Revolutionizing Beta-Lactam Intermediate Production: A Deep Dive into Efficient 4-Ethyl-2,3-Dioxopiperazine-1-Carboxylate Synthesis
The pharmaceutical industry's relentless pursuit of safer, more efficient synthetic routes for beta-lactam antibiotics has led to significant innovations in intermediate manufacturing, as exemplified by the technology disclosed in patent CN101921237A. This pivotal document outlines a robust preparation method for 4-ethyl-2,3-dioxopiperazine-1-carboxylate, a critical building block for renowned antibiotics such as Piperacillin and Cefoperazone. Traditionally, the synthesis of key intermediates in this class relied on hazardous acyl chloride derivatives, necessitating the use of toxic phosgene or its surrogates, which posed severe environmental and safety challenges. The novel approach described herein shifts the paradigm towards stable carboxylate esters, utilizing a direct esterification strategy that bypasses the need for dangerous activating agents. By leveraging 1-ethyl-2,3-dioxopiperazine and chloroformates in a controlled organic solvent system, this method achieves exceptional product purity exceeding 99% and yields as high as 96%. For R&D directors and procurement specialists alike, this represents a transformative opportunity to enhance supply chain resilience while adhering to stringent environmental, health, and safety (EHS) standards. The transition from unstable acyl chlorides to stable esters not only simplifies storage and handling but also streamlines the downstream coupling reactions required for final API assembly.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical manufacturing processes for piperacillin and cefoperazone intermediates were plagued by significant technical and economic inefficiencies, primarily centered around the instability and toxicity of 4-ethyl-2,3-dioxopiperazine-1-acyl chlorides. The conventional synthesis of these acyl chlorides invariably required the deployment of highly toxic substances such as phosgene, trichloromethyl chloroformate (diphosgene), or triphosgene, creating an operating environment that was inherently dangerous for personnel and difficult to manage from a regulatory compliance standpoint. Furthermore, these routes often necessitated the use of group activation agents like trimethylchlorosilane, which, while effective, added substantial material costs and generated stoichiometric amounts of silicate by-products that complicated waste treatment. The chemical instability of the resulting acyl chloride intermediate was another major bottleneck; being prone to facile hydrolysis and decomposition even at room temperature, it forced manufacturers to carry the crude methylene dichloride solution directly into the next reaction step without purification. This "telescoped" approach inevitably introduced impurities from the previous step into the final reaction system, compromising the quality of the active pharmaceutical ingredient and necessitating complex, energy-intensive purification protocols later in the pipeline. Additionally, the reliance on expensive and difficult-to-recover solvents like dioxane and anhydrous tetrahydrofuran further inflated the operational expenditure, making the overall process economically unsustainable for large-scale commercial production.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the innovative protocol detailed in CN101921237A introduces a streamlined, one-step esterification process that fundamentally alters the economic and safety profile of producing this vital intermediate. By reacting 1-ethyl-2,3-dioxopiperazine directly with methyl or ethyl chloroformate in the presence of an acid-binding agent, the process completely eliminates the need for toxic phosgene reagents and expensive silylating activators. The selection of acetone as the primary reaction solvent is a masterstroke of process chemistry, replacing the costly dioxane and THF mixture with a ubiquitous, low-cost, and easily recoverable commodity chemical. This substitution not only drastically reduces raw material costs but also simplifies the solvent recovery infrastructure, as acetone can be distilled and reused with high efficiency. The reaction conditions are meticulously optimized, operating within a mild temperature range of -40°C to 10°C, preferably between -10°C and -5°C, which effectively suppresses side reactions and ensures high selectivity. The result is a stable, isolable solid product—4-ethyl-2,3-dioxopiperazine-1-carboxylate—that can be purified via simple recrystallization to achieve purity levels greater than 99%, thereby providing a high-quality feedstock for subsequent antibiotic synthesis.
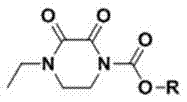
Mechanistic Insights into Base-Mediated Nucleophilic Substitution
The core chemical transformation driving this synthesis is a base-mediated nucleophilic acyl substitution, where the secondary amine nitrogen of the 1-ethyl-2,3-dioxopiperazine ring acts as a nucleophile attacking the electrophilic carbonyl carbon of the chloroformate. The presence of a suitable acid-binding agent, such as triethylamine, pyridine, or inorganic bases like sodium carbonate, is critical to scavenging the hydrogen chloride by-product generated during the reaction. Without effective acid scavenging, the accumulation of HCl would protonate the piperazine nitrogen, rendering it non-nucleophilic and stalling the reaction progress. The choice of triethylamine is particularly advantageous due to its solubility in organic solvents like acetone and its ability to form a solid hydrochloride salt that can be easily removed via filtration, thus driving the equilibrium towards product formation. The reaction kinetics are highly sensitive to temperature; maintaining the system below -5°C during the dropwise addition of the chloroformate prevents thermal runaway and minimizes the formation of di-substituted by-products or hydrolysis products. This precise thermal control ensures that the mono-substituted ester is formed exclusively, preserving the integrity of the dioxopiperazine ring structure which is essential for the biological activity of the final antibiotic.
Impurity control in this process is achieved through a combination of kinetic regulation and thermodynamic purification. The initial reaction mixture, after filtration of the amine salt, contains the crude ester along with trace amounts of unreacted starting material and potential hydrolysis products. The patent specifies a recrystallization step using mixed solvent systems, such as acetone and petroleum ether or ethyl acetate and petroleum ether, to refine the product. This step leverages the differential solubility of the target ester versus impurities; the desired 4-ethyl-2,3-dioxopiperazine-1-carboxylate crystallizes out as a high-purity white powder, while soluble impurities remain in the mother liquor. High-performance liquid chromatography (HPLC) analysis confirms that this purification strategy consistently yields products with purity exceeding 99%, meeting the rigorous specifications required for pharmaceutical intermediates. The structural integrity of the final molecule, characterized by the ethyl group at the 4-position and the carboxylate ester at the 1-position, is preserved throughout this gentle processing, ensuring it serves as a reliable precursor for the complex coupling reactions needed to construct the side chains of Piperacillin and Cefoperazone.
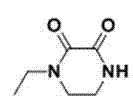
How to Synthesize 4-Ethyl-2,3-Dioxopiperazine-1-Carboxylate Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity and scalability, making it an ideal candidate for technology transfer from laboratory to pilot plant. The process begins with the dissolution of the starting piperazine derivative in a polar aprotic solvent, followed by the addition of a tertiary amine base to create a reactive nucleophilic environment. Detailed standard operating procedures regarding stoichiometry, addition rates, and crystallization parameters are essential for reproducibility. For a comprehensive guide on the exact molar ratios and specific workup techniques validated in multiple embodiments, please refer to the standardized protocol below.
- Dissolve 1-ethyl-2,3-dioxopiperazine in an organic solvent such as acetone and add an acid-binding agent like triethylamine.
- Cool the reaction mixture to a temperature between -40°C and 10°C, preferably -10°C to -5°C.
- Slowly dropwise add methyl chloroformate or ethyl chloroformate, maintain temperature, stir for 3-6 hours, filter, and recrystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers profound strategic advantages that extend far beyond simple chemical yield improvements. The elimination of hazardous reagents like phosgene and triphosgene removes a significant regulatory burden, reducing the costs associated with specialized handling, storage, and disposal of toxic materials. Furthermore, the replacement of expensive, specialized solvents with commodity chemicals like acetone creates a more resilient supply chain that is less susceptible to market volatility and sourcing disruptions. The simplified workup procedure, which involves straightforward filtration and distillation rather than complex extraction or chromatography, translates directly into reduced cycle times and lower utility consumption per kilogram of product. These factors collectively contribute to a substantial reduction in the cost of goods sold (COGS), allowing pharmaceutical manufacturers to improve their margins or pass savings on to healthcare providers. Additionally, the stability of the ester intermediate allows for batch production and inventory stocking, decoupling the synthesis of the intermediate from the immediate needs of the final API coupling step and providing greater flexibility in production scheduling.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of the equimolar trimethylchlorosilane activator, which was a significant cost driver in previous methods. By eliminating this reagent, the atom economy of the reaction is vastly improved, meaning less money is spent on materials that end up as waste. Moreover, the switch from a dual-solvent system of dioxane and THF to a single solvent system of acetone drastically lowers solvent procurement costs and simplifies the distillation train required for solvent recovery. The high yield of up to 96% ensures that raw material utilization is maximized, further driving down the unit cost of the intermediate. These cumulative efficiencies result in a manufacturing process that is not only cheaper to operate but also generates less waste, aligning with modern green chemistry principles and reducing environmental compliance costs.
- Enhanced Supply Chain Reliability: From a logistics perspective, the use of stable, non-hazardous reagents significantly de-risks the supply chain. Unlike phosgene, which requires specialized transport and storage infrastructure, chloroformates and acetone are widely available commodity chemicals with robust global supply networks. This accessibility ensures that production can be maintained continuously without the fear of supply interruptions due to regulatory crackdowns on toxic substances. The ability to isolate and store the intermediate as a stable solid powder also enhances supply chain flexibility; manufacturers can produce large batches during periods of low energy cost or high raw material availability and draw from inventory as needed. This decoupling of production steps reduces the pressure on just-in-time manufacturing schedules and provides a buffer against unexpected demand surges for the final antibiotics.
- Scalability and Environmental Compliance: The simplicity of the reaction setup—a standard stirred tank reactor with cooling capabilities—makes this process highly scalable from kilogram to multi-ton production scales without the need for exotic equipment. The reduction in "three wastes" (waste water, waste gas, and solid waste) is a critical advantage for facilities operating under strict environmental permits. By avoiding the generation of silicate by-products and chlorinated solvent waste streams, the facility's wastewater treatment load is significantly lightened. The high purity of the product (>99%) also means that downstream purification steps in the API synthesis can potentially be simplified, further reducing the overall environmental footprint of the drug manufacturing process. This alignment with sustainability goals makes the technology attractive for companies aiming to improve their ESG (Environmental, Social, and Governance) ratings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and comparative analysis provided in the patent documentation, offering clarity on yield optimization, solvent selection, and safety protocols. Understanding these nuances is essential for process engineers evaluating the feasibility of adopting this route for commercial production.
Q: Why is the ester intermediate preferred over the acyl chloride for Piperacillin synthesis?
A: The ester intermediate offers superior stability compared to the highly reactive and hydrolytically unstable acyl chloride. It eliminates the need for toxic phosgene or triphosgene, significantly improving operational safety and reducing hazardous waste generation.
Q: What are the critical parameters for achieving >99% purity in this synthesis?
A: Maintaining a low reaction temperature between -10°C and -5°C is crucial to suppress side reactions. Additionally, the use of acetone as a solvent and subsequent recrystallization from ethyl acetate/petroleum ether mixtures ensures the removal of impurities and unreacted starting materials.
Q: How does this method reduce production costs compared to traditional routes?
A: This method eliminates the expensive trimethylchlorosilane activator and replaces costly solvents like dioxane and anhydrous THF with affordable acetone. The simplified one-step process also reduces energy consumption and solvent recovery costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Ethyl-2,3-Dioxopiperazine-1-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the long-term viability of pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101921237A are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with advanced cryogenic reactors capable of maintaining the precise low-temperature conditions (-10°C to -5°C) required for this synthesis, guaranteeing consistent product quality. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch of 4-ethyl-2,3-dioxopiperazine-1-carboxylate meets the >99% purity threshold necessary for beta-lactam antibiotic synthesis. Our commitment to excellence extends beyond mere compliance; we actively optimize every step of the process to maximize yield and minimize environmental impact, delivering a sustainable supply solution for our global partners.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their Piperacillin and Cefoperazone supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this ester-based route for your unique production volume. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss detailed route feasibility assessments tailored to your project timelines. Let us help you secure a reliable, cost-effective, and compliant supply of this critical intermediate, empowering your organization to lead in the competitive landscape of generic and branded antibiotic manufacturing.
