Advanced Synthetic Strategy For 3-Oxetanecarboxylic Acid: Scalability And Commercial Viability Analysis
Advanced Synthetic Strategy For 3-Oxetanecarboxylic Acid: Scalability And Commercial Viability Analysis
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing strained heterocyclic motifs, among which the oxetane ring stands out for its unique metabolic stability and ability to modulate physicochemical properties in drug candidates. A pivotal development in this domain is detailed in patent CN108727309B, which discloses a novel synthetic method for 3-oxetanecarboxylic acid, a critical building block for various bioactive molecules. This technology addresses the historical scarcity and high cost of this intermediate by proposing a route that utilizes inexpensive, commercially available starting materials while rigorously avoiding hazardous reagents. The strategic significance of this patent lies not only in its chemical elegance but also in its demonstrated potential for industrial adoption, offering a reliable solution for supply chain bottlenecks that have previously hindered downstream biomedical research and development efforts globally.
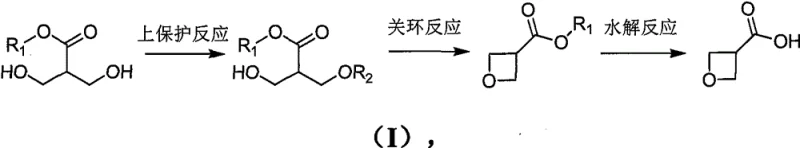
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxetane carboxylic acids has been plagued by significant technical and economic barriers that limit their widespread application in medicinal chemistry. Traditional routes often rely on precursors that are either prohibitively expensive or require handling dangerous reagents capable of causing explosions or severe toxicity, thereby imposing heavy burdens on safety compliance and insurance costs for manufacturing facilities. Furthermore, many legacy processes suffer from poor atom economy and low yields, necessitating complex purification steps that generate substantial chemical waste and drive up the final unit price of the intermediate. These inefficiencies create a fragile supply chain where minor disruptions in raw material availability can lead to prolonged shortages, stalling the progress of critical drug discovery programs that depend on consistent access to high-purity oxetane derivatives for structure-activity relationship studies.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a streamlined three-step sequence that fundamentally reshapes the economic and safety landscape of producing this valuable intermediate. By selecting 3-hydroxy-2-(hydroxymethyl) methyl propionate as the starting scaffold, the process leverages a low-cost feedstock that is readily accessible in the global chemical market, effectively decoupling production costs from volatile specialty chemical pricing. The reaction design cleverly bypasses the need for exotic catalysts or extreme conditions, instead employing standard organic transformations such as selective protection, base-mediated intramolecular substitution, and hydrolysis. This approach not only enhances the safety profile by eliminating explosive risks but also simplifies the operational workflow, making it an ideal candidate for a reliable pharmaceutical intermediate supplier aiming to offer cost reduction in pharmaceutical intermediates manufacturing without compromising on quality or regulatory compliance standards.
Mechanistic Insights into Base-Mediated Cyclization and Ester Hydrolysis
The core chemical innovation of this process resides in the precise orchestration of functional group transformations to construct the strained four-membered oxetane ring with high fidelity. The initial step involves the selective mono-protection of a diol precursor using p-toluenesulfonyl chloride, where the reaction conditions are meticulously controlled to ensure that only the primary hydroxyl group is activated, leaving the secondary hydroxyl free for the subsequent nucleophilic attack. This selectivity is crucial for preventing polymerization or the formation of unwanted bis-protected byproducts, which would otherwise complicate downstream purification. Following protection, the addition of a strong non-nucleophilic base, such as potassium tert-butoxide, triggers an intramolecular SN2 reaction. The deprotonated secondary hydroxyl group attacks the carbon bearing the sulfonate leaving group, forcing the formation of the thermodynamically challenging oxetane ring. This cyclization step is optimized to proceed at moderate temperatures, balancing reaction kinetics with the stability of the strained ring system to maximize yield.
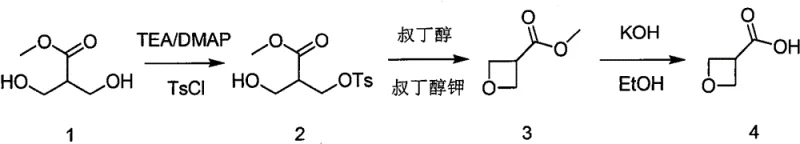
Following the successful construction of the heterocyclic core, the final stage focuses on unveiling the carboxylic acid functionality through a controlled hydrolysis of the methyl ester. This step is executed under alkaline conditions using reagents like potassium hydroxide in ethanol, which efficiently cleaves the ester bond while maintaining the integrity of the sensitive oxetane ring. The mechanism involves the nucleophilic attack of the hydroxide ion on the carbonyl carbon, forming a tetrahedral intermediate that collapses to release methanol and the carboxylate salt. Subsequent acidification to a pH range of 1 to 4 protonates the carboxylate, precipitating the final 3-oxetanecarboxylic acid product. This purification strategy is particularly effective for impurity control, as most organic side products remain soluble in the organic extraction phase or are removed during the aqueous washes, ensuring that the final material meets the rigorous purity specifications required for high-purity pharmaceutical intermediates used in clinical trial material synthesis.
How to Synthesize 3-Oxetanecarboxylic Acid Efficiently
Implementing this synthetic route requires careful attention to stoichiometry and temperature control to replicate the high yields reported in the patent literature. The process begins with the dissolution of the starting diol in a chlorinated solvent, followed by the sequential addition of organic bases and the sulfonylating agent at low temperatures to manage exotherms. Once the protected intermediate is isolated, it undergoes cyclization in an alcoholic solvent with a bulky base, a step that demands precise thermal regulation to ensure complete conversion without degradation. Finally, the ester hydrolysis is conducted under reflux conditions, followed by a careful pH adjustment to isolate the target acid. For detailed operational parameters, stoichiometry ratios, and workup procedures, please refer to the standardized synthesis guide below which outlines the exact experimental conditions validated for commercial production.
- Selective Protection: React 3-hydroxy-2-(hydroxymethyl) methyl propionate with p-toluenesulfonyl chloride in the presence of triethylamine and DMAP to protect the primary hydroxyl group.
- Cyclization: Treat the protected intermediate with potassium tert-butoxide in tert-butyl alcohol at 60-70°C to induce intramolecular ring closure forming the oxetane ester.
- Hydrolysis: Reflux the oxetane ester with potassium hydroxide in ethanol, followed by acidification to pH 2 to isolate the final 3-oxetanecarboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented synthesis offers compelling advantages that directly address the pain points of cost volatility and supply insecurity prevalent in the fine chemical sector. By shifting the raw material base to commodity chemicals, the process insulates buyers from the price fluctuations associated with specialized reagents, leading to substantial cost savings over the lifecycle of a drug product. Moreover, the elimination of hazardous reagents reduces the regulatory burden and safety infrastructure costs required at the manufacturing site, further enhancing the economic viability of the supply chain. The robustness of the reaction conditions ensures that production can be maintained consistently, reducing the risk of batch failures that often lead to delays in project timelines and increased inventory holding costs for pharmaceutical companies seeking to secure their pipeline against market uncertainties.
- Cost Reduction in Manufacturing: The utilization of low-priced starting materials such as 3-hydroxy-2-(hydroxymethyl) methyl propionate serves as a primary driver for lowering the overall cost of goods sold. Unlike traditional methods that may require expensive catalysts or cryogenic conditions, this route operates under mild thermal conditions using standard reagents like triethylamine and potassium hydroxide, which are available in bulk quantities at competitive market rates. The high yield profile, with individual steps exceeding 80% efficiency, minimizes raw material waste and reduces the volume of solvent required per kilogram of product, thereby lowering both material and waste disposal expenses significantly for large-scale operations.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals ensures a resilient supply chain that is less susceptible to geopolitical disruptions or single-source supplier failures. The process has been validated at the pilot plant scale, processing kilogram quantities of starting material with consistent results, which demonstrates its readiness for immediate commercial scale-up of complex pharmaceutical intermediates. This scalability assures procurement managers that suppliers can ramp up production volumes rapidly to meet sudden spikes in demand without the lengthy lead times typically associated with developing new manufacturing processes from scratch.
- Scalability and Environmental Compliance: The synthetic pathway is designed with green chemistry principles in mind, avoiding the generation of toxic byproducts and minimizing the use of hazardous solvents where possible. The workup procedures involve standard aqueous extractions and pH adjustments, which are easily managed in existing wastewater treatment facilities, ensuring compliance with stringent environmental regulations. This environmental compatibility not only mitigates regulatory risk but also aligns with the sustainability goals of modern pharmaceutical companies, making it a preferred choice for sourcing eco-friendly chemical intermediates that support corporate social responsibility initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-oxetanecarboxylic acid based on the disclosed patent technology. These insights are derived from the experimental data and beneficial effects described in the intellectual property documentation, providing clarity on the feasibility and advantages of adopting this synthetic route. Understanding these details is essential for technical teams evaluating the integration of this intermediate into their drug discovery workflows or supply chain strategies.
Q: What are the safety advantages of this synthetic route compared to traditional methods?
A: This patented method avoids the use of highly toxic or explosive reagents often associated with oxetane ring formation. By utilizing stable starting materials like 3-hydroxy-2-(hydroxymethyl) methyl propionate and standard sulfonyl chlorides, the process significantly reduces operational hazards and simplifies waste treatment protocols.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability through pilot plant examples processing 1kg of starting material. The use of common solvents like dichloromethane and ethanol, along with robust inorganic bases, ensures that the reaction conditions are easily transferable from laboratory bench to multi-ton commercial manufacturing without complex engineering controls.
Q: What is the expected purity and yield profile for this intermediate?
A: The documented process achieves an overall yield exceeding 80%, with individual step yields reaching up to 87.5% for the cyclization step. The purification strategy involves standard aqueous workups and crystallization or extraction techniques, which are highly effective at removing organic impurities, ensuring the final acid meets stringent pharmaceutical quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Oxetanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating the journey from molecule to medicine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless and efficient. We are committed to delivering 3-oxetanecarboxylic acid with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our capability to implement the safe and cost-effective synthesis described in patent CN108727309B allows us to offer a stable supply of this valuable building block, empowering our partners to focus on innovation while we manage the complexities of chemical manufacturing.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. By leveraging our manufacturing expertise, we can help you optimize your supply chain for better margins and reliability. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to quality and efficiency can become a cornerstone of your successful drug development program.
