Scalable Metal-Free Synthesis of 6-Aryl-5,6-Dihydrophenanthridines for Pharmaceutical Applications
Scalable Metal-Free Synthesis of 6-Aryl-5,6-Dihydrophenanthridines for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective synthetic routes for complex heterocyclic scaffolds, particularly those found in bioactive molecules. Patent CN107778241B introduces a significant advancement in the synthesis of 6-aryl-5,6-dihydrophenanthridine compounds, a structural motif prevalent in various natural products and therapeutic agents. This technology leverages a novel intramolecular cyclization strategy that bypasses the traditional reliance on transition metal catalysis. By utilizing readily available biphenyl-tertiary amine precursors and promoting cyclization through a strong base in a polar aprotic solvent, this method offers a streamlined pathway to high-value intermediates. For R&D directors and procurement specialists, this represents a critical opportunity to optimize manufacturing protocols, reduce dependency on scarce metal resources, and enhance the overall purity profile of the final active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the dihydrophenanthridine core has relied heavily on transition metal-catalyzed cross-coupling reactions or radical cyclizations that require specialized reagents. Traditional approaches often involve the use of palladium or copper catalysts, which introduce significant challenges in downstream processing. The presence of residual heavy metals necessitates rigorous purification steps to meet stringent regulatory limits for pharmaceutical applications, thereby increasing both production time and costs. Furthermore, many conventional methods require harsh reaction conditions, unstable reactants, or complex multi-step sequences that suffer from moderate to low overall yields. These inefficiencies create bottlenecks in the supply chain, making the reliable sourcing of high-purity dihydrophenanthridine derivatives a persistent challenge for manufacturers aiming for commercial scale-up.
The Novel Approach
In stark contrast to legacy methods, the technology disclosed in CN107778241B employs a transition metal-free protocol that dramatically simplifies the synthetic landscape. The core innovation lies in the base-promoted intramolecular cyclization of biphenyl-tertiary amine compounds. As illustrated in the reaction scheme below, the process utilizes potassium tert-butoxide as a promoter in N,N-dimethylformamide (DMF) at elevated temperatures. This approach not only eliminates the need for expensive catalysts but also tolerates a wide range of functional groups on the aromatic rings. The operational simplicity allows for direct heating under nitrogen protection, resulting in good to excellent yields without the complication of metal scavenging. This paradigm shift enables a more sustainable and economically viable manufacturing process for these valuable heterocyclic intermediates.
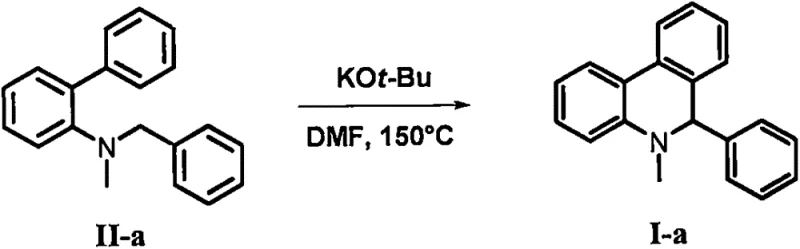
Mechanistic Insights into Base-Promoted Intramolecular Cyclization
The mechanistic pathway of this transformation involves a base-mediated activation of the substrate, leading to ring closure without external oxidants or metal centers. The general reaction scope, depicted in the following figure, highlights the versatility of the method across various aryl substitutions. The strong base, typically potassium tert-butoxide, likely facilitates the deprotonation of a benzylic or adjacent position, generating a reactive nucleophilic species that attacks the proximal aromatic ring. This intramolecular attack forms the new carbon-carbon bond required to close the phenanthridine skeleton. The absence of transition metals suggests a mechanism driven by anionic or radical-anionic intermediates stabilized by the polar solvent environment. Understanding this mechanism is crucial for process chemists aiming to further optimize reaction parameters or adapt the chemistry to novel substrates.
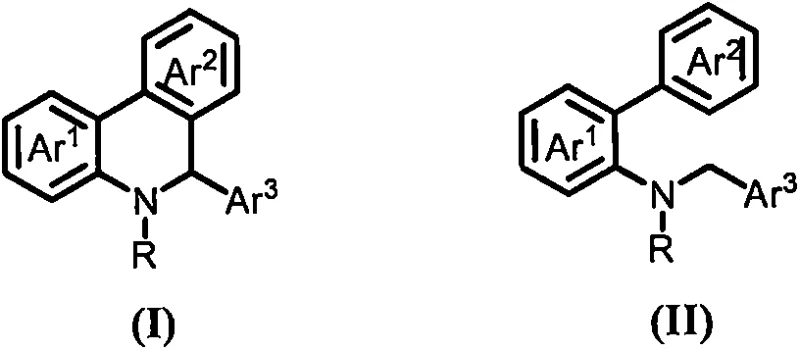
From an impurity control perspective, this metal-free approach offers distinct advantages for maintaining high product quality. In traditional metal-catalyzed reactions, side products often arise from homocoupling of the metal complex or incomplete conversion due to catalyst poisoning. By removing the metal variable, the impurity profile becomes significantly cleaner and more predictable. The primary byproducts are typically derived from over-reaction or solvent interactions, which are generally easier to separate via standard crystallization or chromatography techniques. This inherent cleanliness reduces the burden on analytical teams and ensures that the final API intermediates meet the rigorous specifications required for clinical and commercial batches. The robustness of the base-promoted system ensures consistent batch-to-batch reproducibility, a key metric for supply chain reliability.
How to Synthesize 6-Aryl-5,6-Dihydrophenanthridine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction conditions to maximize yield and safety. The patent data provides clear guidance on the stoichiometry and thermal parameters necessary for success. Typically, the biphenyl-tertiary amine starting material is combined with a slight excess of potassium tert-butoxide in dry DMF. The mixture is then heated to temperatures ranging from 80°C to 180°C, with 150°C identified as the optimal point for balancing reaction rate and product stability. Detailed standardized operating procedures for this synthesis are provided in the structured guide below, ensuring that technical teams can replicate the results with high fidelity.
- Charge a reaction vessel with biphenyl-tertiary amine starting material and potassium tert-butoxide base under nitrogen protection.
- Add N,N-dimethylformamide (DMF) as the solvent and heat the mixture to 150°C for approximately 3 hours to facilitate cyclization.
- Quench the reaction with water, extract with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route translates into tangible strategic benefits beyond mere chemical efficiency. The elimination of transition metal catalysts removes a major cost driver and a potential supply bottleneck, as precious metals like palladium are subject to significant market volatility. Furthermore, the simplified workup procedure reduces the consumption of auxiliary materials such as metal scavengers and specialized filtration media. This streamlining of the manufacturing process leads to substantial cost savings and a reduced environmental footprint, aligning with modern green chemistry initiatives. The ability to produce high-purity intermediates with fewer processing steps enhances the overall agility of the supply chain, allowing for faster response times to market demands.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of expensive transition metal catalysts from the bill of materials. In traditional processes, the cost of the catalyst and the subsequent purification steps to remove metal residues can account for a significant portion of the total production cost. By switching to a base-promoted system using commodity chemicals like potassium tert-butoxide, manufacturers can drastically lower raw material expenses. Additionally, the simplified purification workflow reduces labor and utility costs associated with extended processing times, resulting in a more competitive cost structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Relying on transition metals introduces supply chain risks due to geopolitical factors and mining constraints affecting metals like palladium and platinum. This novel method utilizes widely available organic starting materials and common inorganic bases, ensuring a stable and continuous supply of reagents. The robustness of the reaction conditions means that production is less susceptible to delays caused by reagent shortages or quality variations. For supply chain planners, this reliability is critical for maintaining uninterrupted production schedules and meeting delivery commitments to downstream pharmaceutical clients without the risk of catalyst-related disruptions.
- Scalability and Environmental Compliance: Scaling up chemical processes often amplifies safety and waste management challenges, particularly when dealing with heavy metals. This metal-free methodology is inherently safer and more environmentally friendly, as it generates waste streams that are easier to treat and dispose of in compliance with environmental regulations. The use of standard solvents like DMF and simple thermal conditions facilitates easy translation from gram-scale laboratory experiments to multi-ton commercial production. This scalability ensures that manufacturers can rapidly ramp up production volumes to meet surging demand for dihydrophenanthridine-based therapeutics without requiring complex infrastructure upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within patent CN107778241B, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the practical realities of scaling base-promoted cyclizations in an industrial setting.
Q: Does this synthesis method require transition metal catalysts?
A: No, the method described in patent CN107778241B specifically avoids the use of transition metal catalysts, utilizing potassium tert-butoxide as a promoter instead, which significantly simplifies purification and reduces heavy metal impurities.
Q: What are the optimal reaction conditions for high yield?
A: The patent data indicates that using potassium tert-butoxide in N,N-dimethylformamide (DMF) at a temperature of 150°C provides the highest yields, such as the 68% yield observed in Example 1, compared to lower temperatures or alternative solvents.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly suitable for scale-up due to its operational simplicity, the use of common industrial solvents like DMF, and the elimination of expensive and sensitive metal catalysts, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Aryl-5,6-Dihydrophenanthridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in modern pharmaceutical manufacturing. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries like this base-promoted cyclization are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6-aryl-5,6-dihydrophenanthridine intermediate meets the highest quality standards required by global regulatory bodies. Our commitment to excellence ensures that our partners receive materials that are not only cost-effective but also fully compliant with safety and efficacy requirements.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology for their drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and accelerate your time to market. Let us be your trusted partner in delivering high-quality chemical solutions.
