Scalable Synthesis of P-Mentha-Dienol for Commercial Cannabis API Production
Scalable Synthesis of P-Mentha-Dienol for Commercial Cannabis API Production
The pharmaceutical landscape surrounding cannabinoid therapeutics has evolved rapidly, driving an urgent demand for robust, scalable synthetic routes to key intermediates like (+)-p-mentha-2,8-diene-1-ol. Patent CN1777569A introduces a groundbreaking methodology that addresses the historical bottlenecks in producing this critical precursor for Delta-9-THC synthesis. By leveraging Lewis acid catalysis to control the regioselectivity and stereoselectivity of epoxide ring opening, this technology offers a distinct advantage over traditional enzymatic or singlet oxygen oxidation pathways. For R&D directors and procurement specialists alike, understanding this mechanism is vital for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent purity specifications without the prohibitive costs associated with chiral resolution. The ability to achieve a major-to-minor product ratio greater than 20:1 represents a paradigm shift in process efficiency, directly translating to reduced waste and lower operational expenditures in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of limonene to (+)-p-mentha-2,8-diene-1-ol has been plagued by significant technical hurdles that hinder industrial viability. Conventional approaches often rely on the oxidation of limonene with singlet oxygen or complex enzymatic transformations, both of which introduce substantial variability and cost into the supply chain. These legacy methods frequently result in the formation of extensive isomeric mixtures, necessitating laborious and expensive chiral resolution steps to isolate the desired enantiomer. Furthermore, the handling of singlet oxygen requires specialized equipment and safety protocols that complicate scale-up efforts, while enzymatic routes can suffer from inconsistent batch-to-batch performance and limited substrate tolerance. For a procurement manager evaluating cost reduction in pharmaceutical manufacturing, these factors create a fragile supply chain vulnerable to delays and quality deviations, ultimately inflating the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a Lewis acid-mediated ring opening strategy that fundamentally simplifies the synthetic architecture. By reacting (+)-limonene oxide with specific amines in the presence of catalysts such as lithium acetate or lithium bromide, the process achieves exceptional stereocontrol without the need for exotic reagents. This method operates effectively at moderate temperatures ranging from 30°C to 80°C, significantly lowering energy consumption compared to high-thermal processes. The reaction selectively targets the less substituted carbon atom of the epoxide ring, driven by the electronic modulation provided by the Lewis acid, ensuring that the desired (1S, 2S, 4R) configuration is formed predominantly. This streamlined pathway not only eliminates the need for complex chiral separations but also allows for the recovery of unreacted starting materials, thereby enhancing the overall atom economy and sustainability profile of the manufacturing process.
Mechanistic Insights into Lewis Acid-Catalyzed Epoxide Opening
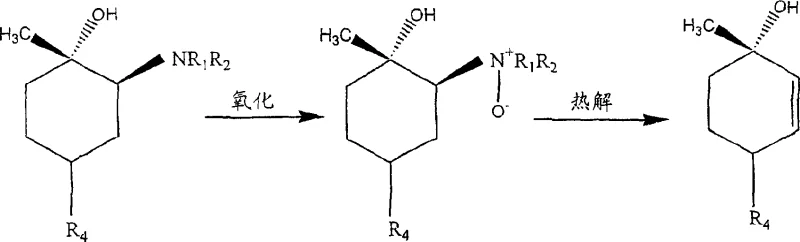
The core innovation of this technology lies in the precise manipulation of electronic density within the epoxide ring through Lewis acid coordination. When a Lewis acid, such as a lithium salt, complexes with the oxygen atom of the epoxide, it withdraws electron density, thereby altering the partial positive charges on the adjacent carbon atoms. This electronic redistribution makes the less substituted carbon atom significantly more susceptible to nucleophilic attack by the amine, overriding the steric hindrance that typically favors attack at the more substituted position in uncatalyzed reactions. Consequently, the reaction proceeds with high regioselectivity, yielding the desired amine adduct intermediate with a ratio exceeding 20:1 relative to unwanted isomers. This level of control is critical for maintaining high purity standards, as it minimizes the formation of difficult-to-remove impurities early in the synthesis, reducing the burden on downstream purification units.
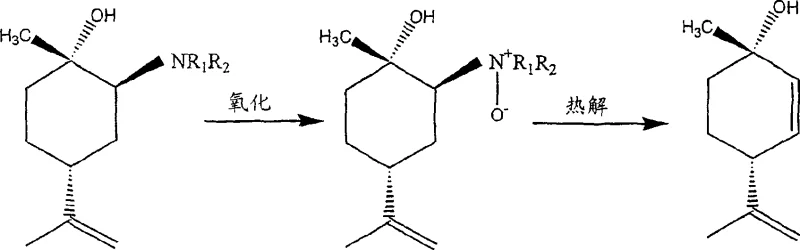
Following the formation of the amine adduct, the pathway diverges into two viable options for generating the final alkene functionality, both of which offer distinct mechanistic advantages. The primary route involves the oxidation of the tertiary amine to an N-oxide using hydrogen peroxide, followed by a thermal Cope elimination. This elimination proceeds via a concerted cyclic transition state that ensures the formation of the double bond with specific stereochemistry, avoiding the carbocation rearrangements common in acid-catalyzed dehydrations. Alternatively, the amine adduct can be quaternized with an alkyl halide and subsequently subjected to base hydrolysis, providing flexibility in process design depending on available infrastructure. Both pathways effectively preserve the chiral integrity established in the initial ring-opening step, ensuring that the final (+)-p-mentha-2,8-diene-1-ol product meets the rigorous optical purity requirements necessary for subsequent conversion into bioactive cannabinoids.
How to Synthesize (+)-p-mentha-2,8-diene-1-ol Efficiently
Implementing this synthesis requires careful attention to reaction conditions, particularly the choice of Lewis acid and the control of temperature during the adduct formation phase. The protocol begins with dissolving (+)-limonene oxide in a suitable alcohol solvent, followed by the addition of the Lewis acid catalyst and the amine nucleophile. Maintaining the reaction temperature around 50°C is crucial for optimizing the selectivity ratio while preventing side reactions that could degrade the epoxide ring. Once the amine adduct is isolated, typically as a hydrochloride salt for enhanced purity, it undergoes oxidation and subsequent elimination to yield the target diene alcohol. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps outlined below.
- React (+)-limonene oxide with a secondary amine in the presence of a Lewis acid catalyst like lithium acetate at 50°C to form the amine adduct with >20: 1 selectivity.
- Oxidize the resulting amine adduct using hydrogen peroxide in an alcohol solvent to generate the corresponding N-oxide intermediate.
- Perform pyrolysis of the N-oxide in toluene at temperatures above 110°C to effect Cope elimination and yield the final diene alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers transformative benefits that directly address the pain points of sourcing high-value pharmaceutical intermediates. By replacing complex enzymatic or photo-oxidative steps with robust chemical catalysis, manufacturers can achieve a drastic simplification of the production workflow. This simplification translates into tangible supply chain reliability, as the reliance on sensitive biological catalysts or hazardous singlet oxygen generators is completely eliminated. For supply chain heads focused on continuity, the ability to source standard chemical reagents like lithium salts and common amines ensures that production is not bottlenecked by specialized material shortages. Furthermore, the high selectivity of the reaction reduces the volume of waste solvents and byproducts, aligning with increasingly strict environmental compliance regulations and reducing disposal costs.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps represents a massive opportunity for cost optimization in the production of cannabinoid precursors. Traditional methods often require multiple crystallization or chromatographic steps to separate enantiomers, which consume significant amounts of solvent and time. By achieving high stereoselectivity directly in the bond-forming step, this process removes these expensive purification stages entirely. Additionally, the recovery of unreacted trans-limonene oxide allows for material recycling, further driving down the raw material cost per kilogram of finished product. These efficiencies compound to offer a substantially lower cost of goods, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the chemical reagents used in this pathway ensures a stable and predictable supply timeline. Unlike enzymatic processes that may vary in activity based on storage conditions or batch origin, Lewis acid catalysts are stable, shelf-stable inorganic or organometallic salts with consistent performance profiles. This consistency allows for precise production planning and reduces the risk of batch failures that can disrupt downstream drug formulation schedules. For procurement managers, this means negotiating contracts with greater confidence, knowing that the supplier can maintain consistent output volumes without the volatility associated with biocatalytic systems.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is facilitated by the use of standard unit operations such as stirred tank reactors and distillation columns. The reaction conditions, operating at moderate temperatures and atmospheric pressure, do not require exotic high-pressure vessels or cryogenic cooling systems, lowering the capital expenditure required for plant retrofitting. Moreover, the reduction in solvent usage and the avoidance of heavy metal catalysts simplify wastewater treatment protocols. This environmental friendliness not only reduces regulatory burdens but also enhances the corporate sustainability profile, a key metric for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your existing supply chain. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of using Lewis acids in this synthesis?
A: The use of Lewis acids alters the partial charges on the epoxide ring carbons, enabling highly regioselective and stereoselective nucleophilic attack by the amine, which drastically reduces isomeric impurities compared to conventional thermal methods.
Q: Can unreacted trans-limonene oxide be recovered?
A: Yes, the process allows for the recovery of unreacted (+)-trans-limonene oxide, which remains inert under the specific reaction conditions, providing an effective method for diastereomer separation and improving overall atom economy.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Absolutely, the method operates at moderate temperatures (30°C to 80°C) and avoids expensive enzymatic steps or singlet oxygen generation, making it highly amenable to commercial scale-up with simplified purification protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (+)-p-mentha-2,8-diene-1-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation cannabinoid therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (+)-p-mentha-2,8-diene-1-ol meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence means we can adapt the Lewis acid catalyzed route to fit your specific capacity requirements while maintaining the highest levels of quality assurance.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your project economics. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential reductions in manufacturing expenses specific to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to move forward with confidence in your supply chain strategy.
