Advanced Cobalt-Catalyzed Asymmetric Synthesis of (S)-Fenoprofen for Commercial Scale-up
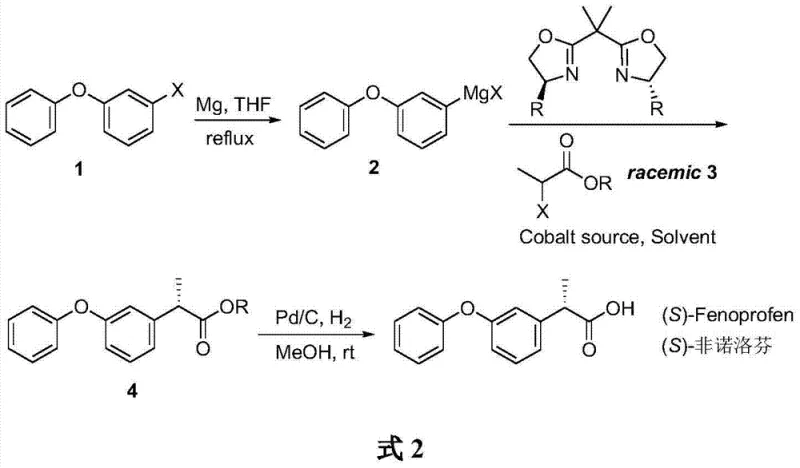
The pharmaceutical industry continuously seeks more efficient pathways to produce high-value chiral intermediates, particularly for non-steroidal anti-inflammatory drugs (NSAIDs) where enantiomeric purity dictates biological efficacy. A pivotal advancement in this domain is detailed in Chinese Patent CN103755554A, which discloses a novel method for the asymmetric catalytic synthesis of (S)-fenoprofen. This specific enantiomer is the pharmacologically active component, exhibiting analgesic potency approximately 35 times greater than its (R)-counterpart, making the exclusion of the inactive isomer critical for patient safety and dosage optimization. The patented methodology leverages a cobalt-catalyzed Kumada cross-coupling reaction, utilizing a self-prepared 3-phenoxyphenyl Grignard reagent and a racemic 2-halopropionate ester. By integrating a specialized bisoxazoline chiral ligand system, this process achieves a remarkable optical purity of 92% ee with a concise three-step synthetic sequence. For global procurement teams and R&D directors, this represents a shift away from expensive precious metal catalysts toward more sustainable base metal chemistry, offering a robust framework for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure (S)-fenoprofen has relied heavily on classical resolution techniques or enzymatic processes, both of which suffer from inherent economic and operational inefficiencies. Traditional kinetic resolution strategies, such as those employing di-9-phenanthrenylmethanol or di-alpha-naphthylcarbinol, are fundamentally constrained by a maximum theoretical yield of 50%, as the unwanted (R)-enantiomer must be discarded or subjected to energy-intensive racemization cycles. Furthermore, enzymatic hydrolysis methods, while selective, often require expensive biocatalysts and strictly controlled aqueous environments that complicate solvent recovery and waste management. Alternative asymmetric catalytic routes utilizing rhodium or nickel have been explored, yet they frequently demand harsh reaction conditions, complex ligand systems, or stoichiometric amounts of chiral inducing agents that drastically inflate the cost of goods sold (COGS). These legacy processes create significant bottlenecks in cost reduction in API manufacturing, as the accumulation of impurities and the loss of half the starting material render them suboptimal for modern, high-volume commercial production requirements.
The Novel Approach
In stark contrast to these legacy limitations, the novel approach outlined in the patent introduces a streamlined, cobalt-mediated strategy that fundamentally alters the economic landscape of fenoprofen production. This method employs an asymmetric Kumada cross-coupling reaction, a powerful carbon-carbon bond-forming tool that connects the aryl Grignard species directly with the propionate backbone in a single stereocontrolling step. By utilizing a cobalt source paired with a chiral bisoxazoline ligand, the reaction proceeds with high enantioselectivity, generating the (S)-fenoprofen ester intermediate with 92% ee directly from racemic precursors. This eliminates the 50% yield ceiling associated with resolution and avoids the high costs of rhodium catalysts. The subsequent catalytic hydrogenation step is straightforward, utilizing standard Pd/C in methanol to cleave the ester protecting group, yielding the final active acid. This concise three-step route not only simplifies the operational workflow but also enhances the overall atom economy, positioning it as a superior choice for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Co-Catalyzed Asymmetric Kumada Coupling
The core innovation of this synthesis lies in the precise orchestration of the cobalt catalytic cycle, which facilitates the enantioselective formation of the chiral center at the alpha-position of the propionic acid moiety. The reaction initiates with the formation of the 3-phenoxyphenyl Grignard reagent, a nucleophilic species generated by the insertion of magnesium into the carbon-halogen bond of 3-phenoxyhalobenzene in anhydrous tetrahydrofuran (THF). This Grignard reagent then interacts with the cobalt catalyst complex, which has been pre-formed by coordinating a cobalt salt, such as cobalt iodide (CoI2), with the chiral bisoxazoline ligand. The steric bulk and electronic properties of the bisoxazoline ligand create a rigid chiral environment around the cobalt center, effectively differentiating between the enantiotopic faces of the incoming electrophile or stabilizing a specific transition state geometry during the transmetallation and reductive elimination steps. This stereochemical control is paramount, as it ensures that the coupling occurs preferentially to form the (S)-configuration, thereby minimizing the formation of the biologically inactive (R)-isomer and reducing the burden on downstream purification processes.
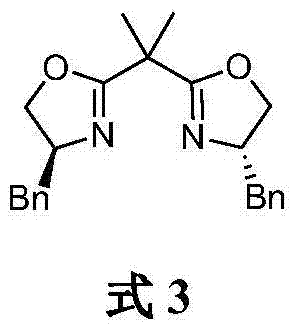
Furthermore, the choice of the chiral ligand is critical for maintaining high optical purity throughout the reaction course. The patent specifies the use of a bisoxazoline ligand (L1), characterized by its rigid bicyclic structure and benzyl substituents that provide the necessary steric hindrance to enforce chirality. During the coupling event at low temperatures (specifically around -80°C), the kinetic energy of the molecules is reduced, allowing the subtle energetic differences between the diastereomeric transition states to dictate the product outcome. This low-temperature protocol suppresses background non-catalyzed reactions that would otherwise lead to racemization. Following the coupling, the resulting ester intermediate retains the stereochemical integrity established during the catalytic cycle. The final hydrogenation step is chemoselective, reducing the benzyl ester to the carboxylic acid without affecting the sensitive ether linkage or the aromatic rings, ensuring that the impurity profile remains clean and manageable for regulatory compliance in drug substance manufacturing.
How to Synthesize (S)-Fenoprofen Efficiently
Implementing this synthesis requires careful attention to moisture exclusion and temperature control, particularly during the Grignard formation and the subsequent asymmetric coupling stages. The process begins with the activation of magnesium powder and the slow addition of the halogenated precursor to ensure steady Grignard reagent generation without exothermic runaway. Once the organometallic species is formed, it is transferred to the cobalt-ligand mixture containing the racemic halopropionate. Maintaining the reaction temperature at -80°C during the addition is crucial for maximizing enantioselectivity, as warmer temperatures could erode the chiral induction provided by the ligand. After the coupling is complete, standard aqueous workup and silica gel chromatography are employed to isolate the chiral ester. The final deprotection is a mild heterogeneous catalysis step that can be easily monitored and controlled. For a detailed breakdown of the specific reagents, molar ratios, and isolation procedures, please refer to the standardized synthesis guide below.
- Preparation of 3-phenoxyphenyl Grignard reagent by reacting 3-phenoxyhalobenzene with magnesium powder in anhydrous THF under reflux.
- Asymmetric Kumada cross-coupling of the Grignard reagent with racemic 2-halopropionate using a cobalt salt and bisoxazoline chiral ligand at low temperature.
- Catalytic hydrogenation of the resulting (S)-fenoprofen ester using Pd/C in methanol to yield the final active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this cobalt-catalyzed route offers substantial strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the substitution of expensive precious metals with abundant base metals, which directly impacts the raw material cost structure. Cobalt salts are significantly cheaper than rhodium or palladium complexes, and the catalyst loading can be optimized to minimize metal residue in the final product, thereby reducing the cost and complexity of heavy metal scavenging steps. Additionally, the elimination of stoichiometric chiral resolving agents removes a major cost driver associated with traditional resolution methods, where half of the synthesized material is essentially wasted. This efficiency translates into a more predictable and stable pricing model for long-term supply contracts, shielding buyers from the volatility often seen in the markets for rare earth metals and specialized enzymes.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive cobalt catalysts and the avoidance of yield-limiting resolution steps. By synthesizing the desired enantiomer directly rather than separating it from a racemate, the effective throughput of the manufacturing plant is nearly doubled without increasing the footprint or raw material intake. Furthermore, the simplified three-step sequence reduces the number of unit operations, solvent exchanges, and purification stages required, leading to lower utility consumption and labor costs. The removal of expensive chiral auxiliaries and enzymes further decreases the bill of materials, allowing for significant margin improvement or competitive pricing strategies in the generic NSAID market.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as 3-phenoxyhalobenzene and simple alkyl halopropionates, are commodity chemicals with robust global supply chains, reducing the risk of raw material shortages. Unlike enzymatic processes that may rely on proprietary biocatalysts with limited suppliers, the reagents here are widely available from multiple chemical vendors. This diversification of the supply base ensures continuity of supply even during market disruptions. Moreover, the synthetic route is less sensitive to the batch-to-batch variability often encountered with biological reagents, providing consistent quality and delivery performance that is critical for just-in-time manufacturing schedules.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route aligns well with green chemistry principles by improving atom economy and reducing waste generation. The absence of stoichiometric chiral waste streams simplifies effluent treatment and lowers the environmental compliance burden. The reaction conditions, while requiring low temperatures for the coupling step, utilize standard industrial refrigeration capabilities and do not involve extreme pressures or hazardous reagents that would necessitate specialized containment infrastructure. This makes the technology highly transferable from pilot scale to multi-ton commercial production, facilitating rapid capacity expansion to meet surging market demand for high-quality anti-inflammatory medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of adopting this route for your specific production needs. Understanding these nuances is essential for making informed decisions about technology transfer and vendor qualification.
Q: What are the primary advantages of using cobalt catalysis over rhodium or nickel for fenoprofen synthesis?
A: Cobalt is a base metal that is significantly more abundant and cost-effective than precious metals like rhodium. Furthermore, the cobalt-bisoxazoline system described in the patent achieves high optical purity (92% ee) without requiring stoichiometric chiral auxiliaries, simplifying the downstream purification process.
Q: How does this synthetic route improve upon traditional resolution methods?
A: Traditional resolution methods are inherently limited to a maximum theoretical yield of 50% for the desired enantiomer. This asymmetric catalytic route bypasses that limitation by directly synthesizing the active (S)-enantiomer from racemic starting materials, effectively doubling the potential yield from the same amount of raw material input.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes standard chemical operations such as Grignard formation, low-temperature coupling, and heterogeneous hydrogenation. While the coupling step requires low temperatures (-80°C), these conditions are manageable in modern pilot and production plants, and the overall three-step sequence is concise enough for efficient scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Fenoprofen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the pharmaceutical marketplace. Our team of expert process chemists has thoroughly analyzed the cobalt-catalyzed Kumada coupling route and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity (S)-fenoprofen that meets stringent purity specifications, utilizing our rigorous QC labs to ensure every batch complies with international regulatory standards. Our facility is equipped to handle the low-temperature requirements and moisture-sensitive operations inherent to this chemistry, guaranteeing a consistent and reliable supply of this vital NSAID intermediate for your formulation needs.
We invite you to collaborate with us to leverage this innovative synthesis method for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data from our recent pilot batches and to discuss detailed route feasibility assessments tailored to your project timelines. Let us help you optimize your sourcing strategy with a solution that balances cost, quality, and supply security.
