Advanced Chiral Ligand Technology For Scalable Pharmaceutical Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking robust catalytic solutions that can withstand rigorous process conditions while delivering exceptional stereochemical control. Patent CN101023092A introduces a groundbreaking class of novel bisphosphane catalysts designed to address the inherent limitations of previous generation ligands. These enantiomerically enriched bidentate organophosphorus ligands provide a versatile platform for transition metal catalysis, particularly in the asymmetric hydrogenation of prochiral substrates. By optimizing the electronic and stereochemical properties around the metal center, this technology enables the efficient transfer of chirality, which is critical for the synthesis of high-value beta-amino acids and other complex intermediates. The innovation lies not just in the catalytic activity but in the enhanced stability of the ligand framework, allowing for operation under more extreme reaction conditions without degradation. For global procurement teams and R&D directors, this represents a significant opportunity to streamline manufacturing processes and secure a reliable supply chain for critical chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on cyclic phosphines such as DuPhos and BPE ligands for asymmetric catalysis, yet these systems often present significant challenges when scaled for industrial applications. One of the primary drawbacks is the relative instability of the five-membered ring backbone under harsh reaction conditions, which can lead to ligand decomposition and subsequent loss of catalytic activity. Furthermore, conventional ligand matrices often lack the necessary flexibility to be broadly modified within wide limits regarding their steric and electronic properties. This rigidity restricts the ability to fine-tune the catalyst for specific substrates, often necessitating extensive and costly screening processes to find a suitable match. In many cases, the reactive groups within the backbone limit the operational window, forcing manufacturers to operate at suboptimal temperatures or pressures to preserve catalyst integrity. These limitations ultimately translate into higher production costs, increased waste generation, and potential supply chain disruptions due to inconsistent batch quality.
The Novel Approach
The novel approach detailed in the patent data overcomes these historical barriers by introducing a ligand framework that combines the beneficial characteristics of known phosphane systems with significantly improved stability. This new class of bidentate and chiral phosphine ligands is engineered to maintain a favorable asymmetric environment around the central metal atom even under more severe reaction conditions. The design allows for diverse modifiability, meaning the steric and electronic properties can be altered within wide limits to perfectly match specific catalytic problems. This adaptability ensures that the catalyst can be optimized for faster and more selective reactivity compared to prior art systems. By controlling the flexibility of the ligand system, the technology effectively exploits the asymmetric environment for efficient chirality transfer. This results in a catalytic process that is not only more robust but also more economically viable for large-scale manufacturing, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
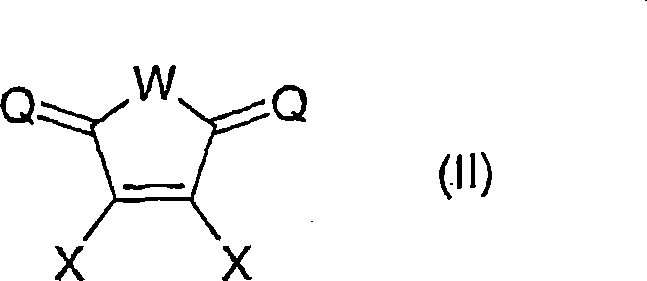
Mechanistic Insights into Bisphosphane-Catalyzed Asymmetric Hydrogenation
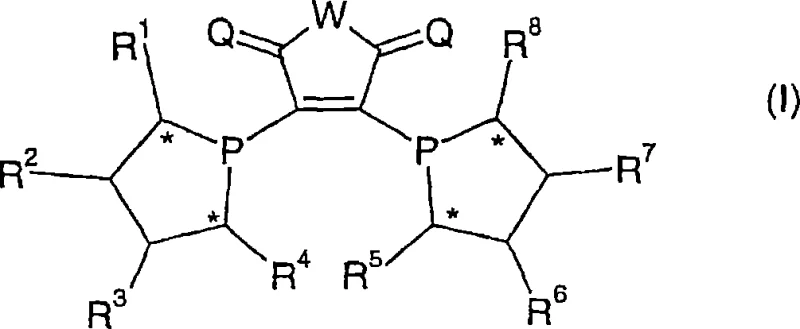
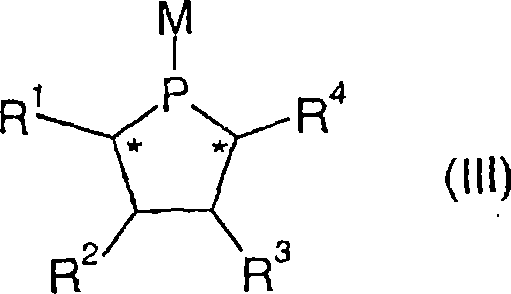
The core mechanism of this technology revolves around the precise construction of the chiral environment surrounding the transition metal center, typically rhodium, palladium, or ruthenium. The ligand of general formula (I) features multiple stereocenters, indicated by asterisks in the structural diagrams, which are crucial for inducing high levels of enantioselectivity during the hydrogenation process. The phosphorus atoms coordinate with the metal to form stable complexes, such as those of general formula (V), where the sum of ligands and solvent molecules creates a specific coordination geometry like octahedral or tetragonal-planar layers. This geometric arrangement is twisted around the transition metal center to create the necessary asymmetry for differentiating between enantiomers of the substrate. The ability to vary substituents R1 through R8 allows chemists to electronically tune the metal center, enhancing its activity towards specific bonds like C=C, C=O, or C=N. Such mechanistic control is essential for achieving the high enantiomeric excess values required in modern drug synthesis.
Impurity control is another critical aspect where this mechanistic design excels, particularly in the context of producing high-purity OLED material or pharmaceutical intermediates. The stability of the ligand backbone prevents the formation of degradation by-products that often contaminate the final product in less robust catalytic systems. By operating under preferred conditions of 0°C to 50°C and hydrogen pressures of 0.5 to 10 bar, the reaction minimizes side reactions that could lead to racemization or over-reduction. The patent highlights that these complexes can hydrogenate E/Z mixtures of prochiral N-acylated beta-aminoacrylic acids without prior separation, yielding products with excellent enantiomeric enrichment. This capability significantly simplifies the downstream purification process, reducing the burden on quality control labs and ensuring that stringent purity specifications are met consistently. The result is a cleaner reaction profile that supports the commercial scale-up of complex polymer additives and fine chemicals.
How to Synthesize Novel Bisphosphane Ligands Efficiently
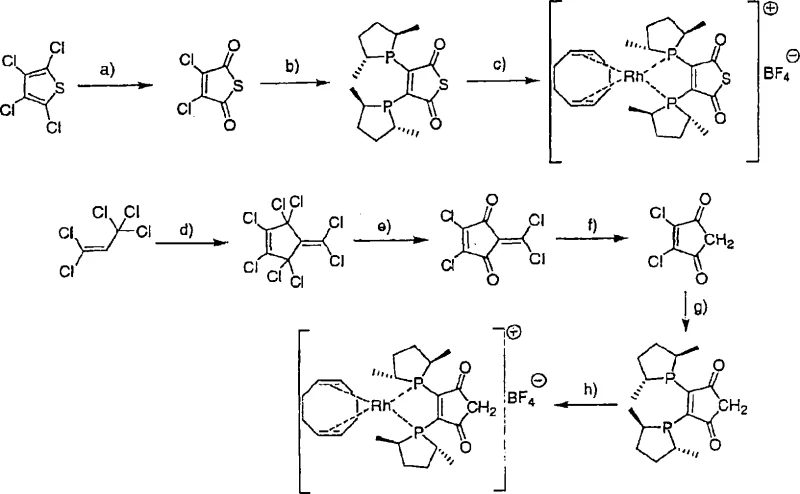
The synthesis of these advanced catalysts follows a logical progression starting from readily available precursors, ensuring that the technology is accessible for commercial adoption. The process typically begins with the preparation of the central dione backbone, followed by coupling with chiral phospholane units to establish the requisite stereocenters. Detailed standardized synthetic steps are provided in the guide below to assist R&D teams in replicating the high yields and purity described in the patent documentation. Understanding the precise stoichiometry and reaction conditions, such as the use of THF at 0°C and subsequent complexation with metal salts, is vital for success. This section serves as a foundational reference for technical teams aiming to integrate this catalytic system into their existing workflows.
- Prepare the central backbone by reacting tetrachlorothiophene or related precursors with nitric acid or ethanol under reflux to form the dichloro-dione intermediate.
- React the dione intermediate with at least 2 equivalents of chiral phospholane derivatives containing lithium, sodium, or magnesium metal centers at 0°C in THF.
- Complex the resulting ligand with a transition metal salt such as [Rh(cod)2]BF4 in dichloromethane to form the active catalyst species for hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel bisphosphane catalyst technology offers substantial strategic advantages beyond mere technical performance. The enhanced stability of the ligand system directly correlates with improved supply chain reliability, as the catalysts are less prone to degradation during storage and transport. This robustness ensures that the active material retains its efficacy over time, reducing the risk of batch failures that can disrupt production schedules. Furthermore, the ability to operate under a wider range of reaction conditions provides manufacturing flexibility, allowing facilities to optimize energy consumption and resource utilization without compromising product quality. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding timelines of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The implementation of this catalytic system drives significant cost savings through qualitative improvements in process efficiency rather than arbitrary percentage claims. By eliminating the need for extensive substrate separation prior to hydrogenation, manufacturers can reduce unit operations and associated labor costs. The high stability of the catalyst also implies a longer operational lifespan, potentially reducing the frequency of catalyst replenishment and lowering the overall consumption of precious metals. Additionally, the simplified workup procedures resulting from cleaner reaction profiles minimize solvent usage and waste disposal expenses. These cumulative efficiencies lead to substantial cost savings in the production of high-value chiral intermediates.
- Enhanced Supply Chain Reliability: The qualitative robustness of these ligands ensures a consistent supply of high-quality catalysts, which is critical for maintaining uninterrupted production lines. The technology supports the use of membrane reactors for continuous processing, which enhances the reliability of the manufacturing process by allowing for the retention and reuse of the catalyst. This capability reduces dependency on frequent raw material deliveries and mitigates the risks associated with supply chain volatility. By securing a stable source of effective catalytic materials, companies can better plan their inventory and meet delivery commitments to their own customers with greater confidence.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is facilitated by the ligand's tolerance to varied reaction conditions and its compatibility with continuous flow systems. The ability to function effectively in membrane reactors supports greener chemistry initiatives by minimizing waste generation and improving atom economy. This aligns with increasingly stringent environmental regulations, reducing the regulatory burden on manufacturing sites. The scalable nature of the synthesis, starting from common precursors, ensures that supply can be ramped up to meet growing demand without encountering significant technical bottlenecks or requiring exotic raw materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bisphosphane catalyst technology. These answers are derived directly from the patent specifications to ensure accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this system into your current manufacturing infrastructure. We encourage technical teams to review these points thoroughly to assess the potential impact on their specific applications.
Q: What distinguishes these bisphosphane ligands from conventional DuPhos systems?
A: Unlike conventional phospholanes which may lack stability under extreme conditions, the novel bisphosphane ligands disclosed in CN101023092A feature a modified backbone that offers improved thermal and chemical stability while maintaining high enantioselectivity.
Q: Can these catalysts be used in continuous flow membrane reactors?
A: Yes, the patent explicitly describes the suitability of these complexes for polymer-amplified systems and membrane reactors, allowing for continuous processing and efficient catalyst retention without frequent replenishment.
Q: What level of enantiomeric excess can be achieved with this technology?
A: The technology supports enantiomeric enrichment greater than 90%, with preferred embodiments achieving over 95% ee, making it highly suitable for the production of high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Novel Bisphosphane Catalysts Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in the development of next-generation pharmaceuticals and fine chemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, providing you with the confidence needed to move forward with your projects. Our expertise in handling complex chiral synthesis allows us to support your R&D efforts with high-quality materials that adhere to the highest industry standards.
We invite you to contact our technical procurement team to discuss how our capabilities can support your specific manufacturing needs. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this novel catalytic system. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions. Partner with us to leverage cutting-edge chemistry for your supply chain success.
