Advanced Organocatalytic Synthesis of ECPPA: Enhancing Selectivity and Commercial Viability for ACE Inhibitors
Introduction to Advanced ECPPA Manufacturing Technologies
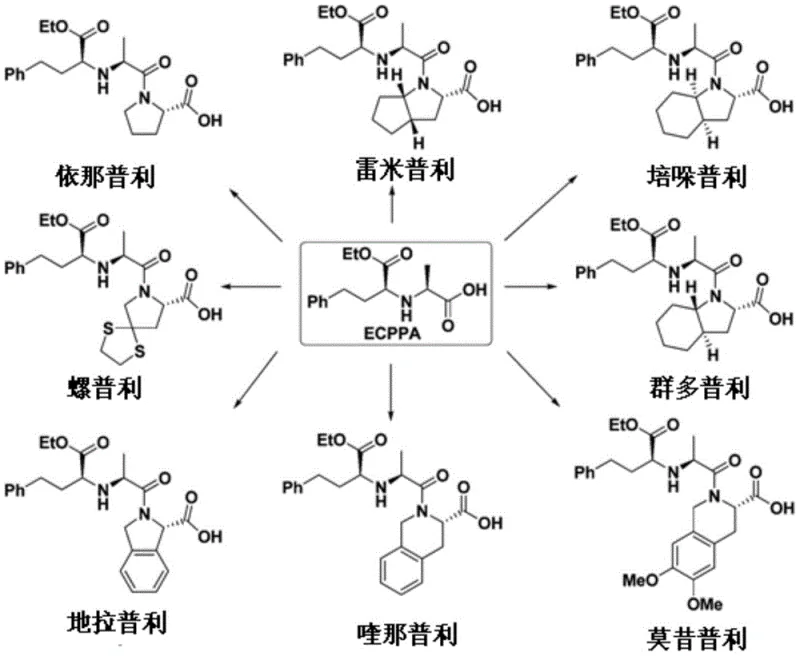
The pharmaceutical industry continuously seeks robust and scalable methods for producing critical intermediates, particularly for widely prescribed classes of drugs such as ACE inhibitors. Patent CN108147973B introduces a groundbreaking preparation method for N-[1-(S)-ethoxycarbonyl-3-phenylpropyl]-L-alanine, commonly known as ECPPA, which serves as a pivotal building block for numerous antihypertensive medications including Enalapril, Ramipril, and Perindopril. This innovation addresses long-standing challenges in stereoselectivity and process efficiency that have plagued traditional synthetic routes. By leveraging specific organocatalysts, namely thiourea and urea derivatives, the disclosed technology achieves exceptional control over chiral centers during the key carbon-carbon bond-forming step. For global procurement teams and R&D directors, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with consistent quality. The strategic implementation of this patented methodology not only enhances the chemical purity profile but also streamlines the manufacturing workflow, directly impacting the cost structure and supply chain resilience of downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ECPPA has been fraught with technical hurdles that complicate large-scale manufacturing and inflate production costs. Traditional routes, as illustrated in various prior art examples, often suffer from poor stereoselectivity, leading to difficult separations of diastereomers and a significant loss of valuable chiral material. For instance, some established methods rely on harsh hydrogenation conditions using concentrated sulfuric acid and acetic acid, which necessitate specialized corrosion-resistant equipment and pose severe safety risks during operation. Furthermore, the removal of acetic acid during work-up requires elevated temperatures under reduced pressure, creating a bottleneck in the production line. Other routes attempting to mitigate these issues by switching solvents to ethanol with p-toluenesulfonic acid have encountered problems with transesterification, where the benzyl protecting group is inadvertently exchanged, introducing impurities that drastically reduce the final hydrogenation yield to ranges as low as 74-76 wt%. These inefficiencies highlight the critical need for a more robust and selective synthetic strategy.
The Novel Approach
The novel approach detailed in the patent data revolutionizes the synthesis landscape by introducing a dual-catalyst system that meticulously controls reaction pathways. Instead of relying on brute-force chemical conditions, this method employs a first catalyst, selected from thiourea or urea compounds, to drive the initial addition reaction with high precision. This organocatalytic step ensures that the formation of the chiral center occurs with superior selectivity, effectively minimizing the generation of unwanted isomers right from the start. Subsequently, the hydrogenation step is refined by the inclusion of a catalytic activity inhibitor, which fine-tunes the reactivity of the heavy metal catalyst. This subtle yet powerful modification prevents the degradation of the ester moiety while efficiently removing the benzyl group and reducing the ketone functionality. The result is a streamlined process that boasts a short synthetic route and simple post-treatment operations, making it ideally suited for commercial scale-up of complex pharmaceutical intermediates without the baggage of legacy processing difficulties.
Mechanistic Insights into Thiourea-Catalyzed Asymmetric Addition
At the heart of this technological breakthrough lies a sophisticated understanding of non-covalent interactions and steric control within the reaction medium. The mechanism relies on the unique ability of the thiourea or urea catalyst to form a network of hydrogen bonds with the amino groups present in the L-alanine derivative. This interaction creates a bulky environment around the reactive imine intermediate, specifically increasing the steric hindrance on the Re face of the molecule. Consequently, the nucleophilic attack by the enamine species is energetically favored to occur from the less hindered Si face. This directional control is paramount for achieving the high diastereomeric ratios (dr > 99%) and enantiomeric excess (ee > 99%) reported in the experimental data. By dictating the spatial orientation of the reactants, the catalyst effectively acts as a molecular mold, ensuring that the resulting addition product possesses the exact stereochemical configuration required for biological activity in the final drug substance.
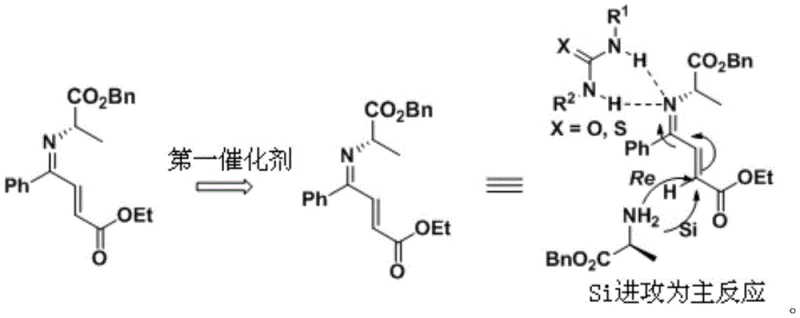
Beyond the initial addition, the mechanism extends to the hydrogenation phase where impurity control is equally critical. In traditional palladium-catalyzed hydrogenations, the high activity of the metal can lead to indiscriminate reduction and side reactions like transesterification. The introduction of a second thiourea compound as a catalytic activity inhibitor serves as a regulatory agent. It partially coordinates with the palladium surface or modifies the local electronic environment, dampening the catalyst's aggressiveness just enough to prevent ester scrambling while maintaining sufficient activity for the desired reduction of the ketone and removal of the benzyl group. This delicate balance ensures that the structural integrity of the ethoxycarbonyl group is preserved, thereby eliminating a major source of impurity that typically requires costly chromatographic purification. Such mechanistic elegance translates directly into a cleaner crude product profile, reducing the burden on downstream purification units.
How to Synthesize ECPPA Efficiently
Implementing this advanced synthesis route requires careful attention to catalyst loading and reaction conditions to maximize the benefits of the patented technology. The process begins with the preparation of the addition product under mild conditions, utilizing alcohol solvents and alkaline agents to facilitate the organocatalytic cycle. Following the isolation of this intermediate, the hydrogenation step is conducted in a pressurized reactor using specific alcohol solvents like isopropanol or t-amyl alcohol, alongside a controlled amount of acid and the activity inhibitor. The detailed standardized synthetic steps, including precise molar ratios, temperature profiles, and work-up procedures necessary to replicate the high yields described in the patent, are outlined in the guide below.
- Perform an asymmetric Michael addition between ethyl (E)-3-benzoylacrylate and L-alanine benzyl ester using a thiourea or urea catalyst to achieve high stereoselectivity.
- Conduct a hydrogenation reaction on the addition product using a palladium catalyst in the presence of an acid and a catalytic activity inhibitor to prevent transesterification.
- Purify the crude hydrogenation product through filtration, decolorization, and crystallization using solvents like methyl tert-butyl ether to obtain high-purity ECPPA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method offers tangible benefits that extend far beyond the laboratory bench. The primary advantage lies in the substantial simplification of the manufacturing process, which directly correlates to reduced operational expenditures and enhanced throughput. By eliminating the need for corrosive acids like concentrated sulfuric acid and avoiding complex multi-step sequences, the facility requirements become less stringent, allowing for production in standard stainless steel reactors rather than exotic alloy vessels. This shift significantly lowers the capital expenditure barrier and reduces maintenance costs associated with equipment corrosion. Furthermore, the high selectivity of the reaction means that less raw material is wasted in the form of incorrect isomers, leading to a more efficient utilization of starting materials and a reduction in the volume of chemical waste generated. These factors collectively contribute to a more sustainable and cost-effective supply chain model.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, combined with the high yield of the reaction, drives down the unit cost of production significantly. The use of organocatalysts, which are generally cheaper and easier to handle than many transition metal complexes used in asymmetric synthesis, further reduces the raw material bill. Additionally, the simplified work-up procedure, which avoids difficult distillation steps for acid removal, saves energy and labor hours. The prevention of transesterification side reactions means that the yield loss typically associated with impurity formation is avoided, ensuring that a higher percentage of input mass is converted into saleable product. This efficiency creates a buffer against raw material price volatility, providing greater financial stability for long-term contracts.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent and reliable supply of high-purity ECPPA, which is critical for maintaining uninterrupted API production schedules. The tolerance of the reaction conditions allows for flexibility in sourcing solvents and reagents, reducing the risk of supply disruptions due to single-source dependencies. The high purity of the crude product (>98%) minimizes the need for extensive reprocessing or third-party purification services, shortening the overall lead time from synthesis to shipment. This reliability is essential for pharmaceutical companies managing tight inventory levels and Just-In-Time manufacturing strategies, ensuring that downstream production lines never stall due to intermediate shortages.
- Scalability and Environmental Compliance: The method is explicitly designed with industrial production in mind, featuring steps that are easily scalable from kilogram to multi-ton quantities without losing efficiency. The use of greener solvents and the avoidance of heavy metal contamination in the early stages align with increasingly strict environmental regulations and corporate sustainability goals. The reduced generation of hazardous waste simplifies disposal protocols and lowers compliance costs. Moreover, the mild reaction conditions enhance operator safety, reducing the risk of accidents and associated downtime. This alignment with green chemistry principles not only future-proofs the supply chain against regulatory changes but also enhances the brand reputation of the manufacturers involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of ECPPA using this patented methodology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical implementation of the technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing portfolios. The answers reflect the specific advantages of the thiourea-catalyzed route over legacy methods.
Q: How does the thiourea catalyst improve the stereoselectivity of ECPPA synthesis?
A: The thiourea catalyst forms hydrogen bonds with the amino groups of the substrate, increasing steric hindrance on the Re face of the imine intermediate. This forces the nucleophilic attack to occur predominantly from the Si face, significantly enhancing the diastereomeric ratio (dr) and enantiomeric excess (ee) of the addition product.
Q: What is the role of the catalytic activity inhibitor in the hydrogenation step?
A: The catalytic activity inhibitor, typically a second thiourea compound, moderates the activity of the palladium catalyst. This prevents unwanted side reactions such as transesterification, where the benzyl ester might exchange with the solvent, thereby maintaining the structural integrity of the molecule and improving the overall yield of the final ECPPA product.
Q: Why is this new method considered superior to conventional routes for industrial production?
A: Unlike conventional routes that often require harsh conditions like concentrated sulfuric acid or complex multi-step sequences with poor selectivity, this method utilizes mild organocatalysis and controlled hydrogenation. This results in a shorter synthetic route, simpler post-treatment operations, and higher overall yields, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ECPPA Supplier
As the global demand for cardiovascular medications continues to rise, securing a stable source of high-quality intermediates like ECPPA is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic technologies to deliver products that meet the rigorous standards of the international pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We operate with stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest benchmarks of quality and safety. Our commitment to excellence ensures that every gram of ECPPA supplied contributes to the efficacy and safety of the final therapeutic product.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our advanced processes can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us partner with you to drive innovation and efficiency in your API manufacturing operations, ensuring a competitive edge in the global marketplace.
