Advanced Synthesis of Ring-Labeled Alpha-Naphthylacetic Acid for Metabolic Research and Agrochemical Applications
Introduction to Advanced Isotopic Labeling Technology
The global agrochemical industry relies heavily on precise metabolic data to ensure the safety and compliance of plant growth regulators like alpha-naphthylacetic acid (NAA). Patent CN113461509A introduces a groundbreaking preparation method for naphthalene ring C-marked alpha-naphthylacetic acid, addressing a critical gap in environmental residue analysis. Unlike traditional methods that often label the unstable acetic acid side chain, this innovation places the 14C isotope directly on the alpha position of the naphthalene ring. This structural modification is paramount for researchers studying the long-term tendency and metabolic pathways of NAA in soil and crops, as the ring structure is far less susceptible to enzymatic degradation than the carboxylic acid tail. By securing the label on the aromatic core, this technology provides a robust tool for regulatory bodies and agricultural scientists to track the true fate of the molecule with high fidelity.
For procurement specialists and supply chain managers in the fine chemical sector, this patent represents a significant leap in process efficiency and raw material utilization. The disclosed method boasts a total synthesis yield exceeding 60 percent, which is exceptionally high for multi-step isotopic labeling syntheses where precious 14C precursors are involved. The route is designed to minimize the consumption of expensive radioisotopes while maximizing the output of the high-value labeled standard. This efficiency translates directly into better cost management for R&D departments requiring these specialized tracers. Furthermore, the use of readily available industrial solvents and reagents suggests a pathway that is not only scientifically elegant but also commercially viable for scale-up, ensuring a steady supply of high-purity labeled standards for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
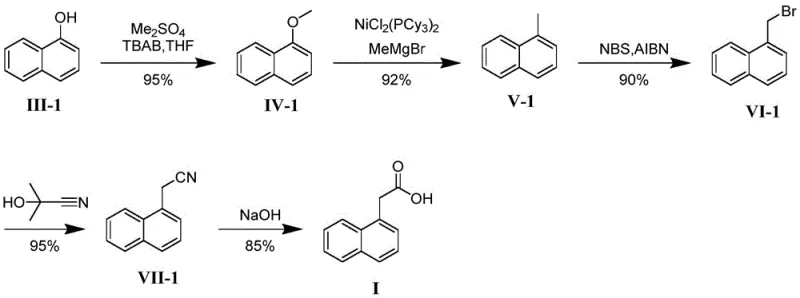
Historically, the synthesis of labeled alpha-naphthylacetic acid has faced significant challenges regarding label stability and precursor availability. Conventional routes often utilize naphthalene or alpha-halogenated naphthalene as starting materials, which can lead to mixtures of isomers or require harsh conditions that degrade sensitive isotopic labels. More critically, many existing methods introduce the 14C label on the acetic acid side chain. While chemically simpler to achieve initially, this placement is metabolically precarious. In biological systems, the acetic acid moiety is frequently cleaved or metabolized rapidly, releasing the label as CO2 or other small molecules that do not reflect the location of the parent naphthalene structure. This results in ambiguous data for environmental impact assessments, forcing researchers to repeat studies or rely on less accurate proxies. Additionally, older synthetic pathways often suffer from low overall yields due to the accumulation of losses over multiple purification steps, making the final labeled product prohibitively expensive for routine screening.
The Novel Approach
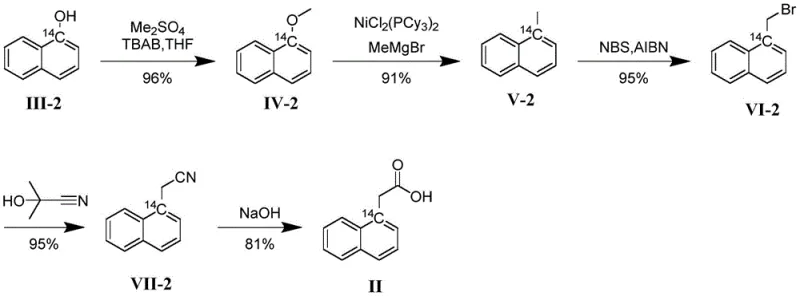
The methodology disclosed in CN113461509A fundamentally shifts the paradigm by utilizing 14C-labeled alpha-naphthol as the primary building block. This strategic choice ensures that the radioactive marker is embedded within the robust aromatic skeleton of the molecule from the very first step. The process involves a streamlined five-step sequence: methylation, nickel-catalyzed coupling, bromination, cyanation, and hydrolysis. As illustrated in the reaction scheme, this route avoids the formation of unstable intermediates that plague traditional methods. The key innovation lies in the efficient conversion of the labeled phenol to the corresponding methyl ether, followed by a highly selective nickel-catalyzed cross-coupling that installs the necessary carbon framework without disturbing the isotopic label. This approach not only guarantees that the label remains at the alpha position of the naphthalene ring but also simplifies the purification process, leading to the reported high total yields. The result is a superior reference standard that offers unparalleled stability for long-term environmental monitoring.
Mechanistic Insights into Nickel-Catalyzed Coupling and Functionalization
The heart of this synthetic breakthrough is the second step: the nickel-catalyzed coupling reaction. In this transformation, the methoxy group of the intermediate (Compound IV) is replaced by a methyl group derived from a Grignard reagent (MeMgBr). This is a chemically demanding reaction, as aryl methyl ethers are typically inert to nucleophilic attack due to the strong C-O bond. The patent specifies the use of specialized nickel complexes, such as NiCl2(PCy3)2 or NiBr2(PCy3)2, which act as powerful catalysts to activate this bond. The tricyclohexylphosphine (PCy3) ligands play a crucial role in stabilizing the low-valent nickel species and facilitating the oxidative addition into the C-O bond. This mechanistic feature allows the reaction to proceed under relatively mild conditions (warming to 80°C in toluene), preserving the integrity of the sensitive 14C label. The high efficiency of this step, with yields reaching up to 92 percent in optimized examples, is a testament to the careful selection of the catalyst system, which minimizes side reactions and homocoupling byproducts.
Following the coupling, the synthesis proceeds through a classic radical functionalization sequence that demonstrates excellent regioselectivity. The methyl group introduced in the previous step is selectively brominated at the benzylic position using N-bromosuccinimide (NBS) and AIBN as a radical initiator. This step is critical because it converts the inert methyl group into a reactive bromomethyl handle (Compound VI) without affecting the aromatic ring or the isotopic label. Subsequent substitution with acetone cyanohydrin introduces the nitrile group, which serves as the precursor to the carboxylic acid. The final hydrolysis step under alkaline conditions (NaOH, 120°C) cleanly converts the nitrile to the desired acetic acid moiety. Throughout this cascade, the 14C atom remains securely anchored on the naphthalene ring, unaffected by the vigorous reaction conditions. This robustness confirms the suitability of the route for producing high-specific-activity materials required for trace-level detection in complex environmental matrices.
How to Synthesize Alpha-Naphthylacetic Acid Efficiently
The synthesis of alpha-naphthylacetic acid via this patented route is designed for operational simplicity and scalability, making it accessible for both laboratory-scale tracer production and larger commercial batches. The process begins with the methylation of alpha-naphthol in a biphasic system using THF and water, facilitated by tetrabutylammonium bromide (TBAB) as a phase transfer catalyst. This ensures rapid and complete conversion to the methyl ether. The subsequent nickel-catalyzed step requires strict exclusion of moisture and oxygen, typically maintained under a nitrogen atmosphere, to preserve the activity of the Grignard reagent and the nickel catalyst. The bromination and cyanation steps utilize common organic solvents like toluene and mixed solvent systems (DMI/THF), which are easy to recover and recycle. Detailed standardized operating procedures for each reaction stage, including precise temperature controls and workup protocols, are essential to replicate the high yields reported in the patent examples. For a complete breakdown of the reaction parameters and stoichiometry, please refer to the technical guide below.
- Methylation of 14C-labeled alpha-naphthol using dimethyl sulfate and TBAB phase transfer catalyst to form the methyl ether intermediate.
- Nickel-catalyzed coupling reaction with methyl Grignard reagent to replace the methoxy group with a methyl group on the naphthalene ring.
- Radical bromination using NBS and AIBN to functionalize the benzylic methyl group, followed by cyanation and alkaline hydrolysis to yield the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route offers compelling advantages that address the core pain points of sourcing specialized isotopic compounds. Traditional methods for producing 14C-labeled standards are often characterized by low throughput and exorbitant costs due to the wasteful consumption of radioisotopes. This new method drastically improves the atom economy of the process. By achieving a total yield of over 60 percent, the process ensures that a significantly larger proportion of the expensive 14C-labeled starting material is converted into the final saleable product. This efficiency gain directly correlates to a reduction in the cost per unit of radioactivity, allowing procurement managers to budget more effectively for metabolic studies. Furthermore, the reliance on commodity chemicals such as dimethyl sulfate, NBS, and sodium hydroxide means that the supply chain is not dependent on obscure or single-source reagents, thereby mitigating the risk of production delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection sequences and the use of a highly efficient nickel catalyst system significantly streamline the manufacturing workflow. In traditional organic synthesis, each additional step introduces yield loss and increases labor and solvent costs. By condensing the synthesis into five high-yielding steps, the overall production cost is substantially lowered. Moreover, the catalyst loading is optimized to be economically viable, avoiding the need for excessive amounts of precious metals. The simplified post-treatment procedures, such as filtration through diatomaceous earth and standard column chromatography, reduce the requirement for specialized purification equipment, further driving down capital and operational expenditures associated with the production of these high-value intermediates.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply chain continuity. Because the reaction conditions are not overly sensitive to minor fluctuations in temperature or pressure (within reasonable limits), the process is easier to control in a manufacturing environment compared to cryogenic or ultra-high-pressure alternatives. The use of stable intermediates allows for potential storage between steps if necessary, providing flexibility in production scheduling. For supply chain heads, this means more predictable lead times and the ability to respond quickly to urgent requests for labeled standards from regulatory agencies or research institutions. The scalability of the reaction, demonstrated by the use of standard solvents like toluene and THF, ensures that production can be ramped up from gram to kilogram scales without encountering fundamental chemical bottlenecks.
- Scalability and Environmental Compliance: Environmental compliance is a critical factor in modern chemical manufacturing. This process generates fewer waste materials relative to the amount of product produced, thanks to the high conversion rates and selectivity. The solvents used are common industrial grades that can be efficiently distilled and recycled, minimizing the volume of hazardous waste requiring disposal. Additionally, the avoidance of heavy metal contaminants (other than the controlled nickel catalyst which is removed during workup) simplifies the quality control process for the final API or intermediate. This aligns with global trends towards greener chemistry and reduces the regulatory burden on the manufacturing facility, ensuring long-term operational sustainability and reducing the risk of environmental compliance violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 14C-labeled alpha-naphthylacetic acid. These answers are derived directly from the experimental data and technical specifications outlined in patent CN113461509A. Understanding these details is crucial for R&D directors planning metabolic studies and procurement officers evaluating supplier capabilities. The stability of the label and the purity of the final product are the primary concerns for end-users, and the patented method provides specific solutions to these challenges through its unique reaction design.
Q: Why is ring-labeling preferred over side-chain labeling for alpha-naphthylacetic acid?
A: Ring-labeling ensures the radioactive carbon remains attached to the stable naphthalene core during metabolic processes. Side-chain labels on the acetic acid moiety are prone to metabolic cleavage, which can lead to inaccurate tracing of the parent compound's environmental fate and residue distribution.
Q: What is the total yield of the novel nickel-catalyzed synthesis route?
A: The patented process achieves a total reaction yield of over 60 percent across the five synthetic steps. Optimized examples in the patent data demonstrate yields reaching up to 63.9 percent for the labeled compound and 63.5 percent for the unlabeled reference standard.
Q: Which catalyst system is used for the key coupling step?
A: The process utilizes a specialized nickel complex catalyst, specifically bis(tricyclohexylphosphine)nickel(II) chloride (NiCl2(PCy3)2) or its bromide analogue. This catalyst facilitates the difficult coupling of the naphthyl methyl ether with the Grignard reagent under mild conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Naphthylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality isotopic standards in advancing agricultural science and environmental safety. Our team of expert chemists has thoroughly analyzed the innovative route described in CN113461509A and is fully prepared to implement this technology for our global clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need milligrams of high-specific-activity tracer or kilograms of unlabeled reference standard, we can deliver with consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including HPLC purity greater than 98 percent and precise radiochemical purity analysis, guaranteeing that every batch meets the exacting demands of regulatory submissions.
We invite you to collaborate with us to optimize your supply chain for agrochemical intermediates. By leveraging our technical expertise in nickel-catalyzed couplings and isotopic labeling, we can offer a Customized Cost-Saving Analysis tailored to your specific project requirements. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us be your partner in delivering reliable, high-purity alpha-naphthylacetic acid solutions that drive your research forward while maintaining cost efficiency and supply security.
