Advanced Solid-Phase Synthesis of Fatty Acyl Cyclic Depsipeptides for Commercial Antibiotic Production
Introduction to Advanced Cyclic Lipopeptide Synthesis
The escalating crisis of antibiotic resistance has intensified the global demand for novel antimicrobial agents, particularly cyclic lipopeptides which exhibit potent activity against Gram-positive bacteria including MRSA. Patent CN102020703A introduces a groundbreaking method for the solid-phase synthesis of fatty acyl cyclic depsipeptide compounds, specifically targeting structures analogous to the natural product A54556. This technology represents a significant leap forward in pharmaceutical intermediate manufacturing, addressing the historical challenges associated with forming the critical ester bond within a peptide backbone. By leveraging a sophisticated orthogonal protection strategy involving Dmab and Trt groups, this process ensures high fidelity in chain assembly and successful macrocyclization. For R&D directors and procurement specialists, this patent outlines a viable pathway to produce high-purity antibiotic intermediates that were previously limited by low-yield fermentation or complex solution-phase chemistry.
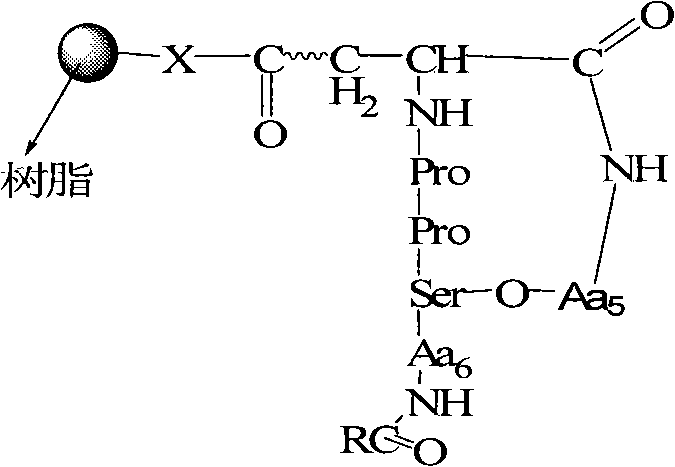
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of cyclic depsipeptides has been plagued by significant chemical hurdles, primarily stemming from the difficulty of forming an ester linkage (lactone) between side-chain hydroxyl and carboxyl groups in the presence of multiple amine functionalities. Conventional solution-phase methods often suffer from poor solubility of the growing peptide chain, leading to aggregation and incomplete reactions. Furthermore, achieving regioselective esterification without protecting every other functional group is notoriously difficult, often resulting in complex mixtures of by-products that are costly and time-consuming to separate. Fermentation, while biologically elegant, is constrained by the metabolic limitations of the host organism, yielding limited quantities and making structural modification for Structure-Activity Relationship (SAR) studies extremely challenging. These inefficiencies create substantial bottlenecks in the supply chain for advanced antibiotic candidates.
The Novel Approach
The methodology described in CN102020703A overcomes these barriers through a meticulously designed solid-phase strategy that prioritizes orthogonality and reaction efficiency. The core innovation lies in the use of Fmoc-Asp-Odmab or Fmoc-Glu-Odmab as the starting anchor, where the Dmab group protects the side-chain carboxyl until the precise moment of esterification. This allows the linear peptide chain to be assembled using standard Fmoc chemistry without interference. Once the linear sequence is complete, the trityl (Trt) group on the serine or threonine side chain is selectively removed, exposing the hydroxyl group for coupling. This precise control eliminates the formation of unwanted oligomers and ensures that the ester bond forms exactly where intended, drastically simplifying the purification process and enhancing the overall viability of the synthetic route for industrial applications.
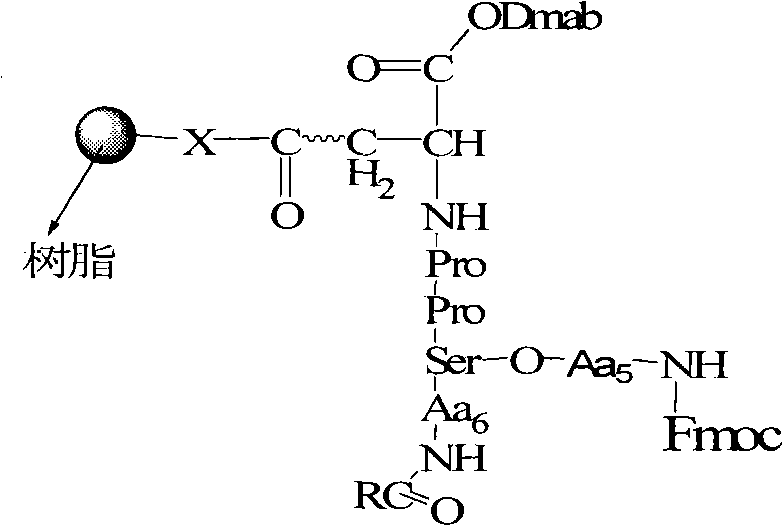
Mechanistic Insights into Orthogonal Protection and On-Resin Cyclization
The chemical elegance of this synthesis is rooted in its triple-orthogonal protection scheme, which manages the reactivity of the amino, hydroxyl, and carboxyl groups independently. The process begins with the loading of the first amino acid onto the resin, followed by iterative coupling cycles using activators like HOBT and DIC. The critical mechanistic step occurs during the side-chain functionalization, where a mild acidic treatment (1% TFA) removes the Trt group from the Ser/Thr residue while leaving the Fmoc and Dmab groups intact. Subsequently, the Dmab group is removed using hydrazine, a reagent that is orthogonal to both acid and base-labile groups. This exposes the side-chain carboxyl of the Asp/Glu residue, allowing it to react with the newly exposed hydroxyl group on the Ser/Thr side chain. This intramolecular esterification is facilitated by an active ester method using DCC and DMAP, ensuring rapid and complete bond formation before the final macrocyclization step.
The final macrocyclization is achieved using a potent coupling cocktail comprising PyBOP, HOBT, and DIEA in NMP solvent. This specific combination is chosen for its ability to activate the C-terminal carboxyl group efficiently while minimizing racemization, a common pitfall in peptide cyclization. The reaction is allowed to proceed for an extended period, typically 48 hours, to ensure thermodynamic equilibrium favors the cyclic monomer over linear oligomers. The solid support plays a crucial pseudo-dilution role here, keeping the reactive ends of the peptide chain in close proximity while preventing intermolecular reactions between different resin beads. This mechanistic precision results in a crude product of exceptional purity, reducing the burden on downstream purification and significantly improving the cost-efficiency of the overall manufacturing process.
How to Synthesize Fatty Acyl Cyclic Depsipeptides Efficiently
The synthesis of these complex molecules requires strict adherence to the optimized protocol detailed in the patent to ensure high yields and purity. The process involves a sequential buildup of the peptide chain on a solid support, followed by selective deprotection and on-resin cyclization. While the general principles of SPPS apply, the specific timing and reagent ratios for the esterification and cyclization steps are critical for success. Operators must carefully monitor the removal of protecting groups using Kaiser tests to prevent deletion sequences. The following guide summarizes the standardized workflow derived from the patent data, providing a clear roadmap for laboratory execution and process scaling.
- Load Fmoc-Asp-Odmab onto resin and elongate the linear peptide chain using standard Fmoc chemistry.
- Selectively deprotect the side-chain hydroxyl group and couple the branching amino acid using an active ester method.
- Perform on-resin macrocyclization using PyBOP/HOBT/DIEA followed by acidic cleavage to release the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from fermentation or solution-phase synthesis to this solid-phase method offers transformative economic and logistical benefits. The primary advantage is the drastic simplification of the production workflow, which eliminates the need for complex fermentation tanks and the associated biological variability. By moving to a chemical synthesis platform, manufacturers gain precise control over batch-to-b consistency and can rapidly adjust production volumes based on market demand without the long lead times associated with biological strain development. Furthermore, the high selectivity of the orthogonal protection strategy means that fewer purification steps are required, directly translating to reduced solvent consumption and lower waste disposal costs. This streamlined approach not only accelerates time-to-market for new antibiotic candidates but also stabilizes the supply chain against biological disruptions.
- Cost Reduction in Manufacturing: The implementation of this solid-phase protocol significantly lowers the cost of goods sold (COGS) by eliminating expensive and inefficient purification stages. Traditional methods often require multiple chromatography steps to separate closely related by-products generated by non-selective esterification. In contrast, the high fidelity of the Dmab/Trt orthogonal strategy ensures that the crude product is already of high purity, allowing for simpler crystallization or precipitation techniques. Additionally, the use of standard, commercially available reagents like PyBOP and DIC avoids the need for custom-synthesized catalysts, further driving down raw material expenses and making the process economically viable for large-scale commercial production.
- Enhanced Supply Chain Reliability: Relying on chemical synthesis rather than fermentation decouples the supply of critical antibiotic intermediates from the vulnerabilities of biological systems. Fermentation processes are susceptible to contamination, strain degeneration, and fluctuating yields, which can cause unpredictable supply shortages. This synthetic method utilizes robust, shelf-stable reagents and standard reactor equipment, ensuring a consistent and reliable output. For supply chain planners, this means more accurate forecasting and the ability to maintain strategic stockpiles of key intermediates without the risk of spoilage or biological failure, thereby securing the continuity of drug manufacturing operations.
- Scalability and Environmental Compliance: The solid-phase nature of this synthesis is inherently scalable, allowing for a seamless transition from gram-scale R&D to kilogram or ton-scale commercial production using automated synthesizers or large stirred-tank reactors. The process is also designed with environmental considerations in mind; the efficient use of reagents and the reduction in solvent-intensive purification steps result in a smaller environmental footprint. The ability to recover and recycle solvents like DMF and DCM, combined with the elimination of biological waste streams, aligns with increasingly stringent global environmental regulations, reducing the compliance burden and potential fines associated with pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the specific chemical mechanisms and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of adopting this technology for their own production lines or research programs.
Q: What is the key advantage of using the Dmab protecting group in this synthesis?
A: The Dmab (4-{N-[1-(4,4-Dimethyl-2,6-dioxocyclohexene)-3-methylbutyl]-amino-}benzyl) group provides orthogonal protection for the side-chain carboxyl group of aspartic or glutamic acid. This allows for selective deprotection and subsequent esterification without affecting the main peptide chain or other protecting groups, solving the critical issue of incomplete esterification.
Q: How does this method address the difficulty of cyclization in depsipeptides?
A: The patent utilizes a potent combination of PyBOP, HOBT, and DIEA in NMP solvent for an extended reaction time (48 hours). This specific reagent system effectively drives the macrocyclization reaction on the solid support, minimizing intermolecular polymerization and ensuring high yields of the desired cyclic structure.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the method relies on solid-phase synthesis (SPPS), which is inherently scalable. The use of standard reagents like DIC, HOBT, and PyBOP, along with straightforward washing and filtration steps, makes the process adaptable for commercial scale-up from grams to kilograms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fatty Acyl Cyclic Depsipeptide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from the laboratory to the marketplace. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fatty acyl cyclic depsipeptide meets the highest international standards. Our expertise in solid-phase peptide synthesis allows us to navigate the complexities of orthogonal protection and macrocyclization with precision, delivering materials that accelerate your drug discovery and development timelines.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project volume and purity requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce your overall development costs.
