Advanced Manufacturing of High-Purity R-Budesonide via Optimized Prednisolone Cyclization
Introduction to Advanced Corticosteroid Manufacturing
The pharmaceutical landscape for respiratory and dermatological treatments has increasingly shifted towards single-isomer glucocorticoids due to their superior therapeutic indices and reduced systemic side effects. Patent CN103724396A introduces a robust and industrially viable preparation method for R-budesonide, also known as dexbudesonide, which exhibits significantly higher anti-inflammatory activity compared to its S-isomer counterpart. This technical disclosure addresses critical bottlenecks in existing manufacturing protocols by utilizing prednisolone as a stable and accessible starting material, thereby streamlining the supply chain for this high-value active pharmaceutical ingredient. By optimizing the cyclization and purification stages, the disclosed methodology not only enhances the overall process yield but also mitigates the environmental and safety hazards associated with traditional heavy metal oxidants. For global procurement teams and R&D directors, this represents a pivotal opportunity to secure a more reliable R-budesonide supplier capable of delivering consistent quality at a competitive operational cost.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dexbudesonide has been plagued by inefficient reaction pathways that rely on hazardous reagents and deliver suboptimal yields. Prior art, such as the method described in patent WO 87/05028, utilizes prednisolone acetate as a starting material but necessitates the use of trifluoroacetic anhydride (TFAA) for esterification and osmium tetroxide for oxidation, both of which pose severe safety and cost challenges. The reliance on osmium tetroxide introduces significant toxicity risks to operators and requires complex waste treatment protocols, while the use of TFAA drastically inflates raw material expenses. Furthermore, the total recovery rate of these legacy processes is notoriously low, often hovering around merely 5%, which results in excessive solvent consumption and a massive carbon footprint per kilogram of final product. The generation of unwanted 11-alpha or beta hydroxyl impurities during sodium borohydride hydrogenation further complicates downstream purification, making commercial scale-up of complex steroid intermediates economically unfeasible for many manufacturers.
The Novel Approach
In stark contrast, the innovative route detailed in CN103724396A circumvents these historical deficiencies by employing a safer and more atom-economical sequence starting directly from prednisolone. This novel approach replaces the toxic osmium tetroxide and expensive TFAA with milder reagents like triethyl orthoacetate and standard oxidants, fundamentally altering the cost structure of the synthesis. The process flow involves a strategic sequence of cyclization, ring opening, esterification, elimination, and oxidation, culminating in a high-yield formation of the target budesonide acetate intermediate. As illustrated in the comprehensive reaction scheme below, the pathway is designed to maximize the conversion efficiency at each step, ultimately achieving a total recovery rate of approximately 20%, which is a four-fold improvement over previous benchmarks. This dramatic increase in yield, combined with the elimination of hazardous heavy metals, positions this method as a superior choice for cost reduction in pharmaceutical manufacturing and ensures a more sustainable production lifecycle.
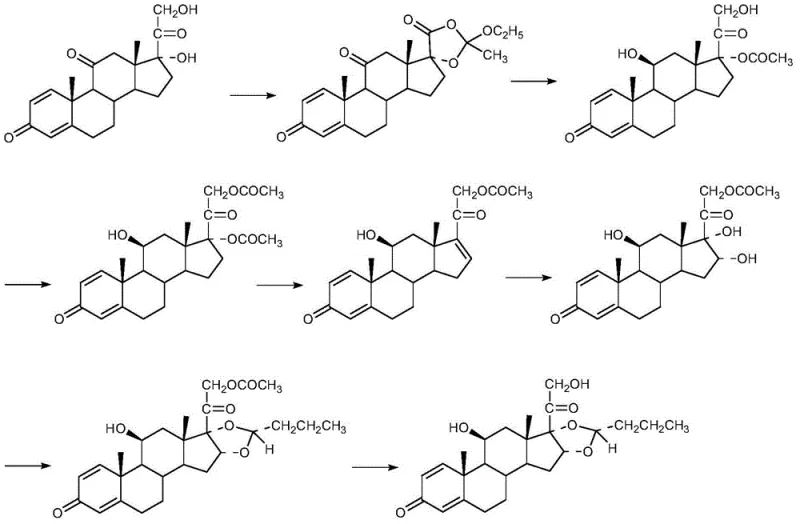
Mechanistic Insights into Triethyl Orthoacetate Cyclization and Isomer Separation
The core chemical innovation of this process lies in the initial cyclization step where prednisolone reacts with triethyl orthoacetate to form the key intermediate compound (II), setting the stereochemical foundation for the subsequent transformations. This reaction is carefully controlled to ensure the correct orientation of the side chain, which is critical for the biological activity of the final R-isomer. Following the formation of compound (II), the process proceeds through a hydrolysis step to open the ring, generating compound (III), which is then subjected to acetylation to protect specific hydroxyl groups during the elimination phase. The elimination reaction under alkaline conditions creates the necessary double bond in the D-ring, forming compound (V), which is subsequently oxidized to introduce the oxygen functionality required for the final acetonide ring closure. Each of these mechanistic steps is optimized to minimize side reactions and impurity formation, ensuring that the crude product entering the purification stage is of sufficiently high quality to allow for effective separation.
A critical aspect of this synthesis is the management of stereoisomers, particularly at the C-22 position, where the R and S isomers are generated. The process generates a mixture of isomers (VII), which must be meticulously separated to isolate the pharmacologically active R-budesonide (VIIa) from the less active S-isomer (VIIb). The patent discloses a refined fractional crystallization technique using alcohols such as methanol or ethanol, which exploits the subtle solubility differences between the diastereomers to achieve high purity. As shown in the structural comparison below, the spatial arrangement of the propyl side chain differs between the isomers, and the purification protocol is specifically tuned to enrich the R-configuration to levels exceeding 99.0wt%. This rigorous control over the impurity profile is essential for meeting the stringent regulatory requirements for hormonal drugs and ensures that the final API possesses the desired potency without the burden of inactive or potentially antagonistic isomeric impurities.
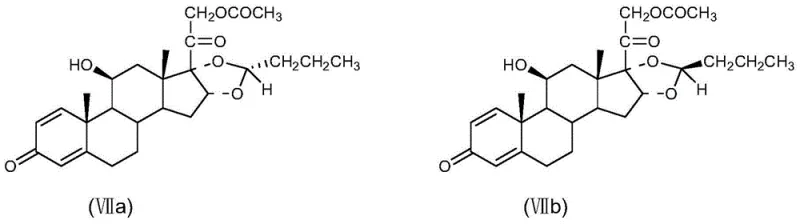
How to Synthesize R-Budesonide Efficiently
The execution of this synthesis requires precise control over reaction parameters such as temperature, pH, and solvent ratios to ensure reproducibility and high yield. The process begins with the cyclization of prednisolone, followed by a series of functional group manipulations that build the complexity of the molecule while maintaining stereochemical integrity. Operators must pay close attention to the crystallization conditions during the separation of isomers VIIa and VIIb, as this step is the primary determinant of the final optical purity. The standardized synthetic steps outlined in the patent provide a clear roadmap for scaling this chemistry from laboratory benchtop to pilot plant operations, minimizing the risk of batch failures. For detailed operational parameters and specific stoichiometric ratios, please refer to the technical guide below.
- Cyclize prednisolone with triethyl orthoacetate to form the intermediate compound (II).
- Perform hydrolysis to open the ring, followed by esterification with acetic anhydride to obtain compound (IV).
- Execute alkaline elimination and oxidation reactions to generate the isomeric mixture (VII), followed by fractional crystallization to isolate the R-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for procurement managers and supply chain directors seeking to optimize their steroid intermediate sourcing strategies. The elimination of osmium tetroxide and trifluoroacetic anhydride removes two of the most volatile cost drivers and safety liabilities from the supply chain, leading to a more predictable and stable manufacturing environment. By shifting to a process that utilizes readily available prednisolone and common organic solvents, manufacturers can significantly reduce their dependency on specialized and hazardous reagent suppliers, thereby enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates. The simplified waste treatment requirements, resulting from the absence of heavy metals, further contribute to operational efficiency and lower environmental compliance costs, making this route highly attractive for large-scale production facilities.
- Cost Reduction in Manufacturing: The transition to this novel synthetic pathway facilitates substantial cost savings by removing the need for expensive and toxic reagents like osmium tetroxide and TFAA, which historically inflated the bill of materials for dexbudesonide production. The quadrupling of the overall process yield from 5% to 20% means that significantly less raw material and solvent are required to produce the same amount of final product, drastically lowering the variable cost per kilogram. Additionally, the reduced complexity of the waste stream allows for simpler and cheaper effluent treatment processes, further contributing to the overall economic viability of the project without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing prednisolone as the starting material leverages a well-established and abundant supply base, ensuring that production schedules are not disrupted by the scarcity of exotic precursors. The robustness of the reaction conditions, which avoid extreme temperatures or pressures, minimizes the risk of unplanned downtime due to equipment failure or safety incidents, guaranteeing a continuous flow of goods to downstream customers. This stability is crucial for maintaining long-term contracts with pharmaceutical companies that require consistent quality and on-time delivery for their respiratory medication portfolios.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial production, with steps that are easily transferable from pilot scale to multi-ton commercial manufacturing without loss of efficiency. The avoidance of persistent heavy metal contaminants simplifies the regulatory approval process for new drug master files (DMFs) and aligns with increasingly strict global environmental regulations regarding chemical manufacturing. This forward-looking approach ensures that the production facility remains compliant with future sustainability mandates, protecting the long-term value of the manufacturing asset and securing its place in the green chemistry supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this R-budesonide synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement specialists assessing supplier capabilities.
Q: What are the primary advantages of the CN103724396A synthesis method over prior art?
A: The method described in CN103724396A significantly improves total yield to approximately 20%, compared to only 5% in older methods like WO 87/05028. Furthermore, it eliminates the need for highly toxic osmium tetroxide and expensive trifluoroacetic anhydride, reducing both safety risks and raw material costs.
Q: What is the starting material for this R-budesonide preparation?
A: The synthesis utilizes prednisolone as the primary starting raw material, which undergoes a series of transformations including cyclization with triethyl orthoacetate to build the necessary steroid framework.
Q: What purity levels can be achieved with this purification process?
A: Through optimized fractional crystallization and hydrolysis steps, the process achieves an R-isomer purity greater than 99.0wt%, with preferred embodiments reaching exceeding 99.5wt%, ensuring high quality for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-Budesonide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe manufacturing processes for high-value hormonal intermediates like R-budesonide. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103724396A are fully realized in practical, large-volume operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of corticosteroid intermediate meets the highest international standards for potency and impurity profiles. Our commitment to technological excellence allows us to offer partners a secure and high-quality supply source that can adapt to fluctuating market demands while maintaining consistent product integrity.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your respiratory drug manufacturing programs.
