Advanced Rhodium-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral Phosphoryl Amides
Advanced Rhodium-Catalyzed Asymmetric Hydrogenation for High-Purity Chiral Phosphoryl Amides
The landscape of asymmetric synthesis is continually evolving, driven by the demand for high-purity chiral building blocks essential for modern pharmaceutical and agrochemical development. A significant breakthrough in this domain is detailed in patent CN112209965A, which discloses a highly efficient preparation method for chiral N-(2-(phosphoryl)-1-phenylethyl) amides. This technology leverages a sophisticated rhodium-catalyzed asymmetric hydrogenation strategy to transform (Z)-(alpha-aryl-beta-phosphonyl) enamides into valuable chiral intermediates with exceptional yield and stereoselectivity. For R&D directors and procurement specialists seeking reliable sources of complex organophosphorus compounds, this methodology represents a paradigm shift from labor-intensive resolution processes to streamlined catalytic manufacturing. The ability to access these structures with enantiomeric excess values approaching 99% under relatively mild conditions underscores the potential for substantial cost reduction in fine chemical manufacturing and ensures a more robust supply chain for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of optically pure beta-aminophosphines and their derivatives has been fraught with significant technical and economic hurdles. Conventional strategies predominantly relied on asymmetric induction using chiral auxiliaries or the classical resolution of racemic mixtures derived from natural or unnatural amino acids. These legacy approaches suffer from inherent inefficiencies, most notably the 50% theoretical yield ceiling associated with resolution techniques, which necessitates the recycling or disposal of the unwanted enantiomer, thereby inflating raw material costs and waste generation. Furthermore, alternative catalytic methods involving phospha-Michael additions followed by nitro group reduction have exhibited poor substrate applicability and inconsistent enantioselectivity, often requiring excessive catalyst loadings that complicate downstream purification. The reliance on harsh reaction conditions and multi-step sequences in these traditional pathways introduces additional points of failure, increasing the risk of impurity formation and compromising the overall process safety profile, which is a critical concern for supply chain heads managing large-scale production.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the novel approach outlined in the patent utilizes a highly active chiral rhodium catalyst system generated in situ from a rhodium metal precursor and a specialized phosphine-phosphoramidite ligand. This methodology enables the direct asymmetric hydrogenation of (Z)-(alpha-aryl-beta-phosphonyl) enamides, bypassing the need for stoichiometric chiral reagents or resolution steps. The reaction proceeds with remarkable efficiency under mild conditions, typically at ambient temperature (25°C) and moderate hydrogen pressures (50-60 bar), utilizing common solvents such as dichloromethane or trifluoroethanol. This streamlined single-step transformation not only maximizes atom economy but also delivers products with superior stereocontrol, achieving conversion rates of nearly 100% and enantiomeric excess values up to 99%. The versatility of this catalytic system is further evidenced by its wide substrate scope, accommodating various aryl substitutions without significant loss in performance, thereby establishing a new standard for the synthesis of high-purity chiral phosphoryl amides.
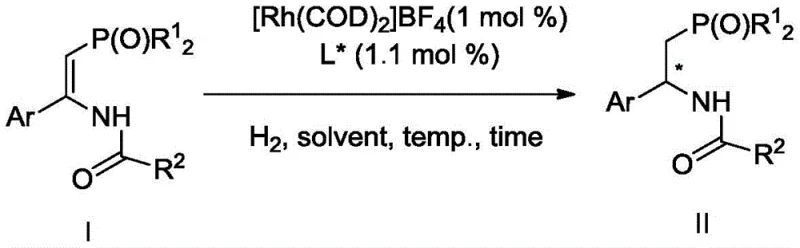
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
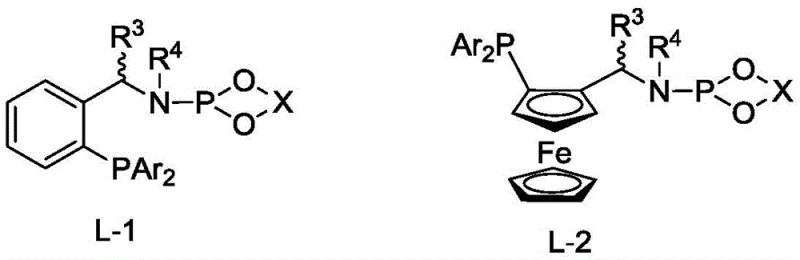
The exceptional performance of this synthetic route is fundamentally rooted in the precise architectural design of the chiral phosphine-phosphoramidite ligands employed in the catalytic cycle. These ligands, characterized by a binaphthyl or biphenyl backbone possessing axial chirality, create a highly defined chiral pocket around the rhodium metal center. This steric environment dictates the facial selectivity of the hydrogen addition to the carbon-carbon double bond of the enamide substrate. The coordination of the phosphine and phosphoramidite moieties to the rhodium atom stabilizes the active catalytic species, facilitating the oxidative addition of hydrogen and the subsequent migratory insertion steps with high fidelity. The presence of bulky aryl groups on the phosphorus atoms further enhances the discrimination between the prochiral faces of the substrate, ensuring that the hydride transfer occurs exclusively to generate the desired (S) or (R) configuration. This level of mechanistic control is critical for R&D teams aiming to minimize impurity profiles, as the high enantioselectivity reduces the burden on downstream chiral separation processes.
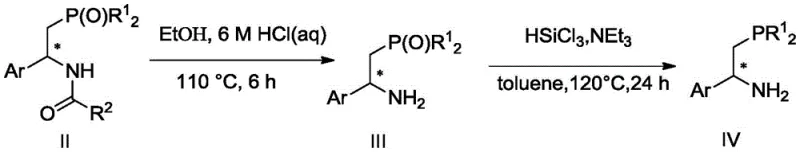
Beyond the primary hydrogenation event, the utility of the resulting chiral amides extends to the facile preparation of free beta-aminophosphines, which are prized ligands in their own right. The patent elucidates a robust downstream processing sequence where the amide bond is hydrolyzed under acidic conditions, followed by the reduction of the phosphoryl group using trichlorosilane and a base like triethylamine. Crucially, this derivatization sequence proceeds with the retention of configuration at the chiral center, meaning the high optical purity established during the initial hydrogenation is preserved in the final amine product. This mechanistic feature allows manufacturers to produce a diverse library of chiral beta-aminophosphine ligands from a common set of amide intermediates. The ability to toggle between the protected amide form and the free amine form provides significant flexibility in inventory management and application testing, enabling chemical producers to serve a broader range of clients in the catalysis and medicinal chemistry sectors without maintaining separate synthesis lines for each derivative.
How to Synthesize Chiral N-(2-(phosphoryl)-1-phenylethyl) Amide Efficiently
Implementing this advanced catalytic technology requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield. The process begins with the in situ generation of the active catalyst, where a rhodium precursor such as [Rh(COD)2]BF4 is combined with the chiral ligand in a molar ratio of approximately 1:1.1 under an inert nitrogen atmosphere. This pre-activation step is vital for forming the coordinatively unsaturated species capable of binding the substrate. Once the catalyst is formed, the reaction is charged with the enamide substrate and subjected to hydrogen pressure. The detailed standardized synthesis steps below outline the precise operational window, including solvent selection, temperature control, and workup procedures, ensuring that technical teams can replicate the high enantioselectivity reported in the intellectual property.
- Prepare the chiral rhodium catalyst in situ by stirring a rhodium metal precursor such as [Rh(COD)2]BF4 with a chiral phosphine-phosphoramidite ligand in a solvent like dichloromethane under nitrogen protection.
- Transfer the catalyst solution to a reactor containing the (Z)-(alpha-aryl-beta-phosphonyl) enamide substrate, replace the atmosphere with hydrogen three times, and maintain a hydrogen pressure of 50-60 bar.
- Stir the reaction mixture at room temperature (25°C) for approximately 24 hours, then remove the solvent via rotary evaporation and purify the product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this rhodium-catalyzed hydrogenation technology offers transformative benefits that extend far beyond simple yield improvements. By shifting from resolution-based methods to direct asymmetric synthesis, organizations can drastically simplify their manufacturing workflows, reducing the number of unit operations and the associated capital expenditure on equipment. The mild reaction conditions eliminate the need for extreme heating or cryogenic cooling, leading to significantly reduced energy consumption and a lower carbon footprint for the production facility. Furthermore, the high turnover frequency of the rhodium catalyst means that precious metal usage is minimized, directly impacting the cost of goods sold. The robustness of the process across a wide range of substrates ensures supply continuity, as the same catalytic system can be utilized to produce various analogues without extensive re-optimization, thereby enhancing the agility of the supply chain in responding to market demands for diverse chiral intermediates.
- Cost Reduction in Manufacturing: The transition to this catalytic asymmetric hydrogenation route eliminates the inherent 50% yield loss associated with classical resolution techniques, effectively doubling the output from the same quantity of starting materials. Additionally, the low catalyst loading required (as low as 1 mol%) combined with the use of commodity solvents like dichloromethane or ethanol minimizes raw material expenses. The streamlined one-step synthesis reduces labor costs and waste disposal fees, contributing to substantial overall cost savings in the production of high-value chiral phosphorus compounds.
- Enhanced Supply Chain Reliability: The broad substrate tolerance of this method ensures that supply chains are less vulnerable to disruptions caused by the scarcity of specific chiral pool starting materials. Since the chirality is induced catalytically rather than derived from expensive natural amino acids, the process relies on readily available synthetic precursors. This independence from biological feedstocks stabilizes pricing and availability, allowing for long-term contracting and reliable delivery schedules for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Operating at ambient temperature and moderate pressure makes this process inherently safer and easier to scale from laboratory to commercial production volumes. The reduction in solvent usage and the elimination of hazardous reagents often required in older synthetic routes align with green chemistry principles, simplifying regulatory compliance and environmental permitting. This scalability ensures that the technology can meet the growing global demand for chiral ligands and API intermediates without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational feasibility and strategic value of adopting this technology for the production of chiral organophosphorus compounds.
Q: What are the primary advantages of this rhodium-catalyzed method over traditional resolution techniques?
A: Unlike traditional chiral resolution which is limited to a maximum theoretical yield of 50%, this asymmetric hydrogenation method achieves near-quantitative conversion with enantiomeric excess values reaching up to 99%, effectively doubling the material efficiency and eliminating the need for recycling unwanted enantiomers.
Q: Can this process be scaled for industrial production of beta-aminophosphine ligands?
A: Yes, the process utilizes mild reaction conditions (25°C) and standard hydrogenation equipment, making it highly suitable for commercial scale-up. The broad substrate scope demonstrated in the patent indicates robustness against various aryl substitutions, ensuring consistent quality across different batches.
Q: How is the final chiral beta-aminophosphine obtained from the amide product?
A: The chiral amide intermediate undergoes a two-step derivatization involving acid-catalyzed hydrolysis to remove the acyl group followed by reduction with trichlorosilane, preserving the stereochemical configuration to yield the target beta-aminophosphine compound in high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphoryl Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-purity chiral building blocks for the development of next-generation therapeutics and advanced catalytic systems. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated rhodium-catalyzed processes described in CN112209965A can be seamlessly transferred to an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral HPLC and NMR capabilities to guarantee that every batch of chiral N-(2-(phosphoryl)-1-phenylethyl) amide meets the exacting standards required by global pharmaceutical partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient synthesis route can optimize your budget. Please contact us today to request specific COA data for our available intermediates and to discuss route feasibility assessments for your target molecules, ensuring a secure and cost-effective supply of these vital chiral components.
