Scalable Metal-Free Synthesis of Z-3-Amino-2-Nitroacrylonitrile for Advanced Pharmaceutical Intermediates
Scalable Metal-Free Synthesis of Z-3-Amino-2-Nitroacrylonitrile for Advanced Pharmaceutical Intermediates
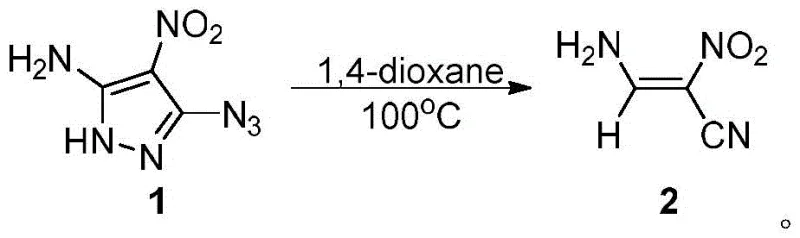
The landscape of organic synthesis is constantly evolving towards greener, more efficient methodologies that reduce reliance on toxic catalysts and complex purification steps. A significant breakthrough in this domain is detailed in patent CN116354852A, which discloses a novel preparation method for (Z)-3-amino-2-nitroacrylonitrile, a compound characterized by strong π-electron delocalization and high thermal stability. This specific alpha,beta-unsaturated nitrile serves as a critical synthon in the construction of complex heterocyclic systems, finding extensive utility in medicinal chemistry and material science. The patented process utilizes a thermal cracking denitrification strategy starting from 5-amino-4-nitro-3-azido-1H-pyrazole, offering a streamlined alternative to traditional multi-step nitrile formations. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the mechanistic robustness and commercial viability of this metal-free route is essential for securing supply chains and optimizing manufacturing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of functionalized alpha,beta-unsaturated nitriles often involves harsh conditions, transition metal catalysis, or multi-step sequences that generate significant waste streams. Conventional routes may require the use of toxic cyanide sources or expensive palladium/copper catalysts, which introduce severe challenges in downstream processing, particularly regarding residual metal limits in active pharmaceutical ingredients (APIs). Furthermore, many existing methods suffer from poor regioselectivity or low thermal stability of the intermediates, leading to polymerization or decomposition before the final product can be isolated. These inefficiencies not only drive up the cost of goods sold (COGS) but also create bottlenecks in the supply chain due to extended purification times and environmental compliance issues associated with heavy metal disposal.
The Novel Approach
In stark contrast, the methodology described in CN116354852A leverages a unique thermal ring-opening strategy that eliminates the need for external metal catalysts entirely. By utilizing 5-amino-4-nitro-3-azido-1H-pyrazole as a high-energy precursor, the reaction proceeds through a controlled thermal cracking mechanism in 1,4-dioxane. This approach is remarkably specific; comparative experiments revealed that common solvents such as toluene, water, and chloroform failed to initiate the reaction, yielding 0% conversion even after prolonged heating. The success of 1,4-dioxane highlights the importance of solvent-solute interactions in facilitating the denitrification process. This novel pathway not only simplifies the operational procedure but also ensures a cleaner reaction profile, directly addressing the industry's demand for cost reduction in pharmaceutical intermediate manufacturing by removing expensive catalytic systems and complex workup procedures.
Mechanistic Insights into Thermal Cracking Denitrification
The core of this synthetic innovation lies in the thermal decomposition of the azido-pyrazole ring system. Upon heating to temperatures between 90°C and 110°C, the azide group (-N3) undergoes thermolysis, releasing nitrogen gas (N2) and generating a highly reactive nitrene or radical intermediate. This energetic species triggers the fragmentation of the five-membered pyrazole ring, effectively contracting the carbon-nitrogen framework into the linear alpha,beta-unsaturated nitrile structure. The presence of the electron-withdrawing nitro group at the 2-position and the amino group at the 3-position stabilizes the resulting double bond through strong conjugation and intramolecular hydrogen bonding, favoring the formation of the (Z)-isomer. This stereoselectivity is crucial for downstream applications where geometric purity influences biological activity or further cyclization efficiency.
From an impurity control perspective, the mechanism offers distinct advantages. Since the reaction is driven purely by thermal energy in a specific solvent environment, side reactions typical of metal-catalyzed cross-couplings, such as homocoupling or over-reduction, are inherently suppressed. The primary byproduct is nitrogen gas, which escapes the reaction matrix, driving the equilibrium forward according to Le Chatelier's principle. The remaining impurities are largely unreacted starting material or minor decomposition products that are easily separated via standard silica gel chromatography. This clean mechanistic profile ensures that the final high-purity OLED material or pharmaceutical precursor meets stringent quality standards without requiring specialized scavenging resins or additional purification stages, thereby enhancing the overall process mass intensity (PMI).
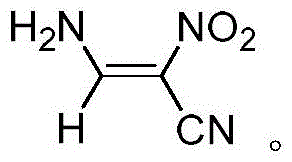
How to Synthesize (Z)-3-Amino-2-Nitroacrylonitrile Efficiently
Executing this synthesis requires precise control over solvent quality and thermal parameters to maximize yield and minimize degradation. The protocol dictates dispersing the azido-pyrazole starting material in 1,4-dioxane at a specific concentration of 0.05 g/mL to ensure optimal solubility and heat transfer. The reaction mixture must be subjected to ultrasonic agitation to guarantee complete dissolution before heating commences. Maintaining the temperature at 100°C for a duration of 24 to 36 hours is critical; shorter reaction times (e.g., 12 hours) result in incomplete conversion, while extending beyond 36 hours offers diminishing returns. Following the thermal treatment, the crude product is isolated via filtration and purified using a mobile phase of petroleum ether and ethyl acetate (4:1 ratio), yielding a characteristic yellow solid that can be further recrystallized to white block crystals.
- Dissolve 5-amino-4-nitro-3-azido-1H-pyrazole in 1,4-dioxane solvent at a concentration of 0.05 g/mL under stirring and ultrasonic conditions.
- Heat the reaction mixture to a temperature between 90°C and 110°C, optimally 100°C, and maintain for 24 to 36 hours to ensure complete thermal cracking.
- Cool the mixture, filter to remove insolubles, and purify the filtrate using silica gel column chromatography with a petroleum ether and ethyl acetate mobile phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free thermal process represents a strategic opportunity to de-risk the supply of critical nitrile intermediates. The elimination of transition metals removes a major variable from the sourcing equation, as there is no longer a dependency on volatile precious metal markets or complex catalyst recovery logistics. Furthermore, the simplicity of the reaction setup—requiring only standard heating equipment and common solvents—means that production can be scaled rapidly across multiple manufacturing sites without the need for specialized reactor linings or high-pressure infrastructure. This operational flexibility significantly enhances supply continuity, ensuring that downstream API production schedules are not disrupted by raw material shortages or quality failures associated with complex catalytic batches.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete removal of expensive transition metal catalysts. In traditional nitrile synthesis, catalyst costs and the subsequent purification steps required to meet residual metal specifications (often in the ppm range) constitute a significant portion of the manufacturing budget. By adopting this thermal cracking route, manufacturers eliminate these line items entirely. Additionally, the use of 1,4-dioxane, a solvent with well-established recovery protocols, allows for efficient recycling, further driving down raw material expenses. The high conversion rates observed at optimized temperatures (up to 77% yield in 36 hours) also mean less starting material is wasted, improving the overall atom economy and reducing the cost per kilogram of the final active intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route against solvent variability is a key supply chain asset. While the process specifically requires 1,4-dioxane, the fact that it tolerates standard thermal conditions without needing inert atmospheres or anhydrous extremes simplifies logistics. Suppliers can maintain larger safety stocks of the stable azido-pyrazole precursor, which can be converted to the final nitrile on demand. This "just-in-time" capability reduces inventory holding costs and minimizes the risk of product degradation during long-term storage. Moreover, the absence of sensitive catalytic systems reduces the likelihood of batch-to-batch variability, ensuring consistent quality delivery to global partners and strengthening the reliability of the commercial scale-up of complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is markedly superior to metal-catalyzed alternatives. The primary gaseous emission is nitrogen, an inert and harmless byproduct, which simplifies exhaust gas treatment requirements. The liquid waste stream consists mainly of organic solvents that can be distilled and reused, aligning with green chemistry principles and reducing the volume of hazardous waste requiring disposal. This reduced environmental footprint facilitates easier regulatory approval in jurisdictions with strict emission standards. Consequently, scaling this process from pilot plant to multi-ton production does not incur the prohibitive costs associated with heavy metal waste management, making it a sustainable choice for long-term reducing lead time for high-purity pharmaceutical intermediates while maintaining corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of (Z)-3-amino-2-nitroacrylonitrile. These insights are derived directly from the experimental data and beneficial effects outlined in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their own synthesis pipelines.
Q: Why is 1,4-dioxane critical for this thermal denitrification reaction?
A: Experimental data from patent CN116354852A demonstrates that alternative solvents like toluene, water, and chloroform result in zero conversion. 1,4-dioxane provides the specific polarity and thermal stability required to facilitate the ring-opening of the azido-pyrazole precursor without decomposing the sensitive nitrile product.
Q: What are the purity specifications for the synthesized acrylonitrile derivative?
A: The process yields a yellow solid which can be recrystallized into white block crystals. Analytical data including 1H NMR, 13C NMR, and HRMS confirms the structure, with elemental analysis showing close alignment to theoretical values (C 31.81%, H 2.60%, N 37.11%), indicating high chemical purity suitable for downstream pharmaceutical applications.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method avoids expensive transition metal catalysts and uses standard thermal conditions (100°C). The absence of metal residues simplifies the purification workflow, making it highly amenable to commercial scale-up for producing complex pharmaceutical intermediates without the burden of heavy metal clearance steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-3-Amino-2-Nitroacrylonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free thermal cracking technology described in CN116354852A for the next generation of pharmaceutical and material science applications. As a leading CDMO partner, we possess the technical expertise to translate this laboratory-scale innovation into robust, GMP-compliant manufacturing processes. Our facilities are equipped to handle the specific thermal requirements and solvent handling protocols necessary for this synthesis, ensuring that every batch meets stringent purity specifications. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we offer a seamless transition from process development to full-scale supply, supported by our rigorous QC labs that verify structural integrity and impurity profiles using advanced NMR and MS techniques.
We invite global partners to collaborate with us to leverage this cost-effective and environmentally friendly synthetic route. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this metal-free approach can optimize your bill of materials. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects, ensuring a secure and competitive supply of this vital alpha,beta-unsaturated nitrile intermediate.
