Advanced Synthesis of Butoconazole Nitrate Intermediate via Stabilized Grignard Chemistry
Advanced Synthesis of Butoconazole Nitrate Intermediate via Stabilized Grignard Chemistry
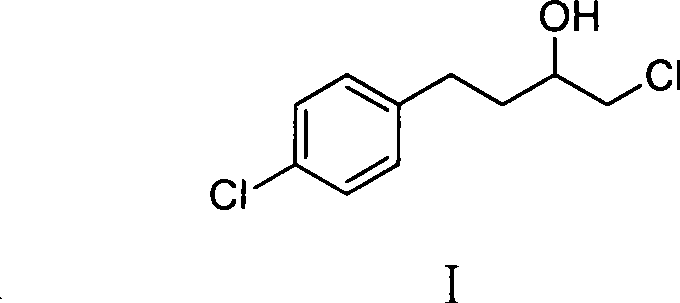
The pharmaceutical landscape for antifungal treatments continues to evolve, driven by the persistent demand for effective therapies against gynecological infections. Central to this domain is Butoconazole Nitrate, a potent imidazole derivative known for its efficacy against Candida albicans. The economic and technical viability of producing this API hinges on the efficient synthesis of its critical precursor, 1-chloro-4-(4-chlorophenyl)-2-butanol. Patent CN101328110B discloses a refined methodology for preparing this essential intermediate, addressing historical limitations in solvent stability and reaction control. By leveraging a Grignard coupling strategy in tetrahydrofuran (THF), this innovation offers a pathway that balances high yield with operational safety, presenting a compelling value proposition for reliable pharmaceutical intermediate suppliers seeking to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-chloro-4-(4-chlorophenyl)-2-butanol has relied on protocols such as those described in US4078071, which utilize diethyl ether as the primary reaction solvent. While chemically feasible on a small laboratory scale, the use of diethyl ether presents significant challenges for industrial application. Ether is highly volatile and possesses a low flash point, creating substantial safety hazards regarding fire and explosion, particularly when managing the exothermic nature of Grignard reagent formation. Furthermore, the reaction profile in ether can be violent and difficult to moderate, leading to inconsistent conversion rates and potential safety incidents during scale-up. These factors contribute to higher operational costs related to specialized containment equipment and rigorous safety monitoring, ultimately rendering the traditional ether-based route less economically attractive for high-volume manufacturing of fine chemical intermediates.
The Novel Approach
The methodology outlined in CN101328110B introduces a strategic shift by employing tetrahydrofuran (THF) as the non-protonic solvent of choice. THF provides a more robust coordination environment for the magnesium species, stabilizing the Grignard reagent formed from p-chlorobenzyl chloride (Formula III). This solvent switch not only enhances the thermal stability of the reaction mixture but also allows for operation at higher, more controllable temperatures ranging from 40°C to 80°C. The improved solvation power of THF ensures a smoother reaction kinetics profile when docking with monochloroacetaldehyde (Formula IV), significantly reducing the risk of runaway exotherms. This transition represents a critical engineering improvement, transforming a hazardous laboratory procedure into a viable, safe, and economically efficient industrial process suitable for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into THF-Mediated Grignard Coupling
The core of this synthetic route lies in the precise generation and utilization of the Grignard reagent. The process initiates with the activation of magnesium metal, often facilitated by a catalytic amount of methyl iodide, within the THF medium. This activation step is crucial for overcoming the passivation layer on the magnesium surface, ensuring rapid and complete formation of the organomagnesium species from p-chlorobenzyl chloride. The choice of THF is mechanistically significant; its oxygen atom acts as a Lewis base, coordinating strongly with the magnesium center to stabilize the carbon-magnesium bond. This stabilization prevents premature decomposition or Wurtz-type coupling side reactions that often plague Grignard syntheses in less coordinating solvents. Consequently, the concentration of the active nucleophile remains high and consistent throughout the addition phase, directly correlating to the observed high conversion efficiency.
Following the formation of the Grignard reagent, the subsequent nucleophilic attack on the carbonyl carbon of monochloroacetaldehyde (Formula IV) dictates the final product quality. The patent specifies a controlled temperature window of 40°C to 80°C during this addition. Maintaining this thermal range is vital for kinetic control; temperatures that are too low may result in incomplete reaction or accumulation of unreacted aldehyde, while excessive heat could promote elimination side reactions or degradation of the sensitive chlorohydrin product. The exothermic nature of the carbonyl addition requires careful heat management, which is facilitated by the higher boiling point of THF compared to ether. Post-reaction, the workup involves acidic quenching to protonate the alkoxide intermediate, followed by standard separation and vacuum distillation. This streamlined purification sequence effectively removes magnesium salts and solvent residues, yielding the target alcohol with the high purity required for downstream antifungal API synthesis.
How to Synthesize 1-chloro-4-(4-chlorophenyl)-2-butanol Efficiently
Implementing this synthesis requires adherence to strict operational parameters to maximize the 80% yield reported in the patent examples. The process begins with the preparation of the Grignard reagent under reflux, ensuring the magnesium is fully consumed before proceeding to the coupling stage. The subsequent addition of the Grignard solution to the aldehyde must be performed dropwise to manage the heat of reaction effectively. Detailed standard operating procedures regarding stoichiometry, addition rates, and distillation cuts are essential for reproducibility. For a comprehensive breakdown of the specific experimental conditions, reagent grades, and step-by-step operational guidelines, please refer to the standardized synthesis protocol below.
- Preparation of Grignard Reagent: React p-chlorobenzyl chloride (Formula III) with magnesium powder in tetrahydrofuran (THF) under reflux conditions, utilizing methyl iodide as an initiator to ensure complete activation of the magnesium surface.
- Grignard Addition and Coupling: Slowly add the prepared Grignard reagent to a solution of monochloroacetaldehyde (Formula IV) in THF, maintaining strict temperature control between 40°C and 80°C to manage the exothermic reaction profile.
- Workup and Purification: Quench the reaction mixture with frozen water and dilute hydrochloric acid, separate the organic phase, dry over anhydrous magnesium sulfate, and purify via vacuum distillation to isolate the target alcohol with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this THF-based protocol offers tangible benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of safety infrastructure. By eliminating the need for handling large volumes of highly volatile diethyl ether, facilities can reduce the complexity of their explosion-proof requirements and lower insurance premiums associated with high-risk solvent storage. This inherent safety improvement translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it allows for more flexible batch scheduling and reduced downtime for safety inspections. Furthermore, the robustness of the THF system minimizes the likelihood of batch failures due to thermal runaway, ensuring a more predictable output volume which is critical for maintaining continuous supply lines to API manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive safety mitigation measures required for ether-based chemistry. The higher stability of the reaction mixture reduces the loss of raw materials due to side reactions or thermal degradation, thereby improving the overall mass balance of the process. Additionally, the use of THF, a widely available commodity solvent, ensures that raw material costs remain stable and competitive. The simplified workup procedure, involving straightforward aqueous quenching and distillation, reduces labor hours and energy consumption associated with complex purification steps, contributing to substantial cost savings per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the volatility of chemical processes; however, this stabilized Grignard method significantly mitigates such risks. The use of readily available starting materials like p-chlorobenzyl chloride and monochloroacetaldehyde ensures that the supply chain is not dependent on niche or scarce reagents. The operational robustness of the THF system means that production campaigns can be extended with fewer interruptions for maintenance or cleaning caused by polymeric byproducts. This reliability allows suppliers to offer more consistent lead times, reducing the inventory buffer stocks that downstream API manufacturers typically need to hold, thus optimizing the working capital for the entire value chain.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often reveals hidden inefficiencies, but this protocol is explicitly designed for industrial suitability. The controlled exotherm and manageable viscosity of the THF reaction mixture facilitate efficient heat transfer in large reactors, a common bottleneck in Grignard chemistry. From an environmental perspective, the process generates less hazardous waste compared to ether-based methods, as THF is easier to recover and recycle through standard distillation trains. This aligns with modern green chemistry principles and regulatory expectations, reducing the burden of waste disposal and ensuring long-term compliance with increasingly stringent environmental regulations governing fine chemical production.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its adoption. The following questions address common inquiries regarding the practical implementation and quality attributes of the intermediate produced via patent CN101328110B. These insights are derived directly from the patent's experimental data and technical disclosures, providing a factual basis for decision-making regarding process validation and vendor qualification.
Q: Why is THF preferred over diethyl ether for this Grignard reaction?
A: THF offers superior solvation properties for the Grignard reagent compared to diethyl ether, leading to enhanced stability and reduced volatility risks. The higher boiling point of THF allows for safer reflux conditions during the exothermic addition of the aldehyde, minimizing the risk of runaway reactions common with low-boiling ether solvents.
Q: What is the expected yield and purity of the intermediate using this method?
A: According to the patent data, this optimized protocol achieves a total recovery yield of approximately 80%. The resulting 1-chloro-4-(4-chlorophenyl)-2-butanol demonstrates high purity levels (HPLC purity ~90% crude, improvable with further processing), which is critical for downstream antifungal API synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. By replacing volatile ether with THF and implementing controlled temperature ranges (40-80°C), the method mitigates safety hazards associated with violent exotherms, making it robust for multi-kilogram to ton-scale manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-chloro-4-(4-chlorophenyl)-2-butanol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final antifungal API is inextricably linked to the purity and consistency of its precursors. Our technical team has extensively analyzed the pathway described in CN101328110B and possesses the expertise to execute this THF-mediated Grignard coupling with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot studies to full-scale manufacturing is seamless. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of 1-chloro-4-(4-chlorophenyl)-2-butanol meets the exacting standards required for global pharmaceutical registration.
We invite R&D directors and procurement leaders to collaborate with us to leverage this optimized synthetic route for your Butoconazole Nitrate projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data from our recent batches and to discuss route feasibility assessments that can further enhance your supply chain efficiency and product competitiveness in the global antifungal market.
