Advanced Purification Technology for 7-Ketolithocholic Acid to Ensure High-Quality UDCA Manufacturing
The pharmaceutical industry's relentless pursuit of high-purity bile acid derivatives has led to significant advancements in process chemistry, particularly concerning the synthesis of Ursodeoxycholic Acid (UDCA). As detailed in patent CN110003302B, a novel refining method for 7-ketolithocholic acid (7K-LCA) addresses a longstanding bottleneck in the semi-synthetic production of this critical therapeutic agent. The traditional pathway, which oxidizes Chenodeoxycholic Acid (CDCA) to generate 7K-LCA prior to stereoselective reduction, often suffers from the co-formation of persistent oxidative byproducts. This new technology introduces a strategic esterification-crystallization-hydrolysis sequence that effectively isolates the target intermediate from structurally analogous impurities. By implementing mild reaction conditions and utilizing commercially ubiquitous reagents, this methodology not only enhances the chemical purity of the intermediate but also streamlines the downstream processing required for final API manufacture. For global supply chains, this represents a pivotal shift towards more robust and controllable manufacturing protocols for complex steroid intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
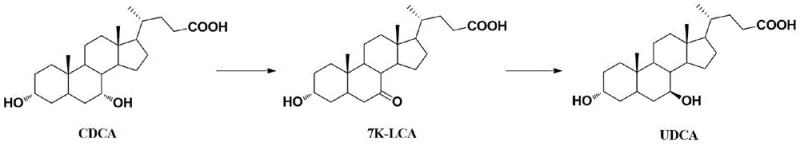
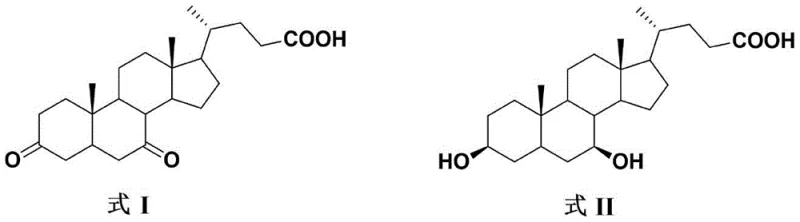
In the conventional synthesis of UDCA, the oxidation of the starting material, CDCA, is a critical yet problematic step. Regardless of the oxidizing agent employed—whether it be N-bromosuccinimide (NBS), bromine, or sodium hypochlorite—the reaction invariably generates a significant quantity of a byproduct known as the "CDCA double oxide." This impurity arises from the non-selective oxidation of the 3-position hydroxyl group alongside the desired 7-position oxidation. The presence of this double oxide is detrimental because, in the subsequent reduction step intended to produce UDCA, it is partially reduced to form "Impurity H." This specific impurity possesses physicochemical properties that are remarkably similar to those of the final product, UDCA, rendering standard purification techniques like recrystallization or chromatography inefficient and costly. Consequently, the accumulation of Impurity H poses a severe risk to the quality profile of the final active pharmaceutical ingredient, necessitating rigorous and often yield-compromising purification strategies at the 7K-LCA stage.
The Novel Approach
The innovative refining method disclosed in the patent circumvents these challenges by exploiting the subtle structural differences between 7K-LCA and the double oxide through derivatization. Rather than attempting the difficult task of separating two highly polar carboxylic acids with similar solubility profiles, the process first converts the crude mixture into their corresponding esters via acid-catalyzed alcoholysis. This chemical modification dramatically amplifies the polarity difference between the target 7K-LCA ester and the double oxide ester. Once esterified, the mixture is subjected to a precise recrystallization process using a binary solvent system, typically comprising an ester solvent and an alkane. This allows the pure 7K-LCA ester to crystallize out selectively while the double oxide ester remains dissolved in the mother liquor. Finally, the purified ester is hydrolyzed under alkaline conditions to regenerate the high-purity 7K-LCA acid, effectively breaking the chain of impurity propagation before it can reach the final UDCA product.
Mechanistic Insights into Esterification-Driven Purification
The core mechanistic advantage of this process lies in the modulation of intermolecular forces and solubility parameters through functional group transformation. Both 7K-LCA and the double oxide contain carboxylic acid groups and hydroxyl functionalities that engage in extensive hydrogen bonding, leading to high lattice energies and similar solubility behaviors in common organic solvents. By converting the carboxylic acid moiety into an ester (e.g., methyl or ethyl ester), the capacity for intermolecular hydrogen bonding is significantly reduced, and the overall lipophilicity of the molecules is altered. Crucially, the structural variance at the 3-position (a hydroxyl group in 7K-LCA versus a ketone in the double oxide) exerts a more pronounced influence on the solubility of the ester derivatives than on the free acids. This amplified difference allows for a thermodynamic separation where the target ester achieves supersaturation and nucleates preferentially, leaving the impurity in the solution phase.
Furthermore, the choice of the recrystallization solvent system is engineered to maximize this selectivity. The use of a good solvent like ethyl acetate combined with a poor solvent like n-heptane creates a gradient of solvation power that is finely tuned to the specific crystal lattice of the 7K-LCA ester. The alkane acts as an anti-solvent, reducing the dielectric constant of the medium and forcing the less soluble component to precipitate. Because the double oxide ester retains higher solubility in this specific mixture due to its distinct dipole moment and lack of the 3-alpha hydroxyl group, it is effectively excluded from the growing crystal lattice. This mechanism ensures that the resulting solid is enriched with the desired isomer, achieving purity levels that are unattainable through direct acid recrystallization, thereby securing the quality of the downstream UDCA synthesis.
How to Synthesize 7-Ketolithocholic Acid Efficiently
The implementation of this refining protocol offers a straightforward yet highly effective route for process chemists aiming to upgrade crude 7K-LCA batches. The procedure begins with the dissolution of the crude acid in a lower alcohol, followed by the controlled addition of a strong acid catalyst to drive the esterification to completion at ambient temperatures. Following workup and isolation of the crude ester, the critical purification step involves dissolving the oil in an ester solvent and slowly introducing an alkane anti-solvent to induce crystallization. The resulting solid is then subjected to saponification to recover the free acid. This sequence transforms a difficult separation problem into a manageable unit operation suitable for large-scale manufacturing. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Dissolve crude 7-ketolithocholic acid in alcohol (e.g., methanol) and catalyze esterification with concentrated sulfuric acid at room temperature.
- Dissolve the resulting crude ester in an ester solvent (e.g., ethyl acetate) and precipitate purified crystals by adding an alkane inert solvent (e.g., n-heptane).
- Hydrolyze the refined ester using an alkaline solution (e.g., NaOH), adjust pH to 2-3 with hydrochloric acid, and filter to obtain high-purity 7-ketolithocholic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this refining technology offers substantial benefits that directly address the pain points of cost volatility and supply reliability in the bile acid market. By shifting the purification burden to the intermediate stage using inexpensive commodity chemicals, manufacturers can avoid the catastrophic yield losses associated with trying to purge Impurity H from the final API. The reliance on common solvents like methanol, ethyl acetate, and heptane eliminates the dependency on specialized or hazardous reagents, simplifying procurement logistics and reducing raw material costs. Furthermore, the mild operating conditions, primarily conducted at room temperature or with moderate heating, reduce energy consumption and minimize the thermal stress on equipment, contributing to longer asset life and lower maintenance overheads.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps or multiple recrystallizations of the final API results in significant operational expenditure savings. By utilizing cheap and easily obtained reagents such as concentrated sulfuric acid and sodium hydroxide, the process avoids the high costs associated with exotic catalysts or proprietary purification media. The high recovery rates observed in the esterification and hydrolysis steps ensure that the overall material throughput is maximized, preventing the financial loss of valuable steroid intermediates. Additionally, the simplicity of the filtration and washing operations reduces labor intensity and processing time, further driving down the unit cost of production.
- Enhanced Supply Chain Reliability: The robustness of this method against variations in crude feedstock quality ensures a consistent output of high-purity 7K-LCA, which is critical for maintaining uninterrupted API production schedules. Since the reagents required are bulk commodities with stable global supply chains, the risk of production stoppages due to raw material shortages is drastically minimized. The ability to effectively scrub the "double oxide" impurity at the intermediate stage acts as a quality firewall, preventing batch failures in the final UDCA synthesis and ensuring that delivery commitments to downstream pharmaceutical partners are consistently met without delay.
- Scalability and Environmental Compliance: The process is inherently scalable, relying on standard unit operations like stirred tank reactions and filtration that are easily transferred from pilot to commercial scale without complex engineering modifications. The use of recyclable solvents like ethyl acetate and heptane aligns with modern green chemistry principles, facilitating easier waste management and solvent recovery. Moreover, the avoidance of heavy metal oxidants or toxic halogenated solvents simplifies the environmental compliance landscape, reducing the burden on wastewater treatment facilities and lowering the overall environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this refining technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on how this method resolves specific purity challenges. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the removal of 'double oxide' critical in 7K-LCA production?
A: The 'double oxide' impurity formed during the oxidation of CDCA is structurally similar to 7K-LCA. If not removed, it converts into Impurity H during the subsequent reduction to UDCA. Impurity H has physicochemical properties nearly identical to UDCA, making it extremely difficult to separate in later stages, thereby compromising the final drug quality.
Q: How does the esterification step improve purification efficiency?
A: Direct purification of the acid forms is difficult due to similar polarities. Converting both 7K-LCA and the double oxide into their ester forms significantly increases the polarity difference between the target molecule and the impurity, allowing for effective separation via solubility differences in a mixed solvent system.
Q: What solvents are preferred for the recrystallization step?
A: The process utilizes a binary solvent system comprising an ester solvent, such as ethyl acetate, and an alkane inert solvent, preferably n-heptane. This combination optimizes the solubility gap, ensuring the target ester crystallizes out while impurities remain in the mother liquor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Ketolithocholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the purity of intermediates like 7-Ketolithocholic Acid is the foundation of a successful pharmaceutical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering materials that meet stringent purity specifications, utilizing our rigorous QC labs to verify that critical impurities, such as the double oxide, are controlled to negligible levels. Our facility is equipped to handle the specific solvent systems and reaction conditions required by this advanced refining process, guaranteeing a supply of intermediates that support the highest standards of drug safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized refining route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this high-efficiency purification method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your production of Ursodeoxycholic Acid remains competitive and compliant in the global market.
