Advanced Manufacturing of Beta-Methyl Carbapenem Intermediates: A Safe, Scalable Dieckmann Condensation Strategy
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the urgent need for potent agents against drug-resistant bacteria. Patent CN103012471A introduces a transformative methodology for preparing key intermediates of beta-methyl carbapenem antibiotics, a class that includes life-saving drugs like meropenem and doripenem. This invention specifically addresses the longstanding challenges associated with the synthesis of the bicyclic core structure, proposing a streamlined three-step route that bypasses the need for hazardous reagents and precious metal catalysts. By leveraging a modified Dieckmann condensation strategy starting from beta-methyl-ADC-8, the process achieves exceptional purity levels exceeding 95% and robust yields, positioning it as a superior alternative for industrial applications. For R&D directors and procurement specialists, this technology represents a significant leap forward in process safety and economic efficiency, offering a reliable pathway to high-quality pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-methyl carbapenem intermediates has been plagued by significant safety and economic hurdles. Traditional routes, such as those depicted in prior art, frequently rely on the use of sulfonyl azide compounds to generate diazo intermediates, which are inherently unstable and pose severe explosion risks during scale-up. Furthermore, the subsequent cyclization step typically necessitates the use of rhodium (Rh) catalysts to facilitate carbene formation. While effective, rhodium is an extremely expensive precious metal, and its use introduces complex downstream processing requirements to ensure residual metal levels meet stringent regulatory limits for API manufacturing. These factors collectively drive up production costs and create supply chain vulnerabilities, making conventional methods less attractive for large-scale commercialization in a cost-sensitive market.
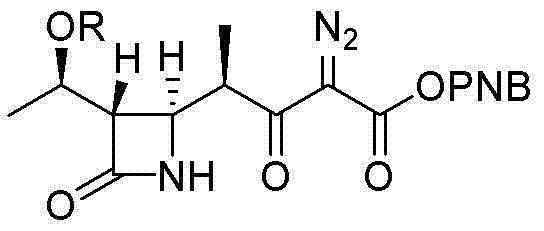
The Novel Approach
In stark contrast, the methodology disclosed in CN103012471A offers a paradigm shift by eliminating both the explosive azide reagents and the rhodium catalyst. The novel approach utilizes a condensation reaction followed by alkylation and a final Dieckmann cyclization, all mediated by inexpensive and readily available reagents such as N,N'-dicyclohexylcarbodiimide (DCC) and sodium hydride (NaH). This strategic substitution not only drastically reduces the raw material costs but also simplifies the purification process by avoiding heavy metal contamination entirely. The process operates under mild conditions, typically between -20°C and 25°C, which enhances operational safety and allows for easier temperature control in large reactors. This robust chemical design ensures that the production of high-purity intermediates is both economically viable and environmentally safer.
Mechanistic Insights into Dieckmann Condensation and Impurity Control
The core innovation of this synthesis lies in the meticulous control of the Dieckmann condensation mechanism and the management of side reactions. In the final cyclization step, the use of a strong base like NaH facilitates the intramolecular condensation to form the critical beta-keto ester ring system. However, a common pitfall in such reactions is the generation of thiophenol as a byproduct, which can react with the intermediate to form stubborn thioether impurities. The patent describes a clever solution: the inclusion of a thiophenol trapping agent, such as benzyl bromide or trimethylchlorosilane, directly in the reaction mixture. This scavenger effectively captures the free thiophenol in situ, preventing it from attacking the electrophilic centers of the product. This mechanistic intervention is crucial for maintaining the high purity specifications required for pharmaceutical grade materials, ensuring that the final product consistently achieves purity levels between 95.4% and 98.3%.
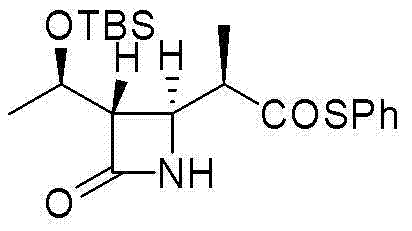
Furthermore, the stereochemical integrity of the molecule is preserved throughout the sequence, which is vital for the biological activity of the final antibiotic. The alkylation step, performed at low temperatures (-70°C to -40°C), ensures high regioselectivity and prevents the epimerization of the methyl group, a degradation pathway that has limited previous methods. By optimizing the molar ratios of the base and the phosphorylating agent (diphenyl chlorophosphate), the reaction drives towards the desired enol phosphate ester with high efficiency. This deep understanding of the reaction kinetics and thermodynamics allows for a reproducible process that minimizes waste and maximizes the yield of the active isomer, providing a solid foundation for regulatory filing and commercial production.
How to Synthesize Beta-Methyl Carbapenem Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step guide for replicating this high-efficiency route in a laboratory or pilot plant setting. The process begins with the activation of the starting material, beta-methyl-ADC-8, followed by a controlled alkylation to extend the carbon chain, and concludes with the ring-closing Dieckmann condensation. Each step is optimized for specific solvent systems, such as acetonitrile for condensation and tetrahydrofuran for alkylation, to ensure maximum solubility and reaction rates. The detailed parameters regarding temperature ramps, addition rates, and quenching procedures are critical for success. For a comprehensive breakdown of the exact operational parameters and stoichiometry required to achieve the reported 90%+ yields, please refer to the standardized synthesis guide below.
- Perform a condensation reaction using beta-methyl-ADC-8 and DCC in acetonitrile at low temperatures to form the thioester precursor.
- Execute an alkylation step using sodium hydride (NaH) as a base in THF to extend the carbon chain efficiently.
- Complete the synthesis via Dieckmann condensation cyclization using diphenyl chlorophosphate and a thiophenol scavenger to ensure high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The elimination of rhodium catalysts removes a major cost driver and supply bottleneck, as the price and availability of precious metals can be highly volatile. Additionally, the avoidance of explosive azide compounds simplifies safety compliance and reduces the need for specialized blast-proof infrastructure, leading to substantial capital expenditure savings. The use of commodity chemicals like NaH and DCC ensures a stable supply chain, reducing the risk of production stoppages due to raw material shortages. Overall, this process represents a significant optimization in cost reduction in API manufacturing, allowing for more competitive pricing of the final antibiotic products.
- Cost Reduction in Manufacturing: The replacement of expensive rhodium catalysts with cheap sodium hydride fundamentally alters the cost structure of the synthesis. By removing the need for costly metal scavenging resins and complex filtration steps associated with heavy metal removal, the overall processing time and consumable costs are significantly reduced. This leaner manufacturing approach translates directly into lower unit costs, enhancing the margin potential for generic manufacturers and improving the affordability of essential medicines.
- Enhanced Supply Chain Reliability: The reliance on widely available organic reagents rather than specialized organometallic complexes ensures a more resilient supply chain. Suppliers can source materials like diphenyl chlorophosphate and thiophenol trapping agents from multiple vendors, mitigating the risk of single-source dependency. This diversification strengthens the continuity of supply, ensuring that production schedules for critical beta-lactam antibiotics can be maintained even during global disruptions in the chemical market.
- Scalability and Environmental Compliance: The mild reaction conditions and simplified workup procedures make this process highly scalable from pilot batches to multi-ton commercial production. The ability to crystallize the product directly from non-polar solvents like n-heptane reduces solvent consumption and waste generation compared to chromatographic purifications. This aligns with green chemistry principles, reducing the environmental footprint of the manufacturing process and facilitating easier compliance with increasingly strict environmental regulations regarding hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: How does this new method improve safety compared to traditional Rh-catalyzed routes?
A: Traditional routes often rely on explosive diazo compounds and expensive rhodium catalysts. This patented method eliminates the need for rhodium and avoids hazardous azide reagents, significantly reducing explosion risks and heavy metal contamination concerns.
Q: What is the role of the thiophenol trapping agent in this synthesis?
A: The thiophenol trapping agent is critical for impurity control. It reacts with free thiophenol generated during the process, preventing the formation of difficult-to-remove thioether impurities that typically lower the yield and purity of the final carbapenem intermediate.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes mild reaction conditions (ranging from -20°C to 25°C), inexpensive bases like NaH, and straightforward crystallization workups, making it highly viable for industrial manufacturing from 100 kgs to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Methyl Carbapenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the modern pharmaceutical supply chain. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our partners receive consistent, high-quality materials regardless of volume. Our state-of-the-art facilities are equipped to handle the specific requirements of beta-lactam chemistry, including strict moisture control and specialized containment for reactive reagents. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of beta-methyl carbapenem intermediate meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this Rh-free route can optimize your budget. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of this vital pharmaceutical building block.
