Scalable Manufacturing of High-Purity Cefotiam Axetil for Global Pharmaceutical Markets
Scalable Manufacturing of High-Purity Cefotiam Axetil for Global Pharmaceutical Markets
The global demand for effective oral cephalosporins continues to drive innovation in the synthesis of key pharmaceutical intermediates. A significant advancement in this field is detailed in patent CN101993449B, which outlines a robust preparation method for high-purity cefotiam axetil and its dihydrochloride salt. This technology addresses critical bottlenecks in the production of this vital antibiotic prodrug, offering a pathway to superior product quality and process efficiency. Cefotiam axetil serves as an orally active prodrug that rapidly hydrolyzes into the active antibiotic cefotiam within the intestinal mucosa, providing broad-spectrum activity against Gram-positive and Gram-negative bacteria. For R&D directors and procurement specialists, understanding the nuances of this synthesis is paramount for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent international quality standards.
The patented methodology represents a substantial leap forward in process chemistry, moving away from labor-intensive purification techniques towards a streamlined, crystallization-driven approach. By optimizing reaction conditions and solvent systems, the process achieves high yields while maintaining impurity profiles well within the limits set by major pharmacopoeias, including the Japanese Pharmacopoeia. This level of control is essential for ensuring the safety and efficacy of the final drug product, making it a cornerstone technology for manufacturers aiming to reduce costs in antibiotic manufacturing without compromising on quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of cefotiam axetil was plagued by significant inefficiencies that hindered large-scale production. Historical literature, such as the work by Yoshinobu et al. (1986), described methods involving the reaction of cefotiam potassium with 1-iodoethyl cyclohexyl carbonate in DMF. While chemically feasible, these early processes suffered from critically low yields, often necessitating multiple recrystallization steps to achieve acceptable purity. Furthermore, alternative methods reported by Nishimura et al. relied on column chromatography for purification, a technique that is notoriously difficult to scale industrially due to high solvent consumption, slow throughput, and significant product loss. These conventional approaches also struggled to control the formation of the Δ2 isomer, a key impurity that frequently exceeded the 2% threshold mandated by regulatory bodies, thereby rendering batches unsuitable for clinical use.
The Novel Approach
In stark contrast, the method disclosed in patent CN101993449B introduces a highly efficient esterification strategy that bypasses the need for chromatographic purification entirely. The core of this innovation lies in the precise control of reaction stoichiometry and the implementation of a sophisticated crystallization protocol. By reacting cefotiam salts with 1-iodoethyl cyclohexyl carbonate in the presence of a carbonate base within specific organic solvents like DMA or DMF, the process achieves conversion rates that significantly outperform historical benchmarks. The subsequent workup and crystallization steps are engineered to selectively isolate the target molecule, effectively suppressing the Δ2 isomer and other by-products. This shift from complex separation techniques to direct crystallization not only simplifies the operational workflow but also drastically enhances the overall economic viability of producing high-purity cefotiam axetil.
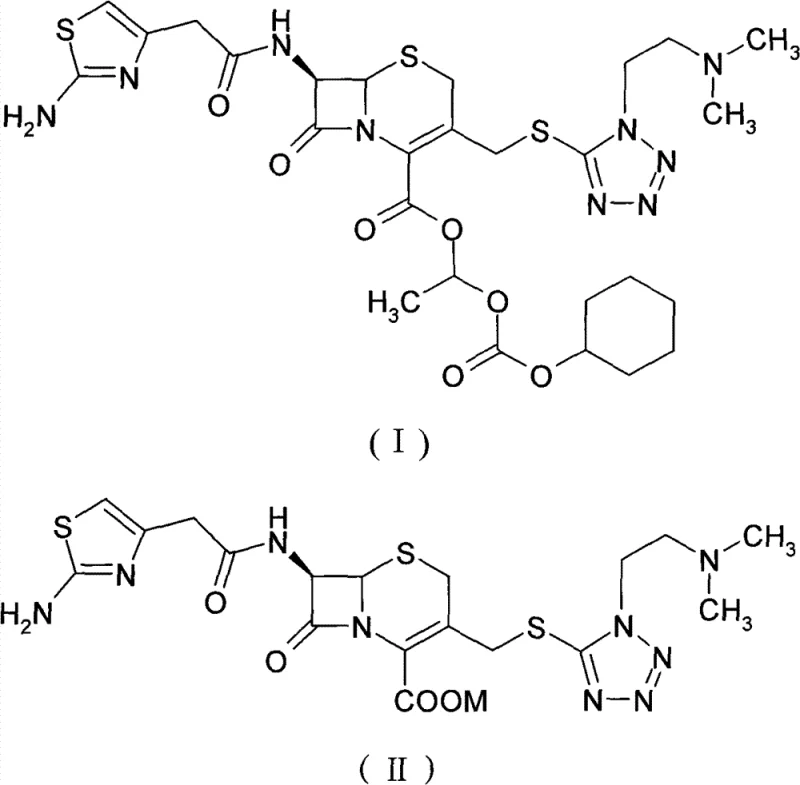
Mechanistic Insights into Carbonate-Mediated Esterification
The chemical transformation at the heart of this process is a nucleophilic substitution reaction where the carboxylate anion of the cefotiam salt attacks the electrophilic carbon of the 1-iodoethyl cyclohexyl carbonate. The presence of a carbonate base, preferably potassium carbonate, is critical for generating the reactive nucleophile from the cefotiam salt precursor. The reaction is conducted at low temperatures, typically between -15°C and 5°C, to minimize side reactions and thermal degradation of the sensitive beta-lactam ring. This temperature control is vital for preserving the structural integrity of the cephem nucleus, which is prone to opening under harsh conditions. The choice of solvent, such as N,N-dimethylacetamide (DMA), facilitates the dissolution of both the ionic salt and the organic esterifying agent, creating a homogeneous reaction medium that promotes efficient molecular collisions and high conversion rates.
Following the reaction, the control of impurities, particularly the Δ2 isomer, becomes the primary focus of the downstream processing. The Δ2 isomer arises from the migration of the double bond within the dihydrothiazine ring, a common degradation pathway for cephalosporins. The patented process mitigates this through a carefully designed crystallization regime. By dissolving the crude product in a "benign dissolving solvent" (such as methanol or acetone) and introducing a "crystallization solvent" (such as isopropyl ether) at controlled temperatures (15-20°C), the system exploits subtle differences in solubility between the target isomer and its impurities. This thermodynamic control ensures that the desired cefotiam axetil precipitates with high fidelity, leaving the Δ2 isomer and other contaminants in the mother liquor. This mechanism is key to achieving the reported purity levels of over 98% and total impurities below 4%.
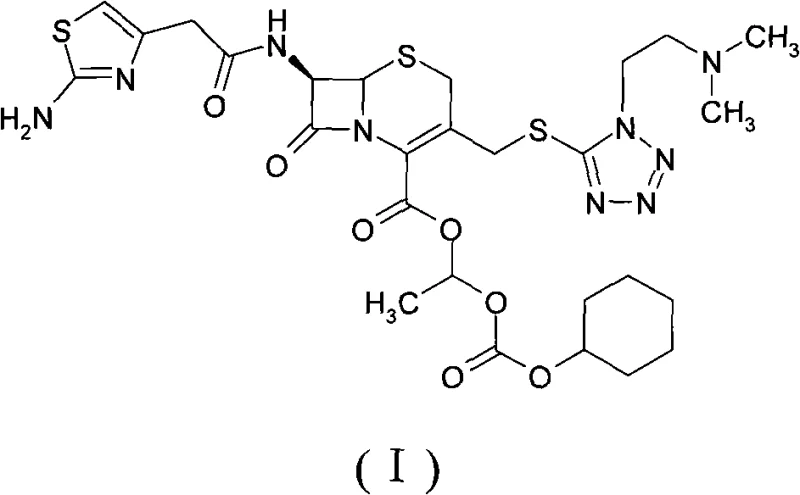
How to Synthesize Cefotiam Axetil Efficiently
The synthesis of cefotiam axetil via this patented route involves a sequence of well-defined unit operations that are amenable to standard pharmaceutical manufacturing equipment. The process begins with the preparation of the reaction mixture under inert atmosphere to prevent moisture ingress, followed by the controlled addition of reagents to manage exotherms. After the reaction reaches completion, a multi-stage extraction and washing protocol removes inorganic salts and unreacted starting materials. The final and most critical step is the crystallization, where the choice of solvent ratios and cooling rates determines the final crystal habit and purity. For a detailed breakdown of the specific operational parameters, reagent grades, and safety considerations required to execute this synthesis, please refer to the standardized guide below.
- React cefotiam salt (potassium or sodium) with 1-iodoethyl cyclohexyl carbonate in an organic solvent like DMF or DMA in the presence of a carbonate base at low temperatures (-15 to 5°C).
- Perform aqueous workup involving extraction with ethyl acetate, acid washing, and pH adjustment to isolate the crude ester product.
- Purify the crude product via crystallization using a mixture of benign dissolving solvents (e.g., methanol, acetone) and crystallization solvents (e.g., isopropyl ether) at controlled temperatures (15-20°C).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible strategic benefits that extend beyond mere technical specifications. The elimination of column chromatography is a game-changer, as it removes a major bottleneck that typically constrains production capacity and inflates operational expenditures. By relying on crystallization for purification, the process reduces solvent usage and waste generation, aligning with modern green chemistry principles and reducing the environmental footprint of manufacturing. This simplification of the workflow translates directly into enhanced supply chain reliability, as the risk of batch failure due to complex purification steps is significantly minimized. Furthermore, the use of commodity chemicals and standard reactors ensures that the commercial scale-up of complex beta-lactams can be achieved rapidly without the need for specialized or custom-built infrastructure.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of chromatographic purification. Column chromatography is not only slow but also consumes vast quantities of silica gel and solvents, which are costly to purchase and dispose of. By replacing this with a crystallization-based purification, the process drastically cuts down on material costs and processing time. Additionally, the improved yield, reported to be around 60% compared to historical lows of 20%, means that less raw material is required to produce the same amount of finished goods. This efficiency gain allows for a substantial reduction in the cost of goods sold (COGS), providing a competitive edge in pricing negotiations for bulk pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that are sensitive to minor variations or rely on scarce reagents. This patented method utilizes widely available starting materials such as cefotiam salts and common organic solvents like DMA and ethyl acetate, reducing the risk of raw material shortages. The robustness of the crystallization step ensures consistent product quality across different batches, which is critical for maintaining regulatory compliance and avoiding costly recalls. By stabilizing the production output, manufacturers can offer more reliable delivery schedules to their clients, effectively reducing lead time for high-purity cephalosporins and strengthening long-term partnerships with global pharmaceutical companies.
- Scalability and Environmental Compliance: Scaling a chemical process from the laboratory to industrial production often reveals hidden challenges, but this method is explicitly designed for scalability. The absence of specialized equipment requirements means that existing manufacturing facilities can be adapted with minimal capital investment. Moreover, the reduced solvent load and elimination of solid waste associated with silica gel columns simplify waste treatment protocols. This ease of handling hazardous materials and waste streams ensures better compliance with increasingly strict environmental regulations, safeguarding the manufacturer against potential fines and operational shutdowns while promoting a sustainable production model.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of cefotiam axetil using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: How does this new method improve upon traditional cefotiam axetil synthesis?
A: Traditional methods reported in literature often suffer from low yields (around 20%) and require cumbersome column chromatography for purification. The patented process eliminates the need for column chromatography, achieves yields around 60%, and ensures total impurities remain below 4%, specifically controlling the Δ2 isomer to meet pharmacopoeia standards.
Q: What are the critical parameters for controlling the Δ2 isomer impurity?
A: Controlling the Δ2 isomer is crucial for meeting Japanese Pharmacopoeia standards (<2%). This is achieved through precise temperature control during the reaction (-15 to 5°C) and, more importantly, through a optimized crystallization step using specific solvent systems like methanol/isopropyl ether mixtures at 15-20°C, which selectively precipitates the desired isomer.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scalability. It avoids special equipment and complex purification steps like freeze-drying or column chromatography. The use of common organic solvents (DMA, DMF, ethyl acetate) and standard crystallization techniques makes it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotiam Axetil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cefotiam axetil meets or exceeds international pharmacopoeial standards. Our commitment to excellence extends beyond mere compliance; we strive to be a strategic partner who understands the complexities of your supply chain and delivers value through superior product performance.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to verify specific COA data and route feasibility assessments for your next campaign, we are ready to provide the data and expertise you need. Contact us today to explore a partnership that combines technical innovation with commercial reliability, ensuring a secure and efficient supply of high-purity cefotiam axetil for your global operations.
