Advanced Palladium-Catalyzed Synthesis of 9-12 Membered Rings for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of 9-12 Membered Rings for Pharmaceutical Applications
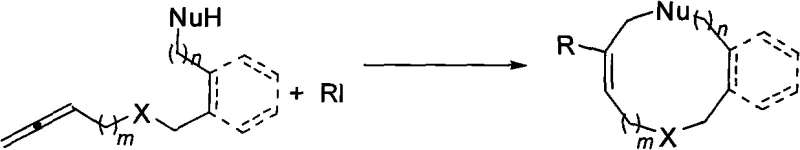
The construction of medium-sized rings, specifically those containing 9 to 12 members, represents one of the most persistent challenges in modern organic synthesis and medicinal chemistry. These mesocyclic scaffolds are ubiquitous in bioactive natural products and serve as critical cores for numerous pharmaceutical candidates, yet their efficient assembly has historically been plagued by entropic penalties and unfavorable transannular interactions. Patent CN101550046A introduces a transformative methodology that addresses these synthetic bottlenecks through a highly stereo- and regioselective palladium-catalyzed cyclization of allenes. By leveraging terminal allenes substituted with nucleophilic groups and reacting them with aryl or alkenyl iodides, this technology enables the robust formation of complex cyclic architectures under remarkably mild conditions. For R&D directors and process chemists seeking reliable pathways to access these difficult motifs, this patent provides a blueprint for efficiency that bypasses the limitations of traditional macrocyclization strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 9-12 membered rings has relied heavily on Ring-Closing Metathesis (RCM), radical-mediated cyclizations, or other metal-catalyzed processes that often demand苛刻 reaction parameters. A primary drawback of these conventional techniques is the necessity for high dilution conditions to suppress intermolecular oligomerization and polymerization, which drastically increases solvent consumption and reduces volumetric productivity. Furthermore, many traditional methods suffer from poor regioselectivity and stereoselectivity, leading to complex mixtures of isomers that are difficult and costly to separate on a commercial scale. When attempting to scale these processes for the production of high-purity pharmaceutical intermediates, the operational complexity and low atom economy associated with high dilution become significant economic barriers, limiting the practical utility of these routes in industrial settings.
The Novel Approach
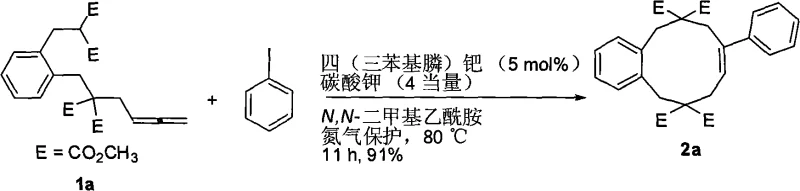
In stark contrast to legacy methods, the technology disclosed in CN101550046A utilizes a palladium-catalyzed cascade reaction that proceeds efficiently without the need for high dilution. By employing tetrakis(triphenylphosphine)palladium as the catalyst and potassium carbonate as the base in N,N-dimethylacetamide, the reaction achieves excellent conversion at a practical concentration of 0.07 mol/L. This approach not only simplifies the operational workflow but also significantly enhances the safety and environmental profile of the synthesis by reducing solvent waste. The method exhibits remarkable substrate universality, accommodating various aryl and alkenyl iodides to produce the desired mesocyclic compounds with yields ranging from 60% to 91%. This robustness makes it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where consistency and purity are paramount.
Mechanistic Insights into Pd-Catalyzed Allene Cyclization
The mechanistic elegance of this transformation lies in the precise orchestration of oxidative addition, migratory insertion, and nucleophilic attack steps facilitated by the palladium center. The cycle initiates with the oxidative addition of the aryl or alkenyl iodide to the Pd(0) species, generating a reactive organopalladium(II) intermediate. This species then undergoes regioselective insertion into the allene moiety, a step that is critical for establishing the correct carbon-carbon bond connectivity required for ring closure. The presence of the terminal nucleophilic group on the allene substrate ensures that the subsequent intramolecular nucleophilic attack occurs with high fidelity, effectively trapping the intermediate and releasing the product while regenerating the active catalyst. This concerted mechanism minimizes the formation of side products and ensures that the stereochemical information inherent in the starting materials is preserved or controlled throughout the process.
From an impurity control perspective, the high regioselectivity of the allene insertion is a decisive factor in maintaining product quality. Traditional radical cyclizations often generate a plethora of byproducts due to non-selective radical additions, necessitating extensive purification efforts that erode overall yield. In this palladium-catalyzed system, the coordination environment around the metal center directs the reaction pathway towards the desired 9-12 membered ring structure, effectively suppressing competing intermolecular reactions even at higher concentrations. This intrinsic selectivity translates directly to a cleaner crude reaction profile, which simplifies downstream processing and allows for the isolation of high-purity intermediates suitable for direct use in subsequent drug synthesis steps without rigorous chromatographic purification.
How to Synthesize 9-12 Membered Cyclic Compounds Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility, particularly regarding the stoichiometry of the base and the purity of the palladium catalyst. The protocol outlined in the patent emphasizes the importance of an inert atmosphere, achieved by flushing the reaction vessel with nitrogen multiple times prior to heating, which prevents catalyst deactivation by oxygen. The reaction mixture is maintained at a moderate temperature of 80°C, balancing reaction kinetics with thermal stability, and is stirred for a duration ranging from 8 to 72 hours depending on the electronic nature of the substituents. Detailed standardized synthesis steps for replicating this high-efficiency transformation are provided in the guide below.
- Charge potassium carbonate and replace atmosphere with nitrogen three times in a reaction vessel.
- Add tetrakis(triphenylphosphine)palladium, nucleophile-substituted allene, and aryl or alkenyl iodide under nitrogen protection.
- Add N,N-dimethylacetamide solvent, heat to 80°C, and stir for 8-72 hours before aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers tangible benefits related to cost structure and operational reliability. The elimination of high dilution conditions represents a major shift in process economics, as it drastically reduces the volume of solvents required per kilogram of product, thereby lowering both raw material costs and waste disposal expenses. Additionally, the use of commercially available reagents such as potassium carbonate and tetrakis(triphenylphosphine)palladium ensures a stable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized catalysts. The robustness of the reaction across a wide range of substrates means that a single platform technology can be applied to manufacture diverse intermediates, streamlining inventory management and reducing the need for specialized equipment.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by operating at standard concentrations rather than high dilution, which substantially decreases solvent usage and energy consumption for solvent recovery. The high yields reported, up to 91% in optimized cases, minimize the loss of valuable starting materials and reduce the burden on purification units. Furthermore, the avoidance of expensive transition metals beyond standard palladium catalysts and the use of simple inorganic bases contribute to a lower overall cost of goods sold, making the final API intermediates more price-competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like aryl iodides and allenes, which are widely produced by the bulk chemical industry, ensures consistent availability and reduces lead times for raw material procurement. The mild reaction conditions (80°C) and ambient pressure operation simplify the engineering requirements for production facilities, allowing for flexible manufacturing in standard glass-lined or stainless steel reactors. This flexibility enhances supply continuity, as the process is less susceptible to disruptions caused by equipment failure or the need for specialized high-pressure or cryogenic infrastructure.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by the absence of hazardous reagents and the generation of minimal toxic waste streams. The simplified workup procedure, involving basic aqueous quenching and extraction, aligns well with green chemistry principles and regulatory expectations for environmental compliance. As production volumes increase from 100 kgs to multi-ton scales, the linear scalability of the batch process ensures that quality attributes remain consistent, supporting the long-term supply needs of pharmaceutical partners without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this palladium-catalyzed cyclization technology. These answers are derived directly from the experimental data and specifications detailed in the patent documentation to provide clarity for technical teams evaluating this route for their specific projects. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing manufacturing workflows.
Q: What are the typical yields for this palladium-catalyzed cyclization?
A: According to patent CN101550046A, the process achieves isolated yields ranging from 60% to 91% depending on the specific substrate and reaction time.
Q: Does this method require high dilution conditions?
A: No, a key advantage of this technology is that it operates effectively at a concentration of 0.07 mol/L, eliminating the need for energy-intensive high dilution protocols common in macrocyclization.
Q: What types of nucleophiles are compatible with this reaction?
A: The method demonstrates broad substrate universality, successfully accommodating nucleophiles such as NTs and C(CO2Me)2 groups to form diverse 9-12 membered ring structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mesocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing complex ring systems for next-generation drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of mesocyclic intermediates meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of palladium-catalyzed reactions, delivering high-quality materials that accelerate your pipeline.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of adopting this route for your target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific rigor and commercial viability.
