For procurement and supply chain managers, this translates to a more reliable production timeline and reduced dependency on volatile reagent markets, ensuring a stable supply of high-purity pharmaceutical intermediates.
Mechanistic Insights into Lewis Acid-Promoted Cyclization
The core of this synthetic innovation relies on the precise activation of the cyclopropane ring by a Lewis acid catalyst, such as Ytterbium(III) trifluoromethanesulfonate (Yb(OTf)3). The Lewis acid coordinates with the electron-rich centers of the reactants, lowering the activation energy required for the ring-opening of the strained cyclopropane system. This activation facilitates a nucleophilic attack by the dioxazole nitrogen, initiating a cascade that leads to the formation of the five-membered 3,4-dihydro-1-pyrroline-N-oxide ring. The elegance of this mechanism lies in its ability to control stereochemistry through the coordination sphere of the metal center, ensuring that the resulting product exhibits high diastereomeric purity. Understanding this catalytic cycle is essential for process chemists aiming to optimize reaction parameters for commercial scale-up, as the choice of Lewis acid and solvent polarity can fine-tune the reaction kinetics and selectivity profiles.
Furthermore, the inclusion of molecular sieves in the reaction mixture plays a pivotal role in maintaining the integrity of the catalytic cycle. Trace amounts of water can coordinate with the Lewis acid, effectively poisoning the catalyst and halting the reaction progress. By rigorously excluding moisture through the use of 4A molecular sieves, the system ensures that the Lewis acid remains available to activate the cyclopropane substrate throughout the reflux period. This attention to detail in the reaction setup underscores the robustness of the method, allowing for consistent yields even with sensitive substrates. For quality control teams, this mechanism implies a lower risk of hydrolysis by-products, simplifying the impurity profile and facilitating easier compliance with regulatory standards for pharmaceutical intermediates.
How to Synthesize 3,4-Dihydro-1-Pyrroline-N-Oxide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational protocols to maximize yield and safety. The process begins with the preparation of a dry reaction environment, where molecular sieves are activated and added to an aprotic solvent such as 1,2-dichloroethane (DCE). The Lewis acid catalyst is then introduced, followed by the precise addition of the substituted dioxazole and cyclopropane reactants in stoichiometric ratios optimized for conversion. The mixture is subsequently heated to reflux, typically for a duration of five hours, to ensure complete consumption of the starting materials. Following the reaction, the workup involves simple filtration to remove the molecular sieves and catalyst residues, followed by solvent removal and purification via silica gel column chromatography. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility.
- Prepare the reaction system by adding dried molecular sieves and a Lewis acid catalyst such as Yb(OTf)3 to an aprotic solvent like dichloroethane.
- Introduce the substituted dioxazole and substituted cyclopropane reactants into the mixture under controlled stoichiometric ratios.
- Heat the reaction mixture to reflux for approximately 5 hours, then filter and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Lewis acid-catalyzed synthesis offers profound benefits for procurement and supply chain operations within the fine chemical sector. By eliminating the need for hazardous strong oxidants, the process significantly reduces the costs associated with reagent procurement, storage, and waste disposal. The mild reaction conditions also lower the energy requirements for heating and cooling, contributing to a more sustainable and cost-effective manufacturing footprint. For supply chain heads, the robustness of this method means fewer batch failures and a more predictable production schedule, which is critical for maintaining continuity in the supply of key pharmaceutical intermediates. The ability to tolerate diverse functional groups further allows for a flexible production line capable of handling various derivatives without extensive retooling.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous oxidizing agents like m-CPBA directly translates to substantial cost savings in raw material procurement. Additionally, the simplified workup procedure, which avoids complex quenching steps required for strong oxidants, reduces labor hours and solvent consumption during purification. The high diastereoselectivity of the reaction minimizes the loss of material during chiral separation, effectively increasing the overall mass balance and reducing the cost per kilogram of the active intermediate. These factors combine to create a leaner manufacturing process that enhances profit margins without compromising on quality.
- Enhanced Supply Chain Reliability: The use of commercially available and stable Lewis acid catalysts ensures that the supply chain is not vulnerable to the volatility often seen with specialized oxidizing reagents. The reaction's tolerance for various substrates means that suppliers can maintain inventory flexibility, switching between different derivatives based on market demand without significant downtime. Furthermore, the mild conditions reduce the risk of equipment corrosion or degradation, leading to lower maintenance costs and higher asset availability. This reliability is crucial for long-term contracts with pharmaceutical companies that require guaranteed delivery schedules.
- Scalability and Environmental Compliance: This synthesis method is inherently scalable, utilizing standard reactor setups and common solvents that are easily managed in large-scale facilities. The absence of heavy metal waste or toxic oxidative by-products simplifies the environmental compliance process, reducing the burden on wastewater treatment systems. The high atom economy of the reaction, driven by the efficient ring-closing mechanism, aligns with green chemistry principles, making it an attractive option for companies aiming to reduce their carbon footprint. This environmental advantage also facilitates smoother regulatory approvals in regions with strict chemical manufacturing guidelines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 3,4-dihydro-1-pyrroline-N-oxide derivatives based on the patented technology. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing production workflows. The answers reflect the practical implications of the chemical mechanism and its impact on operational efficiency.
Q: What are the primary advantages of this Lewis acid-catalyzed method over conventional oxidation routes?
A: Unlike conventional methods requiring harsh oxidants like m-CPBA which suffer from poor functional group tolerance, this novel approach utilizes mild Lewis acid conditions that significantly enhance diastereoselectivity and substrate applicability.
Q: How does the use of molecular sieves impact the reaction efficiency?
A: The addition of molecular sieves effectively removes trace water from the system, preventing hydrolysis or deactivation of the sensitive Lewis acid catalyst, thereby ensuring consistent high yields and reaction stability.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process employs standard reagents and simple workup procedures like filtration and distillation, making it highly suitable for commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydro-1-Pyrroline-N-Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development pipeline. Our technical team has extensively analyzed the pathway described in CN109020862B and possesses the expertise to scale this diverse pathway from 100 kgs to 100 MT/annual commercial production. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that ensure every batch complies with international standards. Our experience in handling complex Lewis acid-catalyzed reactions allows us to optimize the process for maximum yield and safety, providing our partners with a competitive edge in the market.
We invite procurement leaders and R&D directors to collaborate with us for a Customized Cost-Saving Analysis tailored to your specific project needs. By leveraging our manufacturing capabilities, you can secure a stable supply of high-purity 3,4-dihydro-1-pyrroline-N-oxide derivatives while optimizing your overall production costs. Please contact our technical procurement team to request specific COA data and route feasibility assessments. We are ready to support your supply chain with reliable, scalable, and cost-effective chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
