Advanced Synthesis of 4-(p-Acetamidobenzenesulfonamide)-6-methoxypyrimidine for Commercial Scale-up
Advanced Synthesis of 4-(p-Acetamidobenzenesulfonamide)-6-methoxypyrimidine for Commercial Scale-up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical intermediates. A prime example of such innovation is found in Chinese Patent CN115677599A, which discloses a highly efficient method for preparing 4-(p-acetamidobenzenesulfonamide)-6-methoxypyrimidine. This compound serves as a pivotal precursor in the manufacture of Sulfa-6-methoxypyrimidine sodium, a potent veterinary antibiotic known for its broad-spectrum efficacy against Gram-positive and Gram-negative bacteria. The structural integrity and purity of this intermediate are paramount, as they directly influence the therapeutic efficacy of the final drug substance. As depicted in the molecular structure below, the molecule features a delicate balance of sulfonamide and pyrimidine moieties that require precise chemical handling to avoid degradation or side reactions during synthesis.
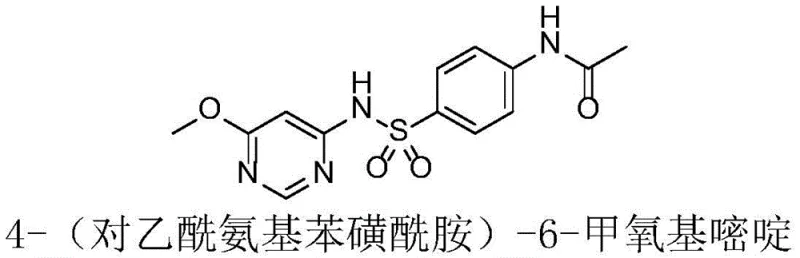
For R&D directors and process chemists, the significance of this patent lies in its departure from hazardous legacy chemistries towards a greener, more controllable process. The patent outlines a condensation reaction between 4-amino-6-methoxypyrimidine and 4-acetamidobenzenesulfonyl chloride, utilizing solvents such as dichloromethane, tetrahydrofuran, or N,N-dimethylformamide. This strategic shift not only enhances the reaction yield but also simplifies the downstream purification process, ensuring that the final product meets the stringent purity specifications required for veterinary applications. By understanding the nuances of this patented technology, manufacturers can secure a reliable supply chain for high-quality pharmaceutical intermediates while adhering to increasingly strict global environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN115677599A, the synthesis of this key intermediate was plagued by significant technical and environmental hurdles. Earlier patents, such as CN108276346, relied on a one-pot method involving carbon tetrachloride and chlorosulfonic acid. While this approach simplified the operational steps, it introduced severe safety risks due to the use of carbon tetrachloride, a solvent largely banned in modern pharmaceutical synthesis due to its hepatotoxicity and ozone-depleting potential. Furthermore, the use of chlorosulfonic acid posed substantial corrosion challenges for reactor equipment and required specialized handling protocols to manage its intense exothermic nature and irritating fumes. Another prevalent method, documented in CN101565418 and CN105294576, utilized pyridine as both the solvent and the acid scavenger. Although this avoided chlorosulfonic acid, it resulted in dismal yields of approximately 53% and created massive environmental burdens. The strong, pungent odor of pyridine necessitated expensive scrubbing systems, and its tendency to form azeotropes with water made solvent recovery energy-intensive and technically difficult, often leading to cross-contamination in subsequent hydrolysis steps.
The Novel Approach
The methodology presented in the current patent represents a paradigm shift in process chemistry for sulfonamide derivatives. By replacing the problematic pyridine solvent system with safer alternatives like dichloromethane (DCM), tetrahydrofuran (THF), or N,N-dimethylformamide (DMF), the process achieves a dramatic improvement in both yield and operational safety. The reaction scheme illustrated below demonstrates the direct condensation of the amine and sulfonyl chloride components under mild conditions. This new route eliminates the need for corrosive chlorosulfonic acid entirely, relying instead on pre-formed 4-acetamidobenzenesulfonyl chloride, which is more stable and easier to handle. The use of catalytic or stoichiometric amounts of milder acid scavengers, such as triethylamine or 2-picoline, further reduces the chemical load on the waste stream. Most critically, this solvent switch facilitates a much cleaner crystallization process, allowing the product to precipitate with high purity (>99%) simply by the addition of polar anti-solvents like methanol or ethanol, thereby bypassing complex chromatographic purifications.
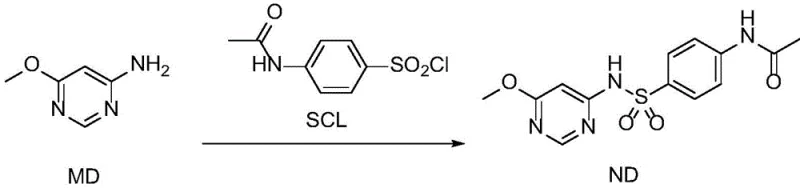
Mechanistic Insights into Sulfonamide Condensation
The core of this synthesis is a nucleophilic substitution reaction where the primary amine group of 4-amino-6-methoxypyrimidine attacks the electrophilic sulfur atom of the sulfonyl chloride. This mechanism is highly sensitive to the reaction environment, particularly the presence of moisture and the basicity of the medium. In the optimized protocol, the reaction is conducted at a controlled temperature range of 10-30°C. Maintaining this thermal window is crucial; temperatures that are too low may result in incomplete conversion of the starting materials, while excessive heat can promote the hydrolysis of the sulfonyl chloride into the corresponding sulfonic acid, a difficult-to-remove impurity that degrades the overall yield. The choice of solvent plays a dual role here: it must solubilize both organic reactants effectively while remaining inert to the sulfonyl chloride functionality. Dichloromethane, for instance, provides excellent solubility for the aromatic components without participating in side reactions, creating an ideal medium for the nucleophilic attack to proceed to completion.
Impurity control is further managed through the precise addition of the acid scavenger. As the reaction proceeds, hydrogen chloride gas is generated as a byproduct. If not neutralized immediately, this acid can protonate the amine reactant, rendering it non-nucleophilic and stalling the reaction. The patent specifies the use of bases like pyridine, triethylamine, or piperidine in molar ratios of 1:1 to 1:2 relative to the amine. This ensures that the HCl is scavenged efficiently, driving the equilibrium towards product formation. Furthermore, the subsequent crystallization step acts as a powerful purification mechanism. By adding anti-solvents like methanol in batches with specific time intervals (8-10 minutes), the process controls the supersaturation level, promoting the growth of large, pure crystals while leaving soluble impurities in the mother liquor. This physical separation technique is far more scalable and cost-effective than chemical purification methods, ensuring the final API intermediate meets the rigorous quality standards demanded by veterinary pharmaceutical manufacturers.
How to Synthesize 4-(p-Acetamidobenzenesulfonamide)-6-methoxypyrimidine Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding solvent ratios and temperature control to maximize yield and purity. The process begins by dissolving the amine substrate in the chosen reaction solvent, followed by the controlled addition of the sulfonyl chloride and the acid scavenger. The reaction mixture is then stirred for a defined period to ensure complete conversion before initiating the isolation phase. The crystallization step is particularly critical, involving the batch-wise addition of a polar anti-solvent to induce precipitation, followed by a cooling cycle to maximize recovery. For detailed operational parameters, including specific stirring rates, filtration techniques, and drying conditions necessary to achieve the reported 93%+ yields, please refer to the standardized protocol outlined below.
- Dissolve 4-amino-6-methoxypyrimidine in a selected solvent (DCM, THF, or DMF) at a mass ratio of 1: 4 to 1:6.
- Add 4-acetamidobenzenesulfonyl chloride (molar ratio 1: 1.0 to 1:1.5) and dropwise add an acid scavenger like pyridine or triethylamine at 10-30°C.
- Precipitate the product by adding a polar anti-solvent (methanol or ethanol) in batches, followed by cooling to 0-20°C for crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible economic and logistical benefits that extend beyond simple yield improvements. The elimination of banned solvents like carbon tetrachloride and the reduction of pyridine usage significantly lowers the cost of waste disposal and environmental compliance. Traditional methods incurred high overheads due to the need for specialized scrubbing systems to handle pyridine vapors and the complex distillation processes required to separate pyridine from water azeotropes. By switching to common industrial solvents like dichloromethane or THF, which have well-established recovery infrastructures, manufacturers can drastically simplify their solvent management protocols. This simplification translates directly into reduced operational expenditures and a lower total cost of ownership for the manufacturing process, making the final intermediate more price-competitive in the global market.
- Cost Reduction in Manufacturing: The most significant driver of cost reduction in this process is the substantial increase in reaction yield, which jumps from roughly 53% in older pyridine-based methods to over 90% in the new protocol. This near-doubling of efficiency means that for every ton of raw material purchased, the output of saleable product is significantly higher, effectively lowering the unit cost of goods sold. Additionally, the avoidance of corrosive reagents like chlorosulfonic acid reduces maintenance costs for reactor vessels and piping, extending the lifespan of capital equipment. The simplified work-up procedure, which relies on straightforward crystallization rather than complex extractions or chromatography, also reduces labor hours and energy consumption per batch, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are readily available from multiple global suppliers. Solvents such as dichloromethane, methanol, and triethylamine are produced at massive scales for various industries, ensuring that production is never halted due to raw material shortages. In contrast, reliance on specialized or hazardous reagents can introduce single points of failure in the supply network. Furthermore, the robustness of the new process, with its wider operating temperature window (10-30°C) and tolerance for standard industrial equipment, reduces the risk of batch failures. This reliability allows for more accurate production planning and shorter lead times, enabling suppliers to respond more agilely to fluctuations in market demand for veterinary antibiotics.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden bottlenecks, particularly regarding heat transfer and safety. This patented method is inherently scalable because it avoids highly exothermic reactions that are difficult to control in large reactors. The use of less toxic solvents aligns with Green Chemistry principles, facilitating easier regulatory approval in regions with strict environmental laws, such as the EU and North America. The ability to recycle solvents efficiently without forming difficult azeotropes means that the process generates less hazardous waste, reducing the environmental footprint of the facility. This compliance advantage is critical for maintaining an uninterrupted supply license and avoiding potential fines or shutdowns associated with environmental violations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this intermediate. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this material into your existing supply chain or R&D pipeline.
Q: Why is the new solvent system superior to pyridine for this sulfonamide synthesis?
A: Traditional methods using pyridine as a solvent resulted in low yields (around 53%) and difficult solvent recovery due to azeotrope formation with water. The new method uses solvents like dichloromethane which allow for easier recovery and significantly higher yields exceeding 90%.
Q: What are the critical temperature controls for minimizing impurities?
A: The condensation reaction should be strictly maintained between 10-30°C. Temperatures outside this range can lead to hydrolysis of the sulfonyl chloride or incomplete reaction, affecting the final purity which targets >99%.
Q: Is this process scalable for industrial production of veterinary intermediates?
A: Yes, the process avoids hazardous reagents like chlorosulfonic acid and banned solvents like carbon tetrachloride, making it environmentally compliant and safe for large-scale manufacturing up to 100 MT annual capacity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(p-Acetamidobenzenesulfonamide)-6-methoxypyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final drug product is inextricably linked to the purity of its precursors. Our technical team has extensively analyzed the pathway described in CN115677599A and possesses the expertise to execute this synthesis with precision. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including HPLC analysis to confirm the absence of critical impurities such as hydrolyzed sulfonic acids or unreacted amines. We are committed to delivering high-purity pharmaceutical intermediates that empower our partners to develop safer and more effective veterinary medicines.
We invite you to collaborate with us to optimize your sourcing strategy for this vital intermediate. Our team is prepared to provide a Customized Cost-Saving Analysis that demonstrates how switching to our optimized supply chain can reduce your overall manufacturing costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities for your evaluation. Let us partner with you to drive efficiency and quality in your pharmaceutical production.
