Advanced Solid Phase Synthesis for High-Purity Peptide Derivatives and Intermediates
Advanced Solid Phase Synthesis for High-Purity Peptide Derivatives and Intermediates
The pharmaceutical industry constantly demands higher purity standards for peptide-based active ingredients, particularly when dealing with complex sequences containing sensitive functional groups. Patent CN100591690C introduces a robust methodology for the preparation of specific peptide derivatives, notably those incorporating arginine, proline, and glycine residues, utilizing a specialized solid-phase synthesis approach. This innovation addresses critical challenges in maintaining stereochemical integrity while streamlining the purification process. By employing a 2-chlorotrityl chloride (CTC) resin as the solid support, the process facilitates the assembly of the peptide chain under mild conditions that prevent premature deprotection of side chains. This technical breakthrough is particularly relevant for manufacturers seeking a reliable peptide intermediate supplier who can deliver materials with stringent optical specifications. The method described not only enhances the overall yield but also significantly reduces the formation of diastereomeric impurities that often plague traditional solution-phase syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solution-phase peptide synthesis often encounters significant hurdles when attempting to purify intermediates, especially when the target molecule contains multiple basic residues like arginine. In conventional workflows, each coupling step typically requires extensive work-up procedures, including extractions and crystallizations, which can lead to substantial material loss and increased operational costs. Furthermore, the risk of racemization at the chiral centers of amino acids is elevated during activation and coupling in solution, necessitating rigorous and often expensive chromatographic separations to achieve the required polarimetric purity. For procurement managers, these inefficiencies translate into higher raw material consumption and longer lead times, creating bottlenecks in the supply chain for high-purity intermediates. The inability to easily wash away excess reagents and by-products in solution phase also complicates the scale-up process, making it difficult to predict yields consistently across different batch sizes.
The Novel Approach
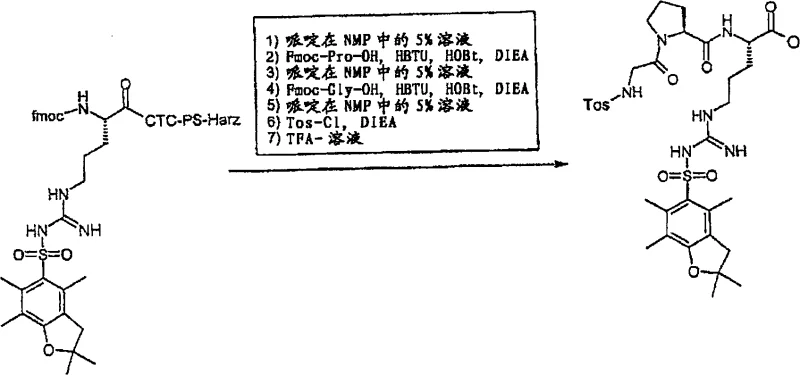
The novel approach detailed in the patent leverages the power of solid-phase peptide synthesis (SPPS) on a CTC resin to overcome these historical limitations. By anchoring the C-terminal arginine to the insoluble polymer support, the synthesis transforms into a series of filtration and washing steps, effectively eliminating the need for intermediate isolations. This "wash-and-react" strategy ensures that excess coupling reagents and soluble by-products are removed simply by rinsing the resin with solvents like NMP or DCM, drastically simplifying the workflow. The use of the CTC linker is particularly strategic because it allows for the cleavage of the full-length peptide from the resin under very mild acidic conditions, such as 1% trifluoroacetic acid in dichloromethane. This mild cleavage preserves the acid-labile Pbf protecting group on the arginine side chain, allowing the resulting peptide intermediate to remain fully protected for subsequent solution-phase couplings, such as the attachment of the p-aminophenyl moiety.
Mechanistic Insights into HBTU-Mediated Coupling and Tosylation
The core of this synthesis relies on highly efficient coupling mechanisms to ensure rapid amide bond formation with minimal epimerization. The patent specifies the use of uranium-based coupling reagents like HBTU (O-benzotriazole-N,N,N',N'-tetramethyl-urea hexafluorophosphate) in conjunction with HOBt (1-hydroxybenzotriazole) and a tertiary amine base such as DIEA. Mechanistically, HBTU activates the carboxyl group of the incoming Fmoc-protected amino acid to form an O-acylisourea intermediate, which is rapidly converted into a more stable and less racemization-prone HOBt ester. This activated ester then reacts with the free alpha-amino group on the resin-bound peptide chain. The presence of the bulky Pbf group on the arginine side chain requires careful optimization of steric access, which is achieved by swelling the CTC resin in polar aprotic solvents like NMP. This solvent choice ensures that the polymer matrix expands sufficiently to allow reagents to penetrate the beads and reach the reactive sites, thereby maximizing the coupling efficiency and preventing the formation of deletion sequences.
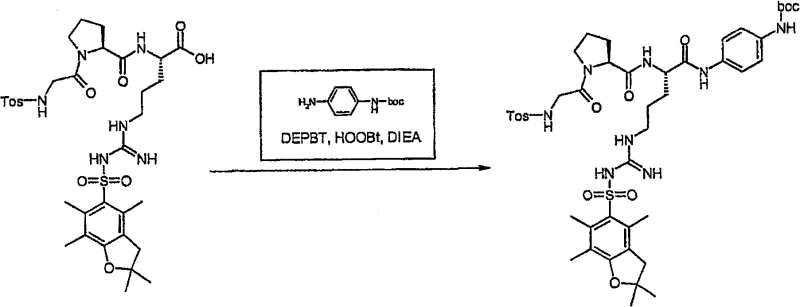
Following the assembly of the tripeptide backbone, a critical tosylation step is performed on the N-terminal glycine residue. This transformation involves the reaction of the free amine with p-toluenesulfonyl chloride (Tos-Cl) in the presence of DIEA. The mechanism proceeds via a nucleophilic attack of the glycine nitrogen on the sulfur atom of the sulfonyl chloride, displacing the chloride ion. This step is crucial as it installs the tosyl group which is part of the final pharmacophore structure. The reaction is typically conducted at temperatures between 0°C and 40°C to balance reaction rate with selectivity. After tosylation, the peptide is cleaved from the resin. Unlike standard Wang resins that require high concentrations of TFA which would remove the Pbf group, the CTC resin releases the peptide as a carboxylic acid while keeping the side-chain protection intact. This orthogonal protection strategy is key to the success of the subsequent coupling with the aniline derivative, as shown in the reaction scheme below.
How to Synthesize Tos-Gly-Pro-Arg Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these complex molecules with high fidelity. It begins with the loading of the first amino acid onto the resin, followed by iterative cycles of deprotection and coupling, and concludes with a specific functionalization and cleavage sequence. This structured approach minimizes variability and ensures that each batch meets the rigorous quality standards expected in pharmaceutical manufacturing. The detailed标准化 synthesis steps见下方的指南 provide the specific molar ratios, solvent volumes, and reaction times necessary to replicate the high yields reported in the examples. By adhering to these parameters, manufacturers can avoid common pitfalls such as incomplete couplings or resin degradation.
- Load Fmoc-Arg(Pbf)-OH onto CTC-resin in DCM with DIEA, followed by washing and drying.
- Perform sequential deprotection with piperidine and coupling of Fmoc-Pro-OH and Fmoc-Gly-OH using HBTU/HOBt.
- Execute N-terminal tosylation with Tos-Cl, cleave from resin with mild TFA, and couple with protected aniline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid-phase methodology offers tangible benefits that extend beyond mere technical feasibility. The shift from solution-phase to solid-phase synthesis fundamentally alters the cost structure of manufacturing these peptide derivatives. By eliminating the need for multiple isolation and purification steps between amino acid additions, the process significantly reduces solvent consumption and labor hours. This streamlined workflow translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the operational overhead associated with handling hazardous intermediates is minimized. Furthermore, the high loading capacity of the CTC resin means that a smaller volume of resin is required to produce a given mass of product, optimizing reactor utilization and reducing the physical footprint required for production.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction in solvent usage for intermediate purifications lead to substantial cost savings. Since the resin-bound intermediates are purified by simple filtration and washing, the expensive chromatography columns often required for solution-phase peptides are reserved only for the final product, if necessary. This reduction in downstream processing complexity lowers the overall cost of goods sold (COGS) and improves the margin profile for the final API. Additionally, the high optical purity achieved (up to 96%) reduces the waste associated with discarding off-spec material, further enhancing economic efficiency.
- Enhanced Supply Chain Reliability: The reagents used in this process, such as Fmoc-protected amino acids, HBTU, and CTC resin, are commodity chemicals available from multiple global suppliers. This commoditization reduces the risk of supply disruptions caused by reliance on single-source custom synthons. The robustness of the solid-phase method also means that the process is less sensitive to minor fluctuations in reaction conditions, ensuring consistent batch-to-batch quality. For supply chain heads, this reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream API manufacturers without the need for excessive safety stock.
- Scalability and Environmental Compliance: Solid-phase synthesis is inherently scalable, as the same principles apply whether running a gram-scale optimization or a multi-kilogram production campaign. The closed nature of the reactor systems used for SPPS minimizes operator exposure to potent compounds and reduces volatile organic compound (VOC) emissions compared to open vessel solution chemistry. The ability to recycle solvents like NMP and DCM through distillation further aligns the process with modern environmental, health, and safety (EHS) standards. This scalability ensures that the commercial scale-up of complex peptides can be achieved smoothly, transitioning from pilot plant to full commercial production without the need for major process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this technology for their specific pipeline projects. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of using CTC-resin for this peptide synthesis?
A: CTC-resin allows for mild cleavage conditions (e.g., 1% TFA) which preserves acid-labile side-chain protecting groups like Pbf, enabling further solution-phase modifications without deprotection.
Q: How does this method ensure high polarimetric purity?
A: The solid-phase approach minimizes racemization risks during coupling by using efficient activation systems like HBTU/HOBt and allows for thorough washing of impurities between steps, achieving up to 96% optical purity.
Q: Can this process be scaled for commercial production?
A: Yes, the protocol utilizes standard SPPS reagents and conditions compatible with large-scale reactors, making it suitable for commercial scale-up of complex peptide intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tos-Gly-Pro-Arg Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We utilize state-of-the-art facilities equipped with rigorous QC labs to verify that every batch meets stringent purity specifications, including the high polarimetric purity demanded by modern regulatory bodies. Our commitment to quality assurance means that we can reliably supply complex peptide derivatives that adhere to the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to optimize your supply chain for these critical materials. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your next project. By partnering with us, you gain access to a supply chain partner dedicated to driving innovation and efficiency in the pharmaceutical sector.
