Advanced Synthesis of Milobalin Intermediate via Asymmetric Acetal Resolution
Introduction to the Novel Preparation Method
The pharmaceutical landscape for treating Peripheral Neuropathic Pain (PNP) has been significantly advanced by the approval of Milobalin, a therapeutic agent developed for conditions such as Diabetic Peripheral Neuropathic Pain (DPNP) and postherpetic neuralgia. 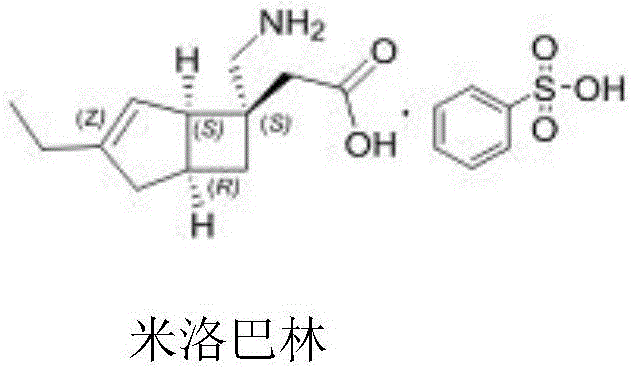 Central to the commercial viability of this drug is the efficient synthesis of its key chiral intermediate, (1R,5S)-3-ethylbicyclo[3.2.0]hept-3-en-6-one. Patent CN115745764A discloses a groundbreaking preparation method that addresses critical bottlenecks in the supply chain of this high-value pharmaceutical intermediate. By leveraging an asymmetric acetal reaction strategy, this technology offers a robust alternative to traditional resolution techniques, promising enhanced stereochemical control and operational simplicity. For global procurement teams and R&D directors, understanding this technological shift is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent purity specifications while optimizing manufacturing economics.
Central to the commercial viability of this drug is the efficient synthesis of its key chiral intermediate, (1R,5S)-3-ethylbicyclo[3.2.0]hept-3-en-6-one. Patent CN115745764A discloses a groundbreaking preparation method that addresses critical bottlenecks in the supply chain of this high-value pharmaceutical intermediate. By leveraging an asymmetric acetal reaction strategy, this technology offers a robust alternative to traditional resolution techniques, promising enhanced stereochemical control and operational simplicity. For global procurement teams and R&D directors, understanding this technological shift is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent purity specifications while optimizing manufacturing economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of the Milobalin intermediate has been plagued by inefficiencies inherent in older resolution technologies. Prior art, such as that described in patents CN10424595 and CN103987687, relied heavily on chromatographic resolution or the use of expensive chiral amine catalysts. These conventional approaches suffer from inherently low production efficiency, often capping yields at around 50%, which effectively doubles the material cost per unit of active product. Furthermore, methods utilizing chiral amines introduce significant supply chain risks due to the high cost and limited availability of these specialized catalysts. Biological resolution methods, while selective, impose rigorous demands on production equipment and environmental controls, rendering them unsuitable for standard chemical manufacturing facilities. The cumulative effect of these limitations is a high-cost, low-yield process that hinders the broader commercialization of Milobalin-based therapies.
The Novel Approach
The methodology outlined in Patent CN115745764A represents a paradigm shift by employing an asymmetric acetal reaction using readily available chiral alcohols. 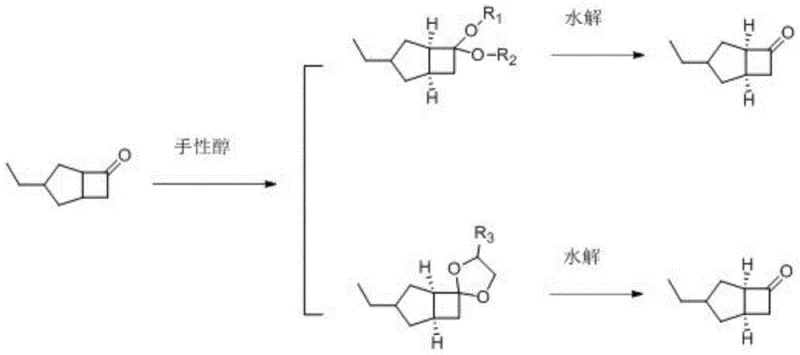 This novel route transforms the racemic ketone substrate into a chiral acetal derivative, which can be easily purified through crystallization before being hydrolyzed back to the target ketone with superior optical purity. The patent data indicates a dramatic improvement in process metrics, with resolution yields increasing to approximately 70% and optical purity (e.e.%) exceeding 99.9%. By replacing scarce chiral amines with abundant chiral alcohols, the process not only simplifies the synthetic pathway but also drastically reduces the raw material burden. This approach effectively bridges the gap between laboratory feasibility and industrial scalability, offering a streamlined solution for cost reduction in API manufacturing.
This novel route transforms the racemic ketone substrate into a chiral acetal derivative, which can be easily purified through crystallization before being hydrolyzed back to the target ketone with superior optical purity. The patent data indicates a dramatic improvement in process metrics, with resolution yields increasing to approximately 70% and optical purity (e.e.%) exceeding 99.9%. By replacing scarce chiral amines with abundant chiral alcohols, the process not only simplifies the synthetic pathway but also drastically reduces the raw material burden. This approach effectively bridges the gap between laboratory feasibility and industrial scalability, offering a streamlined solution for cost reduction in API manufacturing.
Mechanistic Insights into Asymmetric Acetal Resolution
The core of this innovative synthesis lies in the stereoselective formation of acetals. The process begins with the reaction of 3-ethylbicyclo[3.2.0]hept-3-en-6-one with a chiral alcohol in the presence of an acid catalyst, such as dodecylbenzenesulfonic acid or p-toluenesulfonic acid. This step involves the nucleophilic attack of the chiral alcohol on the carbonyl carbon of the ketone, facilitated by azeotropic dehydration in solvents like toluene at temperatures between 70°C and 80°C. The resulting diastereomeric acetals exhibit different physical properties, particularly solubility, allowing for the preferential crystallization of the desired isomer. This crystallization step is crucial as it acts as a powerful purification engine, enriching the stereochemical purity of the intermediate before the final deprotection step. The versatility of this mechanism is underscored by the wide range of compatible chiral alcohols, including monohydric alcohols like (R)-(+)-1-phenylethyl alcohol and polyhydric alcohols like erythritol.
Following the isolation of the chiral acetal, the second critical phase involves acid-catalyzed hydrolysis. 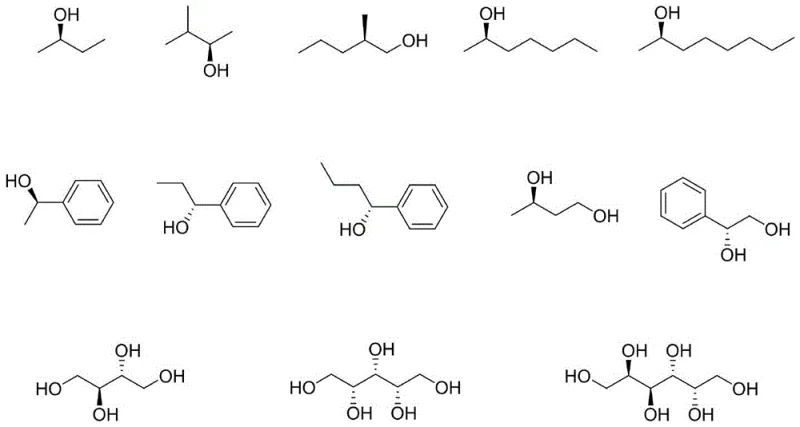 In this step, the acetal protecting group is cleaved using mineral or organic acids such as hydrochloric acid, sulfuric acid, or trifluoroacetic acid in solvents like dichloromethane. This hydrolysis regenerates the carbonyl group of the bicyclic ketone while releasing the chiral alcohol, which can potentially be recovered and recycled, further enhancing the atom economy of the process. The mild conditions of this hydrolysis step ensure that the sensitive bicyclic framework remains intact, preventing racemization or decomposition. The result is a high-purity target molecule with an e.e.% value consistently above 99.9%, demonstrating the robustness of the chiral information transfer from the alcohol auxiliary to the final product. This mechanistic clarity provides R&D teams with a predictable and controllable pathway for scaling complex pharmaceutical intermediates.
In this step, the acetal protecting group is cleaved using mineral or organic acids such as hydrochloric acid, sulfuric acid, or trifluoroacetic acid in solvents like dichloromethane. This hydrolysis regenerates the carbonyl group of the bicyclic ketone while releasing the chiral alcohol, which can potentially be recovered and recycled, further enhancing the atom economy of the process. The mild conditions of this hydrolysis step ensure that the sensitive bicyclic framework remains intact, preventing racemization or decomposition. The result is a high-purity target molecule with an e.e.% value consistently above 99.9%, demonstrating the robustness of the chiral information transfer from the alcohol auxiliary to the final product. This mechanistic clarity provides R&D teams with a predictable and controllable pathway for scaling complex pharmaceutical intermediates.
How to Synthesize (1R,5S)-3-ethylbicyclo[3.2.0]hept-3-en-6-one Efficiently
The synthesis protocol described in the patent offers a clear, two-stage workflow that balances high yield with operational ease. The process initiates with the condensation of the racemic ketone and the chosen chiral alcohol under reflux conditions to drive water removal, followed by a workup involving neutralization and crystallization to isolate the pure acetal. The subsequent hydrolysis step is performed at ambient temperature, minimizing energy consumption and thermal stress on the product. This standardized approach allows for precise replication across different manufacturing sites, ensuring consistent quality output essential for regulatory compliance in the pharmaceutical sector.
- Perform asymmetric acetal reaction by mixing 3-ethylbicyclo[3.2.0]hept-3-en-6-one with a chiral alcohol (e.g., (R)-(+)-1-phenylethyl alcohol) in a solvent like toluene with an acid catalyst, heating to 70-80°C for azeotropic dehydration.
- Isolate the chiral acetal product by cooling, neutralizing with dilute alkali, crystallizing, and filtering the solid precipitate.
- Hydrolyze the chiral acetal product using an acid (e.g., trifluoroacetic acid or hydrochloric acid) in a solvent like dichloromethane at room temperature to remove the protecting group and obtain the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this asymmetric acetal methodology offers tangible strategic benefits beyond mere technical superiority. The primary advantage lies in the substantial optimization of raw material costs. By utilizing chiral alcohols which are reported to be approximately 60% lower in average price compared to traditional chiral amine catalysts, the overall cost of goods sold (COGS) for the intermediate is significantly depressed. Additionally, the increase in resolution yield from 50% to roughly 70% implies a direct reduction in waste generation and raw material consumption per kilogram of final product. This efficiency gain translates into a more competitive pricing structure for the finished API, allowing pharmaceutical companies to better manage their budget allocations for neuropathic pain treatments without compromising on quality standards.
Supply chain reliability is further enhanced by the use of commodity chemicals and standard unit operations. Unlike enzymatic methods that require specialized bioreactors and strict sterile conditions, this chemical resolution can be executed in standard glass-lined or stainless steel reactors using common solvents like toluene and dichloromethane. The availability of diverse chiral alcohol options, ranging from simple secondary alcohols to sugar derivatives like xylitol, mitigates the risk of single-source supplier dependency. This flexibility ensures continuity of supply even in volatile market conditions. Furthermore, the simplicity of the workup procedures—primarily filtration and extraction—reduces the turnaround time between batches, thereby increasing the overall throughput capacity of existing manufacturing facilities without the need for capital-intensive infrastructure upgrades.
From an environmental and scalability perspective, the process aligns well with modern green chemistry principles and regulatory expectations. The ability to recycle the chiral alcohol auxiliary after hydrolysis reduces the chemical footprint of the synthesis. Moreover, the avoidance of heavy metal catalysts or complex biological agents simplifies the impurity profile, making downstream purification and regulatory filing more straightforward. The method's robustness has been demonstrated across various scales in the patent examples, confirming its suitability for commercial scale-up of complex pharmaceutical intermediates. This scalability ensures that as demand for Milobalin grows, the supply of its critical intermediate can be ramped up seamlessly, preventing bottlenecks that could delay product launches or market expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a transparent view of the technology's capabilities. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this intermediate into their broader drug development pipelines.
Q: What are the advantages of this new method over traditional chromatographic resolution?
A: The new asymmetric acetal method improves the yield of chiral resolution to approximately 70% compared to 50% in prior art, and increases optical purity (e.e.%) from about 98% to over 99.9%, significantly reducing production costs and improving product quality.
Q: Why are chiral alcohols preferred over chiral amines in this synthesis?
A: Chiral alcohols used as resolving agents are significantly cheaper, with average prices reported to be about 60% lower than the expensive chiral amine catalysts used in previous methods, facilitating easier industrial adoption.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method uses common solvents and acids, avoids complex biological equipment required for enzymatic methods, and offers simple operation steps, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milobalin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical partners. We are committed to delivering Milobalin intermediates with stringent purity specifications, leveraging advanced analytical capabilities in our rigorous QC labs to guarantee batch-to-batch consistency. Our expertise in chiral resolution and asymmetric synthesis positions us as a strategic partner capable of navigating the complexities of modern API manufacturing.
We invite you to collaborate with us to explore how this advanced synthesis method can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your regulatory filings and production planning. Let us help you secure a stable, cost-effective supply of this vital intermediate for your neuropathic pain therapy programs.
