Advanced Synthesis of Sugammadex Sodium Dimer Impurity for High-Purity API Manufacturing
Advanced Synthesis of Sugammadex Sodium Dimer Impurity for High-Purity API Manufacturing
The pharmaceutical industry continuously demands higher purity standards for Active Pharmaceutical Ingredients (APIs), particularly for complex molecules like sugammadex sodium, a selective relaxant binding agent used to reverse neuromuscular blockade. Patent CN111471121A introduces a groundbreaking synthetic method for producing the sugammadex sodium dimer impurity, a critical reference standard that has historically been difficult to source. This innovation addresses a significant gap in quality control protocols, enabling manufacturers to accurately quantify trace impurities that could otherwise compromise patient safety. By establishing a reliable pathway to generate this specific dimeric structure, the technology empowers quality assurance teams to move beyond semi-quantitative estimates towards precise, validated analytical methods. For R&D directors and procurement specialists, understanding this synthesis is key to securing a robust supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the identification and quantification of impurities in sugammadex sodium formulations have been hindered by the unavailability of authentic reference standards for dimeric by-products. In the absence of a certified reference material, manufacturers are forced to rely on self-control methods or area normalization techniques, which suffer from significant accuracy deficits regarding both qualitative identification and quantitative determination. This lack of precision creates a blind spot in the impurity profile, potentially allowing harmful levels of dimeric species to go undetected in the final drug product. Furthermore, attempting to isolate sufficient quantities of this dimer impurity directly from the crude API reaction mixture is technically arduous, often yielding insufficient material for comprehensive toxicological or analytical validation. The conventional reliance on external sourcing or isolation creates a bottleneck that delays regulatory filings and compromises the rigor of stability studies required for global market approval.
The Novel Approach
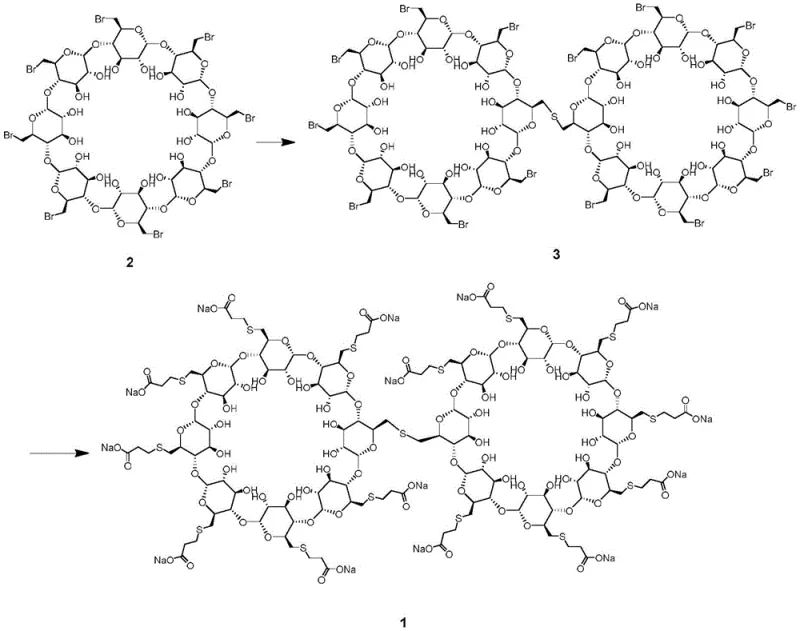
The methodology disclosed in patent CN111471121A offers a transformative solution by synthesizing the dimer impurity de novo rather than isolating it. The process begins with octa-(6-bromo-6-deoxy)-γ-cyclodextrin, a readily available precursor, which undergoes a controlled sulfide bridging reaction to form an intermolecular thioether intermediate. This intermediate is then subjected to a nucleophilic substitution with 3-mercaptopropionic acid under alkaline conditions to yield the final sodium salt of the dimer impurity. This targeted synthetic strategy ensures that the resulting reference standard possesses the exact structural characteristics required for HPLC and MS calibration. By decoupling the production of the reference standard from the main API manufacturing line, this approach significantly enhances the reliability of impurity tracking. It represents a paradigm shift from reactive isolation to proactive synthesis, ensuring that quality control laboratories have consistent access to the tools needed for rigorous safety monitoring.
Mechanistic Insights into Sulfide Bridging and Thiol Substitution
The core of this synthetic innovation lies in the precise control of nucleophilic substitution reactions on the cyclodextrin scaffold. In the first stage, the primary bromide groups at the C6 positions of the gamma-cyclodextrin rings act as excellent leaving groups. When treated with a sulfide source, such as sodium sulfide nonahydrate, the sulfide anion acts as a dinucleophile, attacking two separate cyclodextrin molecules to form a sulfur bridge. This intermolecular linkage is the defining feature of the dimer impurity. The reaction conditions, specifically the use of polar aprotic solvents like DMF and temperature control between -5°C and 45°C, are critical to favoring intermolecular coupling over intramolecular cyclization or polymerization. The stoichiometry is carefully managed, typically using a molar ratio of precursor to sulfide that encourages the formation of the specific dimeric species while minimizing higher-order oligomers.
Following the formation of the thioether-linked intermediate, the second stage involves the displacement of the remaining bromide atoms. The intermediate is reacted with 3-mercaptopropionic acid in the presence of sodium hydroxide. The thiolate anion generated in situ attacks the remaining electrophilic carbon centers, installing the carboxyethyl thio side chains that characterize the sugammadex structure. This step converts the hydrophobic bromo-intermediate into the highly water-soluble sodium salt. The reaction temperature is maintained between 25°C and 80°C to ensure complete conversion without degrading the sensitive glycosidic bonds of the cyclodextrin backbone. This two-step mechanistic pathway allows for the introduction of purification steps between the bridging and the final functionalization, ensuring that the final reference standard is free from mono-substituted or non-bridged contaminants.
How to Synthesize Sugammadex Sodium Dimer Impurity Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for generating this critical reference standard. The process is designed to be operationally simple, utilizing common laboratory solvents and reagents while maintaining strict control over reaction parameters to maximize yield and purity. The initial bridging step requires careful monitoring of the sulfide addition to prevent the formation of polysulfides or excessive cross-linking. Subsequent purification of the intermediate via precipitation and filtration is essential to remove unreacted starting materials before proceeding to the final substitution. For detailed operational parameters, including specific solvent volumes, stirring rates, and crystallization techniques, please refer to the standardized guide below.
- React octa-(6-bromo-6-deoxy)-γ-cyclodextrin with a sulfide source such as sodium sulfide nonahydrate in a polar solvent like DMF to form the intermolecular thioether intermediate.
- Purify the intermediate product through repeated precipitation and filtration to remove unreacted starting materials and intramolecular sulfide by-products.
- React the purified intermediate with 3-mercaptopropionic acid in an aqueous sodium hydroxide solution at controlled temperatures to yield the final sodium sugammadex dimer impurity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the implementation of this synthetic method offers substantial strategic benefits beyond mere technical feasibility. The ability to internally generate or reliably source this specific dimer impurity eliminates a critical vulnerability in the supply chain for sugammadex sodium APIs. Previously, the scarcity of this reference standard posed a risk to batch release timelines, as QC labs struggled to validate impurity levels without proper calibration. By adopting a supply model based on this robust synthesis, organizations can ensure continuous availability of testing materials, thereby preventing production delays caused by analytical bottlenecks. This reliability translates directly into improved inventory management and reduced risk of stockouts for finished dosage forms.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive and inefficient isolation processes. Traditionally, obtaining trace impurities required processing massive quantities of crude API, incurring high solvent and labor costs for minimal yield. This dedicated synthesis route produces the impurity directly from low-cost starting materials, drastically reducing the cost per gram of the reference standard. Furthermore, by avoiding the need for complex preparative chromatography to isolate the dimer from the main product, the process minimizes waste generation and solvent consumption. These efficiencies compound to offer significant cost savings in the overall quality control budget, allowing resources to be reallocated to other critical areas of R&D.
- Enhanced Supply Chain Reliability: Dependence on external vendors for niche reference standards often leads to long lead times and potential quality inconsistencies. This synthetic method utilizes commodity chemicals such as sodium sulfide, 3-mercaptopropionic acid, and gamma-cyclodextrin derivatives, which are widely available from multiple global suppliers. This diversification of raw material sources mitigates the risk of supply disruptions caused by geopolitical issues or single-source vendor failures. Consequently, pharmaceutical manufacturers can maintain a steady flow of reference standards, ensuring that their QC laboratories remain operational without interruption. This stability is crucial for maintaining compliance with stringent regulatory requirements across different international markets.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram-scale production required for large API manufacturing facilities. The use of aqueous workups and standard organic solvents simplifies waste treatment protocols compared to exotic catalytic systems. Additionally, the high selectivity of the sulfide bridging reaction reduces the formation of complex by-product mixtures, simplifying downstream purification and reducing the environmental footprint of the synthesis. This alignment with green chemistry principles supports corporate sustainability goals while ensuring that the manufacturing process remains compliant with increasingly strict environmental regulations regarding solvent discharge and hazardous waste.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of the sugammadex sodium dimer impurity. These insights are derived directly from the patented methodology and are intended to assist technical teams in evaluating the feasibility of adoption. Understanding the nuances of this synthesis helps in making informed decisions about vendor qualification and internal capability building. The answers reflect the current state of the art in impurity reference standard manufacturing.
Q: Why is the synthesis of the sugammadex sodium dimer impurity critical for API quality control?
A: The dimer impurity is a significant reaction by-product in the manufacturing of sugammadex sodium. Without a certified reference standard, accurate qualitative and quantitative analysis is impossible, posing risks to clinical safety. This synthesis method provides the necessary reference material to ensure rigorous impurity monitoring.
Q: What are the key reaction conditions for the sulfide bridging step?
A: The process utilizes octa-(6-bromo-6-deoxy)-γ-cyclodextrin reacting with sodium sulfide nonahydrate in DMF. The reaction is preferably conducted at mild temperatures between 20°C and 30°C to ensure stability and control over the intermolecular linkage formation.
Q: How does this novel synthetic route improve supply chain reliability for reference standards?
A: Previously, there were no reported synthesis methods for this specific impurity, forcing reliance on isolation or unavailable external sources. This robust, scalable two-step synthesis allows for on-demand production of high-purity reference standards, eliminating supply bottlenecks for QC laboratories.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugammadex Sodium Dimer Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final drug product depends on the precision of your analytical controls. As a leading CDMO and supplier of high-value pharmaceutical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at implementing complex synthetic routes, such as the sulfide-bridging methodology described in CN111471121A, ensuring that you receive reference standards with stringent purity specifications. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our materials meet the exacting demands of global regulatory agencies.
We invite you to collaborate with us to optimize your supply chain for sugammadex sodium impurities. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable supply of critical reference standards, ensuring your path to market is smooth, compliant, and cost-effective.
