Advanced Metal-Free Synthesis of Phosphorous Glycine Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for synthesizing complex organic molecules, particularly those containing phosphorus-carbon bonds which are critical in modern drug design. Patent CN113861239A introduces a groundbreaking methodology for the synthesis of phosphorous glycine derivatives, specifically targeting the production of α-phosphinyl amino acids which serve as vital building blocks in medicinal chemistry. This innovation represents a significant departure from traditional transition-metal catalyzed cross-coupling reactions, offering a green, metal-free alternative that operates under ambient conditions. By utilizing visible light induction, specifically blue LED irradiation, this process eliminates the need for hazardous oxidants and expensive metal catalysts, thereby addressing critical purity concerns for R&D directors while simultaneously offering substantial cost reduction in pharmaceutical intermediate manufacturing. The technology demonstrates exceptional substrate tolerance, accommodating various substituents on the aromatic rings, which ensures high versatility for diverse synthetic applications. For supply chain leaders, the simplicity of the reaction conditions translates directly into enhanced supply chain reliability and reduced operational complexity, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships aiming for long-term stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-P bonds in amino acid derivatives has relied heavily on transition metal catalysis, often employing salts of copper or cobalt alongside stoichiometric amounts of peroxide oxidants. As illustrated in the prior art reaction schemes, these conventional methods typically require elevated temperatures, often reaching 80°C or higher, and necessitate strict anhydrous or inert atmosphere conditions to prevent catalyst deactivation. 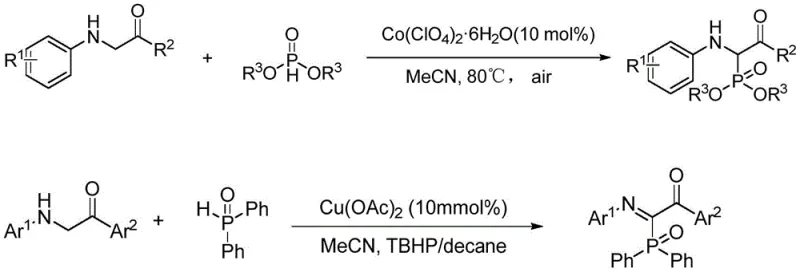 This reliance on heavy metals introduces a significant bottleneck for pharmaceutical applications, as residual metal impurities must be rigorously removed to meet stringent regulatory standards, adding costly purification steps such as chelation or specialized chromatography. Furthermore, the use of external oxidants like TBHP (tert-Butyl hydroperoxide) poses safety risks during scale-up due to their potential instability and exothermic decomposition profiles. The combination of high energy consumption for heating, the expense of metal catalysts, and the complex downstream processing required to ensure product purity creates a substantial economic burden. For procurement managers, these factors result in higher raw material costs and longer lead times, while for R&D teams, the presence of metal residues can complicate biological testing and formulation development, limiting the utility of the final intermediate in sensitive drug discovery programs.
This reliance on heavy metals introduces a significant bottleneck for pharmaceutical applications, as residual metal impurities must be rigorously removed to meet stringent regulatory standards, adding costly purification steps such as chelation or specialized chromatography. Furthermore, the use of external oxidants like TBHP (tert-Butyl hydroperoxide) poses safety risks during scale-up due to their potential instability and exothermic decomposition profiles. The combination of high energy consumption for heating, the expense of metal catalysts, and the complex downstream processing required to ensure product purity creates a substantial economic burden. For procurement managers, these factors result in higher raw material costs and longer lead times, while for R&D teams, the presence of metal residues can complicate biological testing and formulation development, limiting the utility of the final intermediate in sensitive drug discovery programs.
The Novel Approach
In stark contrast to the harsh conditions of the past, the novel method disclosed in the patent utilizes a photo-induced radical mechanism that proceeds efficiently at room temperature without any metal catalyst. 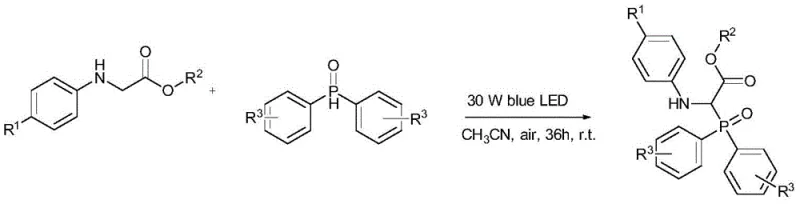 This approach leverages the energy from 30W to 34W blue LEDs to activate the diphenylphosphine oxide, enabling a direct coupling with glycine ester derivatives in a single step. The reaction is conducted in common solvents like acetonitrile under an air atmosphere, completely removing the need for inert gas protection or specialized pressure vessels. This drastic simplification of the reaction setup not only lowers the barrier to entry for production but also significantly enhances the safety profile of the manufacturing process. By operating at 25°C, the energy footprint is minimized, and the risk of thermal runaway is virtually eliminated. The absence of metal catalysts means the resulting α-phosphinyl amino acids are inherently cleaner, reducing the burden on quality control laboratories and accelerating the time from synthesis to application. This paradigm shift offers a compelling value proposition for cost reduction in electronic chemical manufacturing and pharmaceutical sectors alike, where purity and process safety are paramount.
This approach leverages the energy from 30W to 34W blue LEDs to activate the diphenylphosphine oxide, enabling a direct coupling with glycine ester derivatives in a single step. The reaction is conducted in common solvents like acetonitrile under an air atmosphere, completely removing the need for inert gas protection or specialized pressure vessels. This drastic simplification of the reaction setup not only lowers the barrier to entry for production but also significantly enhances the safety profile of the manufacturing process. By operating at 25°C, the energy footprint is minimized, and the risk of thermal runaway is virtually eliminated. The absence of metal catalysts means the resulting α-phosphinyl amino acids are inherently cleaner, reducing the burden on quality control laboratories and accelerating the time from synthesis to application. This paradigm shift offers a compelling value proposition for cost reduction in electronic chemical manufacturing and pharmaceutical sectors alike, where purity and process safety are paramount.
Mechanistic Insights into Photo-Induced C-P Bond Formation
The core of this technological advancement lies in the unique mechanistic pathway that bypasses the need for transition metal mediation. Under blue LED irradiation, the diphenylphosphine oxide undergoes homolytic cleavage or excitation to generate phosphorus-centered radicals, which then attack the electron-deficient center of the glycine ester derivative. 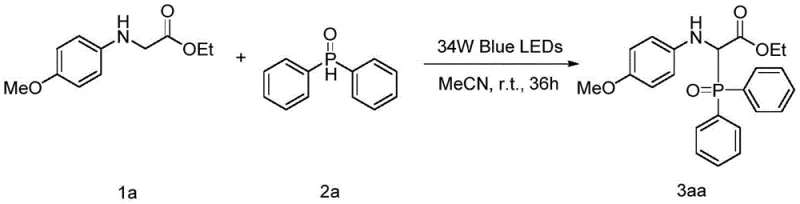 This radical addition is highly selective and proceeds with excellent functional group tolerance, as evidenced by the successful synthesis of derivatives containing electron-donating groups like methoxy and electron-withdrawing groups like chloro and bromo. The mechanism avoids the formation of metal-complex intermediates, which are often the source of side reactions and impurity generation in traditional catalysis. Instead, the reaction relies on the intrinsic reactivity of the excited state species, driven by the continuous input of photon energy. This ensures a consistent reaction rate and high conversion efficiency, with reported yields ranging from 70% to 85% across various substrates. For R&D directors, understanding this mechanism is crucial as it highlights the robustness of the process against variations in substrate electronics, ensuring that the synthetic route remains viable even when structural modifications are required for SAR (Structure-Activity Relationship) studies.
This radical addition is highly selective and proceeds with excellent functional group tolerance, as evidenced by the successful synthesis of derivatives containing electron-donating groups like methoxy and electron-withdrawing groups like chloro and bromo. The mechanism avoids the formation of metal-complex intermediates, which are often the source of side reactions and impurity generation in traditional catalysis. Instead, the reaction relies on the intrinsic reactivity of the excited state species, driven by the continuous input of photon energy. This ensures a consistent reaction rate and high conversion efficiency, with reported yields ranging from 70% to 85% across various substrates. For R&D directors, understanding this mechanism is crucial as it highlights the robustness of the process against variations in substrate electronics, ensuring that the synthetic route remains viable even when structural modifications are required for SAR (Structure-Activity Relationship) studies.
Furthermore, the impurity profile of this metal-free process is significantly cleaner compared to metal-catalyzed counterparts. Without the presence of cobalt or copper ions, there is no risk of metal-induced oxidation of sensitive functional groups or the formation of metal-organic byproducts that are difficult to separate. The primary byproducts are minimal, and the reaction mixture can often be purified using standard silica gel chromatography with common eluent systems like petroleum ether and ethyl acetate. This simplicity in purification translates to higher overall recovery rates and reduced solvent consumption, aligning with green chemistry principles. The ability to produce high-purity OLED material precursors or pharmaceutical intermediates without the specter of heavy metal contamination is a distinct competitive advantage. It allows downstream processors to skip expensive scavenging steps, thereby streamlining the entire production workflow and ensuring that the final product meets the rigorous specifications required for clinical trial materials and commercial drug substances.
How to Synthesize α-Phosphinyl Amino Acid Efficiently
To implement this synthesis effectively, one must adhere to the specific parameters outlined in the patent to ensure optimal yield and reproducibility. The process begins with the precise weighing of glycine ester derivatives and diphenylphosphine oxide, typically maintaining a molar ratio of 1:2 to drive the reaction to completion. The choice of solvent is critical, with acetonitrile proving to be the most effective medium for dissolving both reactants and facilitating the photo-induced electron transfer. Detailed standardized synthesis steps are provided in the guide below to ensure consistency across different batches and scales.
- Prepare the reaction mixture by combining glycine ester derivatives and diphenylphosphine oxide in acetonitrile solvent at a molar ratio of 1: 2.
- Stir the solution at room temperature (25°C) under atmospheric conditions without the need for inert gas protection.
- Irradiate the reaction with 30W to 34W blue LEDs for 36 hours, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers transformative benefits that directly impact the bottom line and operational resilience of chemical manufacturing enterprises. The elimination of transition metal catalysts removes a significant cost center, as high-purity metal salts and the ligands often required for them are expensive and subject to market volatility.
- Cost Reduction in Manufacturing: The removal of metal catalysts and external oxidants drastically simplifies the bill of materials, leading to substantial cost savings in raw material procurement. Without the need for expensive metal scavengers or complex purification protocols to remove trace metals, the downstream processing costs are significantly reduced. This leaner process flow allows for better margin protection and more competitive pricing strategies in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Operating at room temperature and under atmospheric conditions reduces the dependency on specialized equipment such as high-pressure reactors or cryogenic cooling systems. This flexibility allows for production in a wider range of facilities, mitigating the risk of bottlenecks caused by equipment shortages or maintenance downtime. The use of commercially available and stable reagents ensures a steady supply of inputs, reducing lead time for high-purity pharmaceutical intermediates and enhancing overall supply chain continuity.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of hazardous oxidants make this process inherently safer and easier to scale from laboratory to commercial production. The reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations, lowering the costs associated with waste treatment and disposal. This environmental compatibility not only safeguards the company's regulatory standing but also appeals to eco-conscious partners and clients seeking sustainable supply chain solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photo-induced synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers.
Q: What are the primary advantages of this metal-free synthesis method over traditional catalytic approaches?
A: The primary advantage is the elimination of transition metal catalysts such as cobalt or copper, which removes the risk of heavy metal residues in the final pharmaceutical intermediate. Additionally, the reaction proceeds at room temperature without external oxidants, significantly simplifying the workup process and reducing safety hazards associated with high-temperature operations.
Q: What are the typical yield ranges and reaction conditions for this process?
A: The process typically achieves yields between 70% and 85% under mild conditions. The reaction is conducted at room temperature (25°C) in acetonitrile solvent using blue LED irradiation for approximately 36 hours, requiring no special pressure equipment or inert atmosphere.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly suitable for scale-up due to its operational simplicity. It utilizes commercially available reagents, avoids hazardous oxidants, and does not require complex temperature control systems, making it economically viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorous Glycine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art photo-reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of phosphorous glycine derivative meets the highest international standards. We understand the critical nature of supply continuity for our clients and have optimized our workflows to deliver consistent quality and reliability.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green chemistry approach can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the market with our high-purity OLED material and pharmaceutical intermediate solutions.
