Scalable Solid-Phase Synthesis of Antibacterial Cyclic Hexapeptide Thermoactinoamide A for Commercial Production
Scalable Solid-Phase Synthesis of Antibacterial Cyclic Hexapeptide Thermoactinoamide A for Commercial Production
The pharmaceutical industry is constantly seeking robust, scalable pathways for novel antibacterial agents, particularly those derived from marine sources which often present significant supply chain bottlenecks. A pivotal advancement in this domain is detailed in Chinese Patent CN108440654B, which discloses a highly efficient synthesis method for Thermoactinoamide A, a potent cyclic hexapeptide with broad-spectrum antibacterial activity against pathogens such as Staphylococcus aureus. This technology represents a critical shift from unreliable natural extraction to a controlled, high-yield chemical manufacturing process, addressing the urgent need for reliable pharmaceutical intermediate suppliers who can guarantee continuity of supply. By leveraging advanced solid-phase peptide synthesis (SPPS) techniques, this method achieves a total yield of 74.24% and purity exceeding 99%, setting a new benchmark for the commercial production of complex marine-derived peptides.
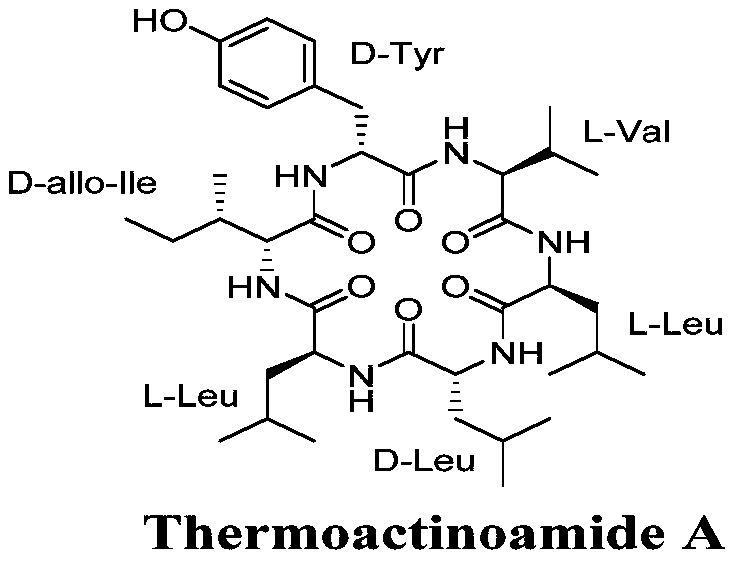
Thermoactinoamide A is structurally defined as cyclo-(D-allo-Ile-Leu-D-Leu-Leu-Val–D-Tyr), a unique arrangement that confers its specific biological activity. The transition from isolating this compound from the bacterial strain ISCAR 2354 found in Iceland to synthesizing it chemically is not merely an academic exercise but a commercial necessity. Natural extraction is plagued by low concentrations, difficult purification from complex biological matrices, and ecological concerns regarding the harvesting of marine organisms. The patented synthetic route overcomes these hurdles by utilizing a 2-chlorotrityl chloride resin support and a strategic combination of coupling reagents, ensuring that the stereochemical integrity of the D-amino acids is preserved while maximizing throughput for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of marine cyclic peptides like Thermoactinoamide A has been constrained by the inherent inefficiencies of natural product isolation. Traditional methods rely on fermenting specific bacterial strains or extracting tissues from marine invertebrates, processes that are notoriously variable and difficult to scale. The yield from natural sources is often negligible, requiring massive volumes of biomass to isolate milligram quantities of the active ingredient, which drives costs prohibitively high for drug development. Furthermore, the purification of natural extracts is complicated by the presence of structurally similar analogs and impurities that are difficult to separate, leading to inconsistent quality and potential safety risks in downstream pharmaceutical applications. This reliance on biological sources also introduces significant supply chain volatility, as environmental factors can drastically alter the production of the target metabolite by the source organism, making it impossible for procurement managers to forecast availability or secure long-term contracts.
The Novel Approach
In stark contrast, the methodology outlined in patent CN108440654B introduces a streamlined solid-phase synthesis protocol that decouples production from biological variability. This novel approach utilizes a linear assembly strategy on a solid support, allowing for the precise addition of each amino acid residue with rigorous quality control at every step. By employing a 2-chlorotrityl chloride resin, the synthesis benefits from mild cleavage conditions that preserve the sensitive peptide backbone, avoiding the harsh acidic treatments often required by other resin systems. The process is designed for operational simplicity, utilizing standard solvents like DMF and DCM, and operates at ambient temperatures (20-29°C), which significantly reduces energy consumption and equipment stress compared to high-temperature solution phase reactions. This shift enables cost reduction in API manufacturing by transforming a scarce natural product into a commodity chemical that can be produced on demand with predictable kinetics and output.
Mechanistic Insights into Dual-System Coupling Strategy
The core innovation of this synthesis lies in its sophisticated dual-coupling reagent system, which is meticulously tailored to the steric and electronic properties of the specific amino acid residues being incorporated. For the coupling of aliphatic amino acids, particularly the leucine and isoleucine residues which are prone to steric hindrance and racemization, the patent specifies the use of the DEPBT/DIEA system. 3-(Diethoxyphosphoryloxy)-1,2,3-benzotriazin-4(3H)-one (DEPBT) acts as a highly efficient condensing agent that forms an active ester in situ, facilitating rapid amide bond formation while minimizing the risk of epimerization at the alpha-carbon. This is critical for maintaining the specific D-configuration of the allo-isoleucine and D-leucine residues, which are essential for the biological activity of the final cyclic peptide. The use of N,N-diisopropylethylamine (DIEA) as the base ensures that the reaction environment remains sufficiently nucleophilic without promoting side reactions, resulting in the reported linear peptide yield of 87.11% with purity over 95%.
Conversely, for the incorporation of the aromatic D-tyrosine and the beta-branched valine, the protocol switches to an Oxyma/DIC coupling system. Ethyl 2-cyano-2-(hydroxyimino)acetate (Oxyma) is known for its superior ability to suppress racemization compared to traditional additives like HOBt, while N,N'-diisopropylcarbodiimide (DIC) serves as the dehydrating agent. This combination is particularly effective for residues that might otherwise form difficult-to-couple intermediates or undergo unwanted side reactions. The strategic alternation between these two coupling systems demonstrates a deep understanding of peptide chemistry, ensuring that each bond in the hexapeptide chain is formed with maximum efficiency. Following the assembly of the linear precursor, the cyclization step utilizes PyBOP (Benzotriazol-1-yl-oxytripyrrolidinophosphonium hexafluorophosphate) in dilute DCM solution. This head-to-tail cyclization is the rate-determining step for macrocyclization, and the use of PyBOP ensures high conversion to the cyclic product (85.23% yield) with minimal dimerization or oligomerization, ultimately delivering a final product with purity exceeding 99% after HPLC purification.
How to Synthesize Thermoactinoamide A Efficiently
The synthesis of Thermoactinoamide A is executed through a nine-step sequence that begins with the loading of the C-terminal amino acid onto the solid support and concludes with the macrocyclization of the linear precursor. The process is designed to be modular, allowing for easy adaptation to automated peptide synthesizers or manual batch processing in standard glass reactors. The initial loading of Fmoc-D-allo-Ile-OH onto the 2-chlorotrityl chloride resin is followed by a capping step to block unreacted sites, ensuring a uniform chain length distribution. Subsequent cycles involve deprotection with 20% piperidine in DMF, followed by the specific coupling reactions described previously. The linear peptide is then cleaved from the resin using a very mild 2% TFA solution in DCM, which preserves the acid-labile side chains and prevents degradation. Detailed standardized synthesis steps for implementing this protocol in a GMP environment are provided in the technical guide below.
- Load Fmoc-D-allo-Ile-OH onto 2-chlorotrityl chloride resin and cap residual sites with DIEA/MeOH.
- Perform iterative deprotection (20% piperidine/DMF) and coupling cycles using DEPBT/DIEA for aliphatic amino acids and Oxyma/DIC for aromatic/valine residues.
- Cleave the linear hexapeptide from the resin using 2% TFA/DCM, purify, and cyclize using PyBOP/DIEA to obtain the final cyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative advantages over traditional sourcing models. The primary benefit is the stabilization of the supply chain; by moving production from a marine extraction model to a chemical synthesis model, companies eliminate the risks associated with seasonal harvesting, geopolitical instability in sourcing regions, and the ecological regulations surrounding marine biodiversity. This transition ensures a continuous, year-round availability of high-purity Thermoactinoamide A, allowing for accurate demand forecasting and inventory planning. Furthermore, the high overall yield of 74.24% significantly reduces the consumption of raw materials per kilogram of finished product, directly impacting the cost of goods sold (COGS). The use of common, commercially available Fmoc-amino acids and standard coupling reagents means that the supply chain for inputs is robust and competitive, preventing single-source bottlenecks that often plague specialty chemical manufacturing.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and inefficient extraction procedures. By utilizing a solid-phase approach with high-loading resins and efficient coupling reagents, the process minimizes solvent usage and waste generation compared to traditional solution-phase synthesis. The high purity achieved (>99%) reduces the burden on downstream purification, lowering the costs associated with preparative HPLC and solvent recovery. Additionally, the mild reaction conditions reduce energy costs for heating or cooling, contributing to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Reliance on natural sources for complex peptides creates a fragile supply chain vulnerable to environmental disruptions. This synthetic method secures the supply by utilizing stable, shelf-stable chemical starting materials that can be sourced from multiple global vendors. The scalability of SPPS allows for rapid ramp-up of production capacity to meet sudden spikes in demand, such as during clinical trial phases or market launch. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that drug development timelines are not delayed by material shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, transitioning smoothly from gram-scale laboratory synthesis to multi-ton commercial production. The use of 2-chlorotrityl resin allows for easy filtration and washing, which simplifies unit operations in large-scale reactors. From an environmental perspective, the high atom economy and reduced solvent waste align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The avoidance of heavy metal catalysts or toxic reagents further simplifies waste treatment and disposal, enhancing the sustainability profile of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Thermoactinoamide A. These answers are derived directly from the technical specifications and experimental data provided in the underlying patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your drug development pipeline.
Q: What is the primary advantage of this synthetic route over natural extraction?
A: The synthetic route described in patent CN108440654B eliminates the reliance on scarce marine bacterial strains, offering a consistent, high-yield (74.24% total) supply chain that is independent of ecological variables and seasonal harvesting limitations.
Q: How is racemization controlled during the synthesis of Thermoactinoamide A?
A: Racemization is minimized by employing a dual coupling strategy: Oxyma/DIC is used for sterically hindered or aromatic residues like D-Tyr and Val, while DEPBT/DIEA is utilized for aliphatic residues like Leu and Ile, ensuring high optical purity throughout the chain elongation.
Q: Is this process suitable for large-scale GMP manufacturing?
A: Yes, the process utilizes standard Fmoc-SPPS protocols with mild reaction conditions (20-29°C) and commercially available reagents, making it highly adaptable for scale-up from laboratory grams to multi-ton commercial production without complex equipment modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thermoactinoamide A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality antibacterial intermediates for the next generation of therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facilities are equipped with state-of-the-art peptide synthesis reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to confirm the stereochemical integrity of D-amino acid residues. We are committed to delivering Thermoactinoamide A that meets the highest international standards, supporting your R&D and commercial manufacturing goals with unwavering consistency.
We invite you to engage with our technical team to discuss how this optimized synthesis route can benefit your specific application. Whether you require custom process optimization or immediate supply of pre-qualified material, our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples to accelerate your development timeline.
