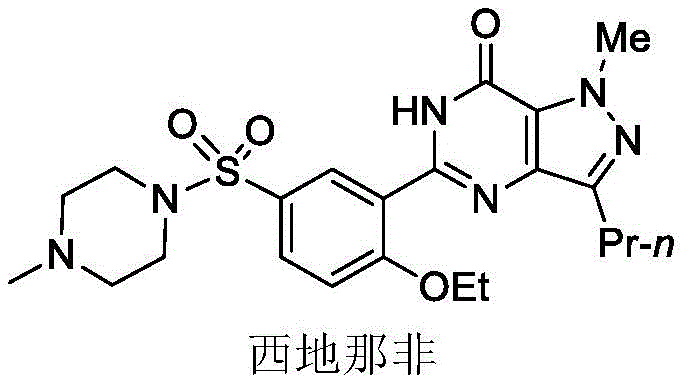
Revolutionizing Sildenafil Intermediate Synthesis via Scalable Electroreduction Technology
Revolutionizing Sildenafil Intermediate Synthesis via Scalable Electroreduction Technology
The pharmaceutical industry is currently undergoing a paradigm shift towards greener, more sustainable manufacturing processes, particularly for high-volume Active Pharmaceutical Ingredients (APIs) like Sildenafil. A pivotal advancement in this domain is detailed in patent CN111005033A, which discloses a novel electroreduction preparation method for 1-methyl-3-propyl-4-aminopyrazole-5-formamide, a critical intermediate in the Sildenafil value chain. This technology replaces traditional, hazardous chemical reduction steps with a clean, controllable electrochemical process, utilizing electrons as the primary reducing agent. By leveraging a diaphragm electrolytic cell system, this method achieves exceptional conversion rates and selectivity while completely avoiding the use of toxic reducing agents such as hydrazine or heavy metal catalysts. For global procurement and R&D teams, this represents a significant opportunity to optimize supply chains, reduce environmental liabilities, and ensure higher purity profiles for the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-methyl-3-propyl-4-aminopyrazole-5-formamide has relied on several chemically intensive reduction strategies, each carrying substantial operational and environmental burdens. Traditional routes often employ stannous chloride (SnCl2) as a reducing agent; however, this approach is plagued by the high cost of the reagent and the formation of tin-complexes that are notoriously difficult to separate from the product, thereby compromising purity. Alternatively, iron powder reduction generates massive quantities of iron sludge, creating severe waste disposal challenges and environmental pollution issues that conflict with modern green chemistry principles. Furthermore, methods utilizing hydrazine hydrate introduce significant safety hazards due to its carcinogenicity and genotoxicity, requiring rigorous and costly containment measures. Even catalytic hydrogenation, while effective, risks leaving residual palladium or nickel in the product, necessitating expensive metal scavenging steps to meet strict regulatory limits for heavy metals in pharmaceuticals.
The Novel Approach
In stark contrast, the electroreduction method described in CN111005033A offers a transformative solution by substituting chemical reductants with electrical energy. This process operates within a diaphragm electrolytic cell where the nitro-group of the precursor is selectively reduced to an amino group at the cathode. The reaction conditions are remarkably mild, typically running between 25°C and 80°C, and the system utilizes inexpensive inorganic ammonium salts as electrolytes. As illustrated in the reaction scheme below, the transformation is direct and efficient, bypassing the complex workup procedures associated with metal removal. This not only streamlines the manufacturing workflow but also fundamentally alters the impurity profile, eliminating the risk of heavy metal contamination and toxic organic residues associated with hydrazine or tin byproducts.
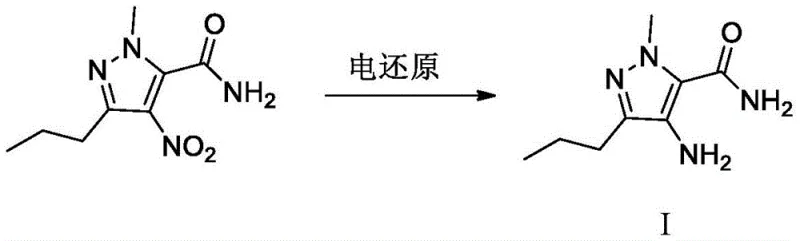
Mechanistic Insights into Diaphragm Electrolytic Reduction
The core mechanism of this innovation lies in the controlled transfer of electrons at the cathode surface within a divided cell configuration. In this setup, the catholyte consists of the nitro-pyrazole substrate dissolved in a mixed solvent system of organic alcohol and water, buffered with inorganic ammonium salts which serve as the proton source and conductivity enhancer. When a potential of 1.20V to 2.50V is applied relative to a reference electrode, the nitro group undergoes a multi-electron reduction sequence. The presence of the diaphragm (specifically a strong acid cation exchange membrane) is critical, as it prevents the oxidation of the newly formed amine at the anode and separates the anodic and cathodic reactions, thereby maximizing current efficiency. The use of robust electrode materials, such as red copper, nickel, or titanium-based platinum, ensures stable performance over extended operation cycles without significant degradation or leaching.
From an impurity control perspective, this electrochemical mechanism offers superior selectivity compared to chemical reduction. In chemical methods, over-reduction or side reactions with the reducing agent can generate complex byproduct mixtures. However, in electroreduction, the reaction rate and selectivity are precisely tunable by adjusting the electrode potential and current density (25.0 mA/cm² to 250.0 mA/cm²). This precise control minimizes the formation of hydroxylamine intermediates or azo-coupling byproducts, leading to a cleaner crude product. Consequently, the downstream purification process is significantly simplified, often requiring only standard extraction and recrystallization to achieve high-purity specifications suitable for API synthesis, thus enhancing the overall robustness of the manufacturing process.
How to Synthesize 1-Methyl-3-propyl-4-aminopyrazole-5-formamide Efficiently
Implementing this electrochemical route requires careful attention to cell configuration and electrolyte composition to maximize yield and safety. The process begins with the preparation of a homogeneous catholyte by dissolving the nitro-precursor in solvents like ethanol or isopropanol, mixed with an aqueous ammonium salt solution. The anolyte typically contains dilute sulfuric acid or ammonium sulfate to maintain conductivity and balance charge transfer. Operationally, the cell is maintained at moderate temperatures (e.g., 40°C) with constant stirring to ensure mass transfer to the electrode surface. The reaction progress is monitored by charge consumption or HPLC analysis, and upon completion, the product is isolated via organic extraction. For a detailed, step-by-step breakdown of the experimental parameters and specific equipment setups described in the patent, please refer to the standardized synthesis guide below.
- Prepare the catholyte by dissolving 1-methyl-3-propyl-4-nitropyrazole-5-formamide in an organic solvent (e.g., ethanol or isopropanol) and mixing with an aqueous solution of inorganic ammonium salt.
- Assemble the diaphragm electrolytic cell with a suitable cathode (e.g., red copper or nickel) and an anode (e.g., DSA or platinum mesh), ensuring the anolyte contains sulfuric acid or ammonium sulfate.
- Apply a constant voltage between 1.20V and 2.50V relative to the reference electrode at a temperature of 25-80°C until the reduction is complete, followed by extraction and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electroreduction technology translates into tangible strategic advantages beyond mere technical novelty. The elimination of expensive and hazardous chemical reagents directly impacts the Bill of Materials (BOM), reducing dependency on volatile commodity markets for metals like palladium or tin. Furthermore, the simplification of the workup process—removing the need for complex metal scavenging resins or extensive sludge treatment—drastically reduces processing time and waste disposal costs. This streamlined workflow enhances the overall agility of the supply chain, allowing for faster batch turnover and more reliable delivery schedules for key intermediates.
- Cost Reduction in Manufacturing: The electrochemical process fundamentally lowers production costs by removing the need for stoichiometric amounts of expensive reducing agents like stannous chloride or precious metal catalysts. Since electrons serve as the reagent, the variable cost associated with chemical consumption is significantly minimized. Additionally, the avoidance of heavy metal catalysts eliminates the capital and operational expenditure related to metal recovery systems and stringent residue testing, leading to substantial overall cost savings in the manufacturing of pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By relying on electricity and common inorganic salts rather than specialized, potentially supply-constrained chemical reductants, the process mitigates supply chain risks. The raw materials required for the electrolyte are commodity chemicals with stable availability, ensuring consistent production continuity. Moreover, the milder reaction conditions reduce the likelihood of safety-related shutdowns or regulatory interventions, providing a more predictable and secure supply of high-purity intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable through the addition of electrode surface area or the use of flow cells, making this technology ideal for commercial scale-up of complex pharmaceutical intermediates. From an environmental standpoint, the process aligns perfectly with green chemistry mandates by generating minimal hazardous waste and avoiding toxic emissions. This compliance reduces the environmental tax burden and simplifies the permitting process for new manufacturing facilities, future-proofing the supply chain against tightening global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electroreduction technology. These insights are derived directly from the experimental data and beneficial effects reported in patent CN111005033A, providing a clear understanding of how this method compares to legacy synthetic routes. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this green chemistry approach into their existing manufacturing portfolios.
Q: How does electroreduction improve the purity profile compared to catalytic hydrogenation?
A: Unlike catalytic hydrogenation which often leaves trace heavy metal residues (Pd, Ni) that form difficult-to-separate complexes, electroreduction uses electrons as the primary reagent. This eliminates the risk of heavy metal contamination exceeding pharmacopeial limits and simplifies downstream purification.
Q: What are the safety advantages of this method over traditional chemical reduction?
A: Traditional methods often rely on hazardous reagents like stannous chloride (toxic, hard to remove), iron powder (generates massive sludge), or hydrazine hydrate (carcinogenic). The electrochemical method operates under mild conditions (25-80°C) without these dangerous chemicals, significantly reducing HSE risks.
Q: Is this electrochemical process scalable for industrial API production?
A: Yes, the patent demonstrates high yields (up to 99%) in a diaphragm cell setup which is inherently scalable. The use of robust electrode materials like DSA and titanium-based platinum ensures long operational life suitable for continuous or large-batch manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Methyl-3-propyl-4-aminopyrazole-5-formamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern pharmaceutical manufacturing. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory techniques like the one described in CN111005033A are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced electrochemical reactors capable of maintaining stringent purity specifications, guaranteeing that every batch of intermediate meets the highest global regulatory standards for safety and efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and environmentally friendly technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in green chemistry can optimize your supply chain and enhance your competitive advantage in the global market.
