Advanced Synthesis of E-Beta-Bromo-Gamma-Hydroxymethylene Sulfone for Commercial Scale-Up
Advanced Synthesis of E-Beta-Bromo-Gamma-Hydroxymethylene Sulfone for Commercial Scale-Up
The chemical landscape for producing high-value functionalized sulfones has evolved significantly with the disclosure of patent CN100497306C, which introduces a robust methodology for synthesizing E-beta-bromo-gamma-hydroxymethylene sulfone derivatives. This specific class of compounds serves as a critical building block in the construction of complex molecular architectures found in modern pharmaceuticals and agrochemical agents. The innovation detailed in this patent addresses long-standing challenges regarding stereocontrol and operational simplicity, providing a pathway that bypasses the need for harsh reagents or multi-step protection-deprotection sequences often associated with traditional halogenation protocols. By leveraging an electrophilic addition strategy using elemental bromine in a mixed solvent system, the process achieves remarkable efficiency while maintaining strict control over the geometric configuration of the resulting double bond. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes for active pharmaceutical ingredients (APIs) that rely on this specific sulfone motif, ensuring higher purity profiles and reduced impurity burdens in the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-bromo-alpha,beta-unsaturated sulfones has been plagued by issues related to poor stereoselectivity and the generation of difficult-to-remove Z-isomer impurities. Conventional methods often require low-temperature cryogenic conditions to manage the exothermic nature of bromination, leading to increased energy consumption and operational complexity in a plant setting. Furthermore, many existing protocols rely on stoichiometric amounts of Lewis acids or specialized brominating agents that introduce heavy metal contaminants, necessitating costly downstream purification steps to meet stringent regulatory limits for residual metals in pharmaceutical products. The lack of regioselectivity in older methodologies frequently results in mixtures of constitutional isomers, drastically reducing the overall mass balance and increasing the cost of goods sold due to material loss during chromatographic separation. These inefficiencies create bottlenecks in supply chains, particularly when scaling up from gram-scale laboratory experiments to multi-kilogram pilot runs, where heat transfer and mixing become critical failure points for non-optimized processes.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes a direct electrophilic reaction between alpha-allenyl sulfones and bromine in an acetonitrile medium, followed by the strategic addition of water to trap the intermediate. This method operates effectively at room temperature or mild cooling (0°C), eliminating the need for extreme cryogenic infrastructure and significantly reducing the energy footprint of the manufacturing process. The reaction demonstrates high regioselectivity, ensuring that the bromine atom adds specifically to the beta-position while the hydroxyl group installs at the gamma-position, driven by the electronic properties of the sulfonyl group. Most critically, the process exhibits excellent stereoselectivity, predominantly yielding the thermodynamically stable E-isomer, which simplifies purification and enhances the quality of the intermediate for subsequent coupling reactions. 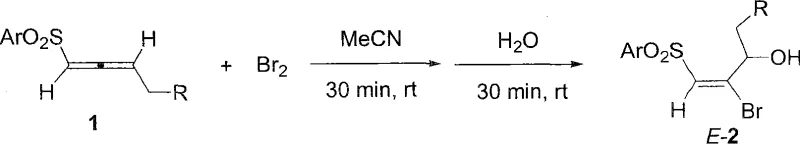
Mechanistic Insights into Electrophilic Bromohydrin Formation
The underlying mechanism of this transformation involves the initial formation of a cyclic bromonium ion intermediate upon the interaction of the electron-rich allene system with molecular bromine. The presence of the electron-withdrawing sulfonyl group adjacent to the allene moiety modulates the electron density, directing the nucleophilic attack of water to the gamma-carbon rather than the alpha-carbon, thereby establishing the observed regiochemistry. This nucleophilic opening of the bromonium ring occurs with anti-addition stereochemistry, which, combined with the subsequent elimination or rearrangement steps facilitated by the reaction conditions, locks the double bond into the E-configuration. Understanding this mechanistic pathway is crucial for process optimization, as it highlights the importance of water concentration and addition rate in controlling the reaction kinetics and preventing over-bromination or polymerization side reactions. The stability of the intermediate species allows for a controlled reaction profile, minimizing the formation of dibromo-byproducts that are common in uncontrolled halogenation reactions of unsaturated systems.
From an impurity control perspective, the mechanism inherently suppresses the formation of Z-isomers due to steric repulsion in the transition state leading to the E-product. The use of acetonitrile as a polar aprotic solvent stabilizes the ionic intermediates without competing as a nucleophile, ensuring that water remains the primary oxygen source for the hydroxyl group installation. This precise control over the reaction trajectory means that the crude reaction mixture contains fewer structurally related impurities, reducing the load on the purification train. For quality assurance teams, this translates to a more consistent impurity profile across different batches, facilitating easier validation and regulatory filing. The ability to tune the substituent effects on the aryl sulfonyl group and the alkyl chain further allows chemists to fine-tune the reaction rate and selectivity for specific target molecules without altering the fundamental process parameters.
How to Synthesize E-Beta-Bromo-Gamma-Hydroxymethylene Sulfone Efficiently
The execution of this synthesis protocol requires careful attention to the order of addition and stoichiometry to maximize yield and selectivity. The process begins with the dissolution of the alpha-allenyl sulfone starting material in acetonitrile, ensuring a homogeneous solution before the introduction of the electrophile. Detailed standardized operating procedures for the precise addition rates, temperature monitoring, and workup protocols are essential for reproducibility at scale. The following guide outlines the critical operational steps derived from the patent examples to ensure successful implementation in a GMP environment.
- Dissolve alpha-allenyl sulfone substrate in acetonitrile solvent at room temperature under stirring conditions.
- Add bromine acetonitrile solution dropwise and stir for 30 minutes, optionally cooling to 0°C for sensitive substrates.
- Introduce water to the reaction mixture, stir for an additional 30 minutes, then proceed to extraction and flash column chromatography for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits in terms of cost structure and supply reliability. The elimination of expensive transition metal catalysts and specialized reagents directly reduces the raw material cost per kilogram, contributing to substantial cost savings in pharmaceutical intermediate manufacturing. The reliance on commodity chemicals such as bromine, acetonitrile, and water ensures that the supply chain is resilient against shortages of exotic reagents, securing continuity of supply even during market fluctuations. Furthermore, the simplified workup procedure, which involves standard extraction and filtration techniques rather than complex distillation or crystallization from hazardous solvents, lowers the operational expenditure associated with waste treatment and solvent recovery. This operational simplicity also shortens the overall cycle time for production batches, allowing facilities to increase throughput without significant capital investment in new equipment.
- Cost Reduction in Manufacturing: The process avoids the use of precious metal catalysts, which eliminates the need for expensive scavenging resins and rigorous metal testing, thereby driving down the total cost of production significantly. By utilizing readily available bulk solvents and reagents, the variable costs associated with raw material procurement are minimized, enhancing the overall margin profile for the final product. The high atom economy of the reaction ensures that a larger proportion of the starting materials are converted into the desired product, reducing waste disposal costs and improving resource efficiency.
- Enhanced Supply Chain Reliability: Since the synthesis relies on stable, shelf-stable reagents that are globally sourced, the risk of supply disruption is markedly reduced compared to routes dependent on custom-synthesized specialty chemicals. The robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-validation effort, providing flexibility in sourcing strategies. This reliability is critical for long-term supply agreements with multinational pharmaceutical companies that require guaranteed delivery schedules for their API production lines.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of highly toxic byproducts make this process inherently safer and easier to scale from pilot plant to commercial tonnage production. The use of water as a reactant and co-solvent aligns with green chemistry principles, reducing the environmental impact and simplifying compliance with increasingly stringent environmental regulations. The ease of purification via flash chromatography or recrystallization ensures that high-purity standards can be met consistently, supporting the production of high-quality intermediates for sensitive applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this chemistry into their existing portfolios.
Q: What is the primary advantage of this bromination method over traditional routes?
A: The primary advantage lies in the exceptional regio- and stereoselectivity, specifically yielding the E-isomer exclusively without requiring complex separation steps, alongside the use of mild reaction conditions.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes common solvents like acetonitrile and water, avoids expensive transition metal catalysts, and operates at near-ambient temperatures, making it highly suitable for large-scale commercial manufacturing.
Q: What represents the typical yield range for this synthesis?
A: According to the patent data, the reaction consistently achieves yields ranging from 60% to 85% across various substituted substrates, demonstrating robust reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable E-Beta-Bromo-Gamma-Hydroxymethylene Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of this advanced synthetic methodology for the production of high-performance pharmaceutical and agrochemical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our capability to handle complex halogenation chemistries safely and effectively positions us as a preferred partner for clients seeking reliable sources of specialized sulfone intermediates.
We invite you to engage with our technical procurement team to discuss how this specific synthesis route can be optimized for your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this more efficient manufacturing process. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your supply chain.
