Advanced Manufacturing of Cefoperazone Sodium: A Green, High-Yield Synthetic Route for Global Pharma Supply Chains
Advanced Manufacturing of Cefoperazone Sodium: A Green, High-Yield Synthetic Route for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable methods for producing critical antibiotics. Patent CN102372729A introduces a groundbreaking approach to synthesizing Cefoperazone Sodium, a third-generation cephalosporin antibiotic widely used for its broad-spectrum activity against both Gram-positive and Gram-negative bacteria. This novel method addresses long-standing challenges in cephalosporin manufacturing, specifically focusing on the use of green solvents and mild reaction conditions to enhance both yield and purity. By leveraging dimethyl carbonate as a recyclable solvent and optimizing the coupling of key intermediates, this technology offers a compelling value proposition for reliable pharmaceutical intermediates suppliers aiming to upgrade their production capabilities. The strategic implementation of this patent allows manufacturers to overcome the limitations of traditional mixed anhydride or acyl chloride methods, which often suffer from low yields and complex purification requirements.
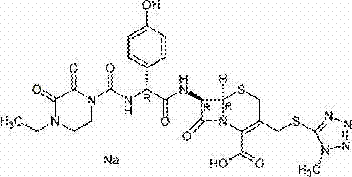
Cefoperazone Sodium, chemically known as (6R,7R)-3-[[(1-methyl-1H-tetrazole-5-yl)thio]methyl]-7-[(R)-2-(4-ethyl-2,3-dioxo-1-piperazinecarboxamido)-2-(4-hydroxyphenyl)acetamido]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid sodium salt, is a vital component in modern antimicrobial therapy. The structural integrity and purity of this molecule are paramount, especially for injectable formulations where clarity is a critical quality attribute. The innovation detailed in this patent provides a streamlined pathway that not only ensures high chemical purity but also significantly reduces the environmental footprint of the manufacturing process. For procurement and supply chain leaders, understanding the nuances of this synthetic route is essential for securing a stable, cost-effective supply of high-quality API intermediates in a competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefoperazone has relied on several established techniques, including the mixed anhydride method, acyl chloride method, thioester method, and amino substituting group substep condensation. While these methods have served the industry for decades, they are plagued by inherent inefficiencies that impact both cost and quality. For instance, literature indicates that traditional routes often achieve total recovery rates as low as 34.2% to 38.2%, representing a significant loss of valuable starting materials like 7-ACA. Furthermore, these conventional processes frequently involve harsh reaction conditions, multiple purification steps, and the use of hazardous solvents that complicate waste management and increase operational risks. A persistent issue in prior art is the difficulty in controlling product clarity, a vital index for injectable drugs, often leading to underproof products that fail stringent quality control standards. These limitations create bottlenecks in cost reduction in API manufacturing and pose challenges for maintaining consistent supply chain reliability.
The Novel Approach
In stark contrast, the method disclosed in CN102372729A revolutionizes the synthesis by introducing a green, high-yield protocol centered around the use of dimethyl carbonate. This innovative approach simplifies the operational complexity by utilizing mild reaction conditions that preserve the delicate beta-lactam structure of the cephalosporin nucleus. The process begins with the efficient condensation of 7-ACA and 1-methyl-5-mercapto tetrazole to form the key intermediate TZA (7-TZCA), followed by a highly selective coupling with the side chain acid chloride. By employing dimethyl carbonate, a solvent that is not only environmentally friendly but also recyclable, the method drastically cuts down on solvent consumption and disposal costs. The result is a process that delivers high yields at every step, with reported purities exceeding 99%, effectively solving the clarity issues associated with older technologies. This represents a significant leap forward in commercial scale-up of complex pharmaceutical intermediates, offering a robust alternative for industrial production.
Mechanistic Insights into Dimethyl Carbonate-Mediated Condensation
The core of this technological advancement lies in the precise activation and coupling mechanisms facilitated by the dimethyl carbonate system. In the first step, 7-ACA is suspended in a mixture containing dimethyl carbonate, glacial acetic acid, and a boron trifluoride dimethyl carbonate complex. This specific combination acts as a powerful yet controlled activating system, promoting the nucleophilic attack of 1-methyl-5-mercapto tetrazole on the C-3 position of the cephalosporin nucleus. The use of the boron complex is critical, as it enhances the electrophilicity of the reaction center without inducing the degradation often seen with stronger Lewis acids. Following this, the reaction mixture undergoes hydrolysis and crystallization, yielding the TZA intermediate with exceptional purity. This mechanistic precision ensures that the stereochemistry at the C-6 and C-7 positions is preserved, which is essential for the biological activity of the final antibiotic.
Subsequent steps involve the activation of the side chain, D-(-)-2-(4-ethyl-2,3-dioxo-1-piperazinecarboxamido)-2-(4-hydroxyphenyl)acetic acid, using phosphorus oxychloride in an organic solvent at low temperatures (-10°C to -40°C). This low-temperature activation minimizes racemization and side reactions, generating a highly reactive acyl chloride species in situ. When this activated side chain reacts with the TZA intermediate, the condensation proceeds efficiently to form Cefoperazone Acid. The final conversion to the sodium salt is achieved through a controlled pH adjustment in an acetone-water system, ensuring the formation of stable, high-quality crystals. This rigorous control over reaction parameters and intermediate stability is what allows the process to achieve such high levels of purity, directly addressing the reducing lead time for high-purity pharmaceutical intermediates by minimizing the need for extensive downstream purification.
How to Synthesize Cefoperazone Sodium Efficiently
The synthesis of Cefoperazone Sodium via this novel route is designed for operational simplicity and industrial scalability. The process is divided into three distinct stages: the formation of the tetrazole intermediate, the coupling with the side chain, and the final salt formation. Each stage is optimized to maximize yield and minimize impurity generation, making it an ideal candidate for technology transfer and large-scale manufacturing. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and crystallization protocols, are outlined below to guide R&D teams in replicating this high-efficiency process.
- Suspend 7-ACA in a mixture of dimethyl carbonate, glacial acetic acid, and boron trifluoride dimethyl carbonate complex, then react with 1-methyl-5-mercapto tetrazole to form the TZA intermediate.
- Activate the side chain acid using phosphorus oxychloride in organic solvent at low temperature, then condense with the TZA intermediate to form Cefoperazone Acid.
- Dissolve Cefoperazone Acid in acetone-water mixture, adjust pH to 7.0 with alkali solution, and crystallize to obtain high-purity Cefoperazone Sodium.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemical efficiency. The shift towards a greener, more streamlined process directly translates into enhanced supply chain resilience and significant cost optimization opportunities. By eliminating the need for hazardous solvents and reducing the number of purification cycles, manufacturers can lower their operational expenditures while simultaneously improving their environmental compliance profile. This alignment with green chemistry principles is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies, making this technology a key differentiator in the marketplace.
- Cost Reduction in Manufacturing: The utilization of dimethyl carbonate as a recyclable solvent is a primary driver for cost savings in this process. Unlike traditional solvents that require expensive disposal or complex recovery systems, dimethyl carbonate can be easily recovered and reused, substantially lowering raw material costs. Additionally, the high yields reported in each step of the synthesis mean that less starting material is wasted, further driving down the cost of goods sold. The elimination of transition metal catalysts or harsh reagents also reduces the need for expensive scavenging steps, streamlining the production workflow and contributing to overall economic efficiency without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 7-ACA and 1-methyl-5-mercapto tetrazole ensures a stable supply base, mitigating the risks associated with sourcing exotic or constrained reagents. The mild reaction conditions employed in this method reduce the likelihood of batch failures due to thermal runaway or equipment corrosion, thereby enhancing production predictability. Furthermore, the robustness of the crystallization steps ensures consistent product quality, reducing the incidence of out-of-specification batches that can disrupt supply schedules. This reliability is crucial for maintaining continuous production lines and meeting the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: From a scalability perspective, the process is inherently designed for industrial application, with parameters that are easily translated from laboratory to pilot and commercial scales. The use of green solvents aligns with increasingly stringent environmental regulations, reducing the regulatory burden and potential fines associated with volatile organic compound (VOC) emissions. The simplified waste stream, characterized by lower toxicity and easier treatment requirements, facilitates smoother permitting and operation in diverse geographic regions. This environmental compatibility not only future-proofs the manufacturing asset but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the global healthcare value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Cefoperazone Sodium using this patented method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical implementation and benefits of this technology for industry stakeholders.
Q: What are the primary advantages of using dimethyl carbonate in Cefoperazone synthesis?
A: Dimethyl carbonate serves as an environmentally friendly, recyclable solvent that replaces traditional hazardous solvents. It facilitates milder reaction conditions, simplifies operation, and significantly reduces production costs while maintaining high product purity.
Q: How does this novel method improve product clarity compared to conventional techniques?
A: Conventional methods often struggle with clarity due to impurities formed during harsh reaction conditions. This new method utilizes mild temperatures and specific crystallization protocols (e.g., controlled pH and seeding) that minimize impurity generation, resulting in superior clarity essential for injectable formulations.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial suitability. It features high yields at each step, uses readily available starting materials like 7-ACA, and employs recyclable solvents, making it economically viable and scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefoperazone Sodium Supplier
The technological potential of this green synthesis route underscores the importance of partnering with a manufacturer who possesses the expertise to execute complex chemical transformations with precision. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is evidenced by our stringent purity specifications and rigorous QC labs, which ensure that every batch of Cefoperazone Sodium meets the highest international standards for safety and efficacy. We understand the critical nature of antibiotic supply chains and are dedicated to providing a seamless, reliable source of high-quality intermediates and APIs.
We invite you to explore how this advanced manufacturing technology can optimize your supply chain and reduce costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your pharmaceutical manufacturing operations. Together, we can build a more sustainable and resilient future for global healthcare.
