Advanced 9-Position Anthracene Fluorene Derivatives for Next-Generation OLED Manufacturing
Introduction to High-Performance Optoelectronic Materials
The rapid evolution of display technology has necessitated the development of organic materials that offer superior thermal stability and charge transport properties. Patent CN1686976A introduces a groundbreaking class of conjugated derivative materials based on 9-position anthracene and pyrene-substituted fluorene. These compounds represent a significant leap forward in the field of organic electronics, specifically targeting applications in organic light-emitting diodes (OLEDs), organic integrated circuits, and organic solar cells. By strategically incorporating large planar aromatic rings such as anthracene and pyrene into the fluorene backbone, the inventors have successfully modulated the electronic structure to enhance carrier mobility without compromising the inherent high fluorescence efficiency of the fluorene system. This technical breakthrough addresses critical bottlenecks in current display manufacturing, particularly regarding the longevity and color purity of blue and red emitting layers.
For research and development directors overseeing material selection, the significance of this patent lies in its dual approach to material modification and electronic structure modulation. The synthesis methodology described allows for the creation of both oligomers and high polymers with repeating units ranging from 1 to 50, providing unparalleled flexibility in tuning the bandgap and emission spectra. The ability to synthesize these materials via robust cross-coupling reactions ensures that they can be produced with the high purity required for commercial electronic applications. As the industry shifts towards flexible and large-area displays, the thermal and spectral stability offered by these 9,9-diaryl substituted structures becomes a decisive factor in device reliability and performance consistency over extended operational lifetimes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional fluorene-based conjugated polymers have long been utilized in optoelectronic devices due to their high quantum efficiency and ease of processing. However, these conventional materials suffer from intrinsic limitations that hinder their performance in high-end applications. A primary issue is the mismatch between the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) energy levels of standard fluorenes and the work functions of common anode and cathode materials. This energetic misalignment creates a barrier for efficient hole and electron injection, leading to imbalanced carrier transport within the emissive layer. Furthermore, standard fluorene derivatives often exhibit poor thermal stability, which predisposes them to forming aggregates and excimer complexes under the heat generated during device operation. These aggregates act as quenching sites, drastically reducing luminescence efficiency and causing undesirable spectral shifts that degrade color purity.
Additionally, the synthesis of conventional high-molecular-weight fluorene polymers often relies on catalysts that leave behind transition metal residues, which can act as deep-level traps for charge carriers. The lack of steric bulk at the 9-position in simple polyfluorenes allows for close packing of polymer chains, facilitating interchain interactions that promote non-radiative decay pathways. For procurement managers and supply chain heads, these material deficiencies translate into lower device yields and higher rejection rates during quality control. The inability to maintain consistent spectral output under thermal stress also limits the operational envelope of the final display products, restricting their use in demanding environments where thermal management is challenging.
The Novel Approach
The novel approach detailed in patent CN1686976A overcomes these deficiencies through the strategic introduction of bulky anthracene and pyrene groups at the 9-position of the fluorene unit. This 9,9-diaryl substitution pattern introduces significant steric hindrance that physically separates the conjugated backbones, effectively suppressing the formation of interchain aggregates and excimers. By integrating these large aromatic systems, the material maintains the high fluorescence efficiency characteristic of fluorenes while simultaneously enhancing the thermal stability and glass transition temperature. The presence of anthracene and pyrene, which are known for their excellent hole-transporting capabilities, rectifies the carrier injection imbalance by raising the HOMO level closer to the anode work function. This structural modification results in a material that is not only more thermally robust but also electronically balanced, leading to significantly improved device efficiency and brightness.
From a manufacturing perspective, this approach offers a versatile platform for creating both discrete oligomers and extended polymers. The synthetic route allows for the precise installation of functional groups that can further tune solubility and film-forming properties, enabling solution-processing techniques like spin-coating. This compatibility with solution processing is a critical advantage for cost reduction in electronic chemical manufacturing, as it facilitates the use of inkjet printing and other scalable deposition methods. The resulting materials exhibit satisfactory results in terms of brightness and luminous efficiency, making them highly attractive candidates for next-generation full-color displays and lighting applications where performance consistency is paramount.
Mechanistic Insights into Suzuki and Yamamoto Coupling Polymerization
The synthesis of these advanced materials relies heavily on transition-metal catalyzed cross-coupling reactions, specifically the Suzuki and Yamamoto protocols, which are renowned for their ability to construct carbon-carbon bonds between aromatic units. The core mechanistic pathway begins with the preparation of the fluorene monomer, which involves a multi-step sequence starting with the oxidation of 2,7-dihalofluorene to the corresponding fluorenone. This oxidation is typically achieved using air in the presence of a phase transfer catalyst, a green chemistry approach that minimizes the use of harsh oxidants. Subsequently, the fluorenone undergoes nucleophilic addition with a Grignard reagent, generated in situ from an aryl halide and magnesium metal. This step installs the first aryl group at the 9-position, creating a tertiary alcohol intermediate that serves as the precursor for the final substitution.
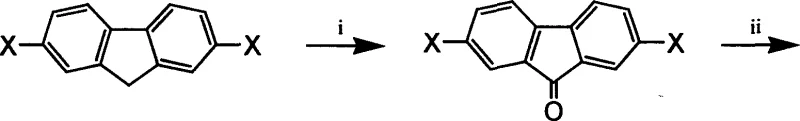
The final step in monomer synthesis involves a Friedel-Crafts alkylation where the tertiary alcohol reacts with a second large aromatic ring, such as anthracene or pyrene, under acidic catalysis. Strong organic acids like trifluoromethanesulfonic acid are employed to facilitate the dehydration and subsequent electrophilic aromatic substitution, locking the bulky groups into place. Once the monomer is secured, polymerization proceeds via the Suzuki coupling mechanism, which utilizes a palladium catalyst cycle involving oxidative addition, transmetallation, and reductive elimination. Alternatively, the Yamamoto coupling employs a nickel(0) catalyst to couple aryl halides directly, offering a complementary route for homopolymerization. These mechanisms allow for the controlled growth of the polymer chain, with the ability to introduce end-capping agents to terminate the reaction and define the molecular weight distribution precisely.
Understanding these mechanistic details is crucial for ensuring high purity and reproducibility in commercial production. The choice of ligands and bases in the Suzuki reaction, such as triphenylphosphine and potassium carbonate, influences the turnover frequency of the catalyst and the tolerance to functional groups. Similarly, the Yamamoto reaction conditions, utilizing Ni(COD)2 and bipyridine in DMF, require strict exclusion of oxygen and moisture to prevent catalyst deactivation. By optimizing these parameters, manufacturers can minimize the formation of homocoupling byproducts and ensure that the final polymer possesses the desired conjugation length and optoelectronic properties. This level of control is essential for meeting the stringent specifications required by R&D teams developing high-performance OLED devices.
How to Synthesize 9-Position Anthracene Fluorene Efficiently
The efficient synthesis of these conjugated derivatives requires a systematic approach that balances reaction yield with product purity. The process begins with the rigorous purification of starting materials, particularly the halogenated fluorenes, to prevent the incorporation of impurities that could act as quenching sites in the final device. The oxidation step to form the fluorenone must be monitored closely to avoid over-oxidation, while the Grignard reaction demands anhydrous conditions to ensure high conversion rates. Following the formation of the monomer, the polymerization step is critical; maintaining the correct stoichiometry between the dibromo monomer and the bis-boronate species is essential for achieving high molecular weights. Detailed standardized synthesis steps for scaling this process from laboratory to commercial production are outlined in the guide below.
- Oxidize 2,7-dihalofluorene using air and a phase transfer catalyst in pyridine to obtain the corresponding fluorenone intermediate.
- React the fluorenone with a Grignard reagent derived from aryl halides in anhydrous ether or THF to form the tertiary alcohol intermediate.
- Perform a Friedel-Crafts reaction between the alcohol and a large aromatic ring (anthracene or pyrene) using a strong organic acid catalyst to yield the monomer.
- Polymerize the monomer using Suzuki or Yamamoto coupling conditions with palladium or nickel catalysts to form the final conjugated derivative material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the synthetic routes described in patent CN1686976A presents several compelling economic and logistical advantages. The primary benefit stems from the use of readily available starting materials such as fluorene, anthracene, and pyrene, which are commodity chemicals produced at scale by the petrochemical industry. This abundance ensures a stable supply chain and mitigates the risk of raw material shortages that often plague the production of exotic specialty chemicals. Furthermore, the reliance on established coupling reactions like Suzuki and Yamamoto means that the manufacturing infrastructure required is already present in many fine chemical facilities, reducing the need for significant capital expenditure on new equipment. This compatibility with existing infrastructure accelerates the timeline from process development to commercial scale-up.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive and difficult-to-remove transition metal catalysts in certain steps, or utilizes catalysts that can be efficiently scavenged, thereby reducing downstream purification costs. The high yields reported in the patent examples, such as the 93% yield in the oxidation step and 91% in the Grignard addition, indicate a highly atom-economical process that minimizes waste generation. By avoiding complex protection-deprotection sequences often required in other conjugated polymer syntheses, the overall number of unit operations is reduced, leading to substantial cost savings in labor and solvent consumption. Additionally, the ability to produce soluble oligomers and polymers allows for solution processing, which is inherently less energy-intensive than vacuum deposition methods used for small molecules.
- Enhanced Supply Chain Reliability: The robustness of the Suzuki and Yamamoto coupling reactions contributes to high batch-to-batch consistency, a critical factor for maintaining supply chain reliability. The chemical stability of the 9,9-diaryl substituted fluorene backbone ensures that the materials have a long shelf life and can withstand transportation and storage conditions without degradation. This stability reduces the risk of inventory write-offs and ensures that customers receive materials that meet specification upon arrival. Moreover, the modular nature of the synthesis allows for the rapid substitution of different aryl groups if supply constraints arise for a specific precursor, providing flexibility in sourcing strategies without compromising the core performance characteristics of the material.
- Scalability and Environmental Compliance: The processes described are amenable to large-scale production, with reaction conditions that can be safely managed in standard stainless steel reactors. The use of phase transfer catalysts in the oxidation step enhances reaction rates and allows for milder conditions, improving safety profiles. Waste streams generated during the synthesis, primarily consisting of aqueous salts and organic solvents, can be managed through standard treatment protocols, facilitating compliance with increasingly stringent environmental regulations. The high thermal stability of the final product also contributes to sustainability by extending the lifespan of the electronic devices in which they are used, reducing electronic waste. This alignment with green chemistry principles and scalable manufacturing makes these materials a sustainable choice for long-term supply partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 9-position anthracene and pyrene-substituted fluorene derivatives. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these aspects is vital for making informed decisions about material integration and process optimization.
Q: What are the primary advantages of 9-position diaryl substituted fluorene derivatives?
A: According to patent CN1686976A, introducing large aromatic groups like anthracene or pyrene at the 9-position significantly improves thermal stability and spectral stability while maintaining high fluorescence efficiency. This structure effectively prevents the formation of aggregates and excimers that typically plague standard fluorene systems.
Q: Which coupling reactions are suitable for polymerizing these materials?
A: The patent specifies two primary methods: Suzuki coupling using palladium catalysts like Pd(PPh3)4 with potassium carbonate, and Yamamoto coupling using nickel catalysts like Ni(COD)2. Both methods allow for the precise control of molecular weight and end-capping to optimize optoelectronic properties.
Q: How does this material improve OLED device performance?
A: The large conjugated aromatic rings enhance hole injection and transport capabilities, addressing the mismatch between HOMO/LUMO levels and electrode work functions. This leads to balanced carrier injection, higher brightness, and improved luminous efficiency in electroluminescent devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Position Anthracene Fluorene Supplier
The technological potential of 9-position anthracene and pyrene-substituted fluorene derivatives is immense, offering a pathway to higher efficiency and longer-lasting organic electronic devices. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. Our rigorous QC labs are equipped to analyze these complex conjugated systems using advanced techniques such as GPC, LDI-TOF-MS, and cyclic voltammetry, guaranteeing that every batch delivers the consistent performance required for high-end OLED manufacturing. We understand the critical nature of supply continuity in the electronics sector and have optimized our logistics to support global demand.
We invite you to collaborate with us to leverage these advanced materials for your next-generation projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and purity requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain partner committed to driving innovation and efficiency in the field of display and optoelectronic materials.
