Advanced Enzymatic Synthesis of Rimegepant Intermediate VI for Commercial Scale-Up
Introduction to Patent CN115976130A and Technological Breakthroughs
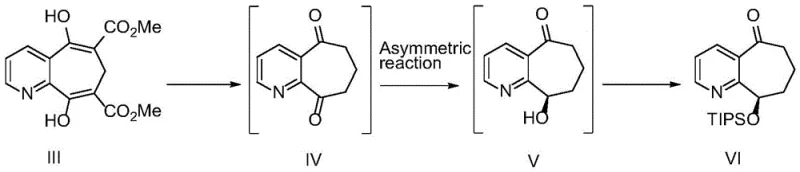
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex small molecules, particularly for migraine treatments like Rimegepant. Patent CN115976130A discloses a highly efficient synthesis process for Compound VI, a critical chiral intermediate in the production of Rimegepant. This technology represents a significant leap forward by replacing traditional transition metal catalysis with a novel biocatalytic approach. The core innovation lies in the use of a specific ketoreductase (amino acid sequence SEQ ID No.1) to introduce the chiral hydroxyl group with exceptional stereoselectivity. Unlike previous methods that struggled with impurity profiles and harsh reaction conditions, this new process utilizes a two-step telescopic strategy that bypasses the purification of intermediates, thereby streamlining the workflow. The result is a three-step overall yield reaching 90.1%, with the final product demonstrating purity levels exceeding 99.9%. For global supply chain stakeholders, this patent offers a blueprint for cost reduction in API manufacturing by simplifying unit operations and enhancing material throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing the core skeleton of Rimegepant have historically relied heavily on precious metal catalysis, presenting substantial barriers to industrial adoption. For instance, earlier reports described the use of Rh-(R-Binapine)(COD)BF4 complexes to achieve asymmetric reduction. While effective in laboratory settings, these metal-based systems suffer from severe drawbacks including the high cost of rhodium, the instability of the catalyst, and the difficulty in sourcing specialized ligands like R-Binapine. Furthermore, these reactions often necessitate hazardous reagents such as hexachlorodisilane and require high-pressure hydrogenation conditions up to 100 psi, which impose rigorous safety protocols and equipment costs on manufacturing facilities. Alternative enzymatic attempts using older strains like ES-KRED-119 resulted in suboptimal outcomes, generating up to 7% of structural impurities and achieving only 81% single-step yields. These inefficiencies accumulate, leading to higher production costs and complex waste streams that complicate regulatory compliance and environmental sustainability efforts.
The Novel Approach
The methodology outlined in CN115976130A fundamentally reengineers the synthesis pathway to overcome these historical bottlenecks through biocatalysis and process intensification. By employing a mutated ketoreductase derived from Arthrobacter sp. TS-15, the process achieves a conversion rate greater than 99% and an enantiomeric excess (ee) value exceeding 99.5% under mild conditions (20-30°C, atmospheric pressure). A key strategic advantage is the implementation of telescopic reactions, where the crude products of the acid hydrolysis (Compound IV) and the enzymatic reduction (Compound V) are carried forward directly without isolation or chromatographic purification. This elimination of intermediate workup steps drastically reduces solvent consumption and processing time. The final protection step utilizes standard silylation reagents like TIPSCl, followed by a robust salt-formation purification using camphorsulfonic acid, ensuring that the final free base meets strict pharmacopeial standards. This approach not only enhances safety by removing high-pressure steps but also significantly improves the economic viability of large-scale production.
Mechanistic Insights into Ketoreductase-Catalyzed Asymmetric Reduction
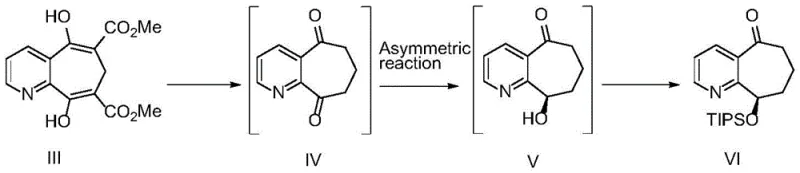
The heart of this synthetic advancement is the specific biocatalytic mechanism employed to establish the chiral center at the 9-position of the cyclohepta[b]pyridine ring. The ketoreductase utilized (SEQ ID No.1) facilitates the hydride transfer from the cofactor NADPH to the prochiral ketone substrate (Compound IV) with precise stereocontrol. This enzymatic pocket is engineered to favor the formation of the (9R)-hydroxy configuration, effectively suppressing the formation of the undesired (9S)-enantiomer. The reaction system incorporates a cofactor regeneration cycle using glucose and glucose dehydrogenase, which maintains the necessary concentration of reduced nicotinamide adenine dinucleotide phosphate (NADPH) throughout the reaction duration of 4 to 5 hours. Operating within a buffered aqueous environment at pH 6.0 to 8.0 ensures enzyme stability and optimal activity. This biological precision contrasts sharply with chemical catalysts that often require cryogenic temperatures or exotic ligands to achieve similar selectivity, highlighting the superiority of modern protein engineering in fine chemical synthesis.
Beyond stereochemistry, the process demonstrates remarkable control over structural impurities, which is critical for regulatory approval. The primary impurities identified in this pathway include the diol Compound VII and its derivatives, as well as the chiral impurity VI'. The enzymatic specificity minimizes the over-reduction to the diol, while the subsequent telescoped silylation and salt formation steps act as powerful purification drivers. Even if the crude enzymatic product contains trace amounts of chiral impurities (up to 2%), the downstream processing effectively purges these to levels below 0.15% in the final API starting material. The fate of impurities is meticulously tracked, ensuring that potential genotoxic or reactive species like Compound VIII are maintained below the threshold of 0.10%. This comprehensive impurity management strategy ensures that the final output aligns with ICH Q3A guidelines for drug substances, reducing the risk of late-stage development failures due to purity issues.
How to Synthesize (R)-9-Triisopropylsilyloxy-6,7,8,9-tetrahydro-5H-cyclohepta[b]pyridin-5-one Efficiently
The practical execution of this synthesis involves a streamlined sequence designed for maximum efficiency and minimal handling. The process begins with the acid-mediated cyclization of the precursor diester to form the dione, followed immediately by the biocatalytic reduction in a single pot or telescoped manner. The detailed standardized operating procedures, including specific reagent grades, mixing rates, and quality control checkpoints, are essential for reproducing the high yields reported in the patent data. Manufacturers aiming to adopt this route should focus on maintaining strict temperature control during the enzymatic step to preserve catalyst activity and ensure consistent stereochemical outcomes.
- Hydrolyze compound III in acidic solution (e.g., 7N HCl) at 85-95°C to form crude compound IV without purification.
- Catalytically reduce crude compound IV using ketoreductase (SEQ ID No.1) with NADP cofactor recycling to generate chiral alcohol compound V.
- Protect the hydroxyl group of crude compound V using TIPSCl and DBU in acetonitrile, followed by salt formation and free basing to isolate pure compound VI.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic telescoped process translates into tangible strategic benefits regarding cost, reliability, and scalability. The shift away from precious metal catalysts removes a significant variable cost driver and mitigates supply risks associated with rare earth elements. Additionally, the simplification of the workflow by eliminating intermediate purification steps reduces the overall manufacturing cycle time and facility occupancy, allowing for higher throughput within existing infrastructure. The robustness of the enzyme under ambient conditions also lowers energy consumption compared to high-pressure hydrogenation processes, contributing to a greener manufacturing profile that aligns with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive rhodium catalysts and specialized ligands results in substantial raw material savings. Furthermore, the telescopic nature of the synthesis reduces solvent usage and waste disposal costs by avoiding multiple isolation and drying steps. The high overall yield of 90.1% maximizes the utility of starting materials, directly improving the cost of goods sold (COGS) for the final intermediate. By avoiding the need for complex chromatographic purification, the process also lowers capital expenditure requirements for specialized equipment.
- Enhanced Supply Chain Reliability: Biocatalysts can be produced consistently via fermentation, offering a more stable supply source compared to chemically synthesized metal complexes which may face geopolitical or logistical constraints. The mild reaction conditions reduce the risk of batch failures due to equipment malfunction or safety incidents, ensuring more predictable delivery schedules. The ability to source generic reagents like TIPSCl and glucose further diversifies the supply base, reducing dependency on single-source vendors for critical inputs.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard reactor configurations without the need for high-pressure vessels. The aqueous nature of the enzymatic step minimizes the release of volatile organic compounds (VOCs), simplifying environmental permitting and waste treatment. The high purity of the final product reduces the burden on downstream API synthesis, preventing impurity carryover that could compromise final drug quality. This alignment with green chemistry principles enhances the long-term viability of the supply chain in an increasingly regulated market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for R&D teams evaluating technology transfer and procurement teams assessing vendor capabilities. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for decision-making processes.
Q: What are the advantages of using ketoreductase over rhodium catalysts for this synthesis?
A: The enzymatic route eliminates the need for expensive and unstable rhodium catalysts like Rh-(R-Binapine)(COD)BF4, avoids high-pressure hydrogenation conditions (100 psi), and achieves superior chiral purity (>99.5% ee) with simpler operational requirements suitable for industrial scale-up.
Q: How does this process control critical impurities like Compound VIII and VI'?
A: The specific ketoreductase (SEQ ID No.1) demonstrates high stereoselectivity, minimizing the formation of the chiral impurity VI'. Furthermore, the telescoped process and subsequent purification via camphorsulfonic acid salt formation effectively reduce structural impurity VIII to below 0.10% and chiral impurities to within 0.15%.
Q: What is the overall yield and purity of the final intermediate VI?
A: The three-step telescoped process achieves a total yield of up to 90.1% from compound III. The final product exhibits an HPLC purity greater than 99.9%, with no single unknown impurity exceeding 0.10%, meeting stringent regulatory standards for starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rimegepant Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development of CGRP antagonists like Rimegepant. Our technical team has thoroughly analyzed the advancements presented in CN115976130A and possesses the expertise to implement this enzymatic route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. Our facilities are equipped with state-of-the-art biocatalysis reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to confirm ee values greater than 99.5%.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project timelines and budget constraints. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this biocatalytic method. We are prepared to provide specific COA data from our pilot batches and conduct comprehensive route feasibility assessments to demonstrate our commitment to delivering reliable, high-purity pharmaceutical intermediates that meet global regulatory standards.
