Revolutionizing P-Methylsulfonylbenzaldehyde Production with Green Solvent-Free Technology
Revolutionizing P-Methylsulfonylbenzaldehyde Production with Green Solvent-Free Technology
In the highly regulated landscape of pharmaceutical manufacturing, the reliability and purity of key intermediates dictate the success of final drug products. P-Methylsulfonylbenzaldehyde serves as a critical starting material for the synthesis of Thiamphenicol, a broad-spectrum antibiotic, as well as various beta-lactam antibacterial agents and antiangina drugs. The traditional supply chain for this essential chemical has often been plagued by complex multi-step processes and significant environmental burdens. However, a groundbreaking technical disclosure found in patent CN103058899A introduces a transformative approach that redefines the production standards for this vital pharmaceutical intermediate. This innovative methodology not only streamlines the synthetic route but also aligns perfectly with modern green chemistry principles by eliminating the need for organic solvents entirely. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, understanding the technical nuances of this solvent-free pathway is crucial for securing a sustainable and cost-effective supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of P-Methylsulfonylbenzaldehyde has relied on routes that are inherently inefficient and environmentally taxing. Conventional methods often involve lengthy reaction sequences that require multiple purification steps, leading to cumulative yield losses that can severely impact overall production economics. Furthermore, many traditional processes utilize volatile organic compounds (VOCs) as solvents, necessitating expensive recovery systems and generating hazardous waste streams that require rigorous treatment before disposal. The use of harsh oxidizing agents in older methodologies can also lead to unpredictable side reactions, resulting in complex impurity profiles that complicate downstream processing and final drug safety assessments. These factors collectively contribute to higher operational expenditures and increased regulatory scrutiny, making it difficult for manufacturers to maintain consistent quality and competitive pricing in a global market that demands both efficiency and sustainability.
The Novel Approach
The novel synthetic strategy outlined in the referenced patent offers a robust solution to these longstanding industry challenges by employing a direct two-step conversion that maximizes atom economy. By utilizing p-methylsulfonyltoluene as the starting material, the process bypasses unnecessary functional group transformations, significantly shortening the overall production timeline. The elimination of organic solvents is perhaps the most striking feature, as it removes the need for solvent recovery infrastructure and virtually eliminates VOC emissions from the production facility. This streamlined approach not only simplifies the operational workflow but also enhances the safety profile of the manufacturing plant by reducing fire hazards associated with flammable solvents. For supply chain leaders, this translates into a more resilient production capability that is less susceptible to fluctuations in solvent availability and pricing, ensuring a steady flow of high-quality intermediates for antibiotic manufacturing.
Mechanistic Insights into Catalytic Bromination and Aqueous Hydrolysis
The core of this advanced synthesis lies in a highly controlled free-radical substitution reaction followed by a selective hydrolysis step. In the first stage, p-methylsulfonyltoluene undergoes bromination in the presence of a catalyst such as copper chloride, iron salts, or iodine at temperatures ranging from 100°C to 200°C. This thermal activation facilitates the substitution of hydrogen atoms on the methyl group with bromine atoms, forming the key intermediate, p-methylsulfonyldibromotoluene. The reaction conditions are meticulously optimized to prevent over-bromination or ring substitution, ensuring that the methyl group is selectively targeted. The molar ratio of toluene to bromine is carefully maintained between 1:1.2 and 1:8.5 to drive the reaction to completion while minimizing the formation of poly-brominated byproducts. This precise control over the reaction kinetics is essential for achieving the high purity levels required for pharmaceutical applications.
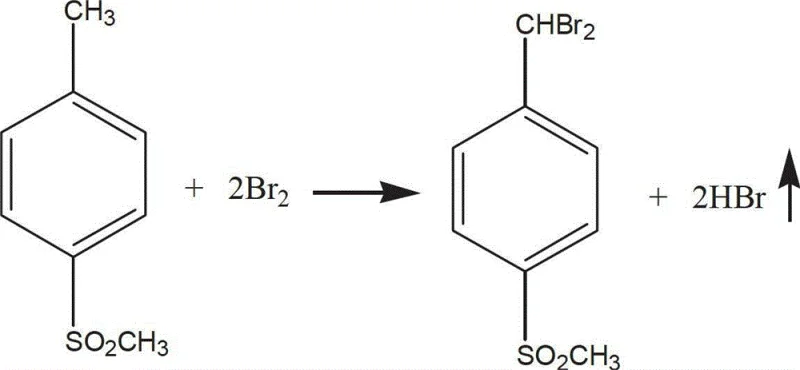
Following the bromination, the crude liquid product undergoes a rigorous purification protocol involving vacuum distillation to remove hydrogen bromide and unreacted bromine, followed by neutralization and washing. The second critical phase involves the hydrolysis of the purified p-methylsulfonyldibromotoluene in an aqueous medium. Conducted at temperatures between 80°C and 100°C, this hydrolysis converts the gem-dibromo group into an aldehyde functionality. The mechanism likely proceeds through the formation of a gem-diol intermediate which spontaneously dehydrates to yield the stable aldehyde product. A remarkable feature of this step is the recyclability of the hydrolysis liquor, which can be reused in subsequent batches, thereby creating a closed-loop water system that drastically reduces fresh water consumption. This aqueous-based transformation stands in stark contrast to traditional methods that might require anhydrous conditions or toxic reagents, highlighting the green chemistry credentials of this process.
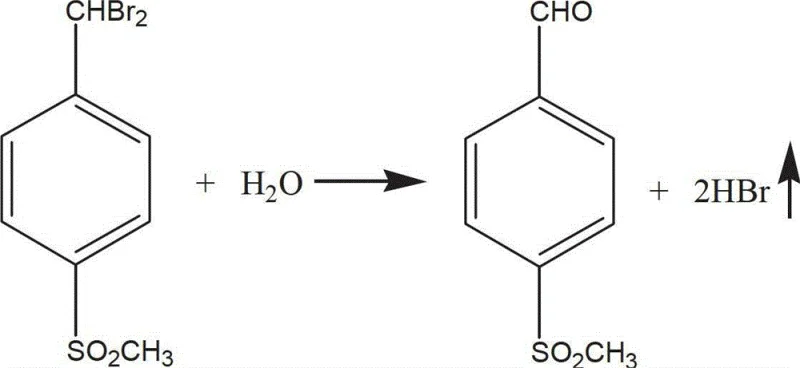
Impurity control is intrinsically built into this mechanistic design through the physical properties of the intermediates and products. The crystallization of the dibromo intermediate from water allows for the effective rejection of soluble impurities, while the final filtration of the aldehyde product ensures the removal of any remaining inorganic salts or catalyst residues. The patent data indicates that this dual-purification strategy consistently delivers product purity exceeding 97%, with total yields surpassing 92%. Such high performance metrics are indicative of a reaction pathway that minimizes side reactions and maximizes the conversion of starting materials into the desired target molecule. For R&D directors evaluating potential suppliers, this level of mechanistic robustness provides confidence in the consistency and scalability of the manufacturing process, ensuring that every batch meets stringent pharmacopeial standards.
How to Synthesize P-Methylsulfonylbenzaldehyde Efficiently
Implementing this synthesis route requires precise adherence to the thermal and stoichiometric parameters defined in the technical literature to ensure optimal results. The process begins with the charging of p-methylsulfonyltoluene into a reactor, followed by the addition of the chosen catalyst and the controlled introduction of bromine under heating. Once the bromination is complete, the removal of acidic byproducts is critical before proceeding to the hydrolysis stage to prevent equipment corrosion and ensure product stability. The subsequent hydrolysis step leverages the unique reactivity of the dibromo intermediate in hot water, transforming it into the final aldehyde without the need for additional organic reagents. Detailed standard operating procedures regarding temperature ramping, vacuum levels, and filtration techniques are essential for replicating the high yields reported in the patent documentation.
- React p-methylsulfonyltoluene with bromine at 100°C-200°C using a copper or iron catalyst to form p-methylsulfonyldibromotoluene.
- Remove HBr and excess bromine via vacuum distillation, then neutralize and wash the crude product to obtain purified p-methylsulfonyldibromotoluene.
- Hydrolyze the purified intermediate in water at 80°C-100°C, then cool, filter, and dry to obtain the final P-Methylsulfonylbenzaldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this solvent-free technology offers profound benefits that extend far beyond simple chemical efficiency. For procurement managers focused on cost reduction in pharmaceutical intermediate manufacturing, the elimination of organic solvents represents a substantial decrease in raw material expenses and waste disposal fees. The absence of solvent recovery units lowers capital expenditure requirements for new production lines and reduces the energy consumption associated with distillation processes. Furthermore, the ability to recycle the hydrolysis water creates a circular economy within the plant, significantly lowering utility costs and minimizing the environmental footprint of the operation. These factors combine to create a highly competitive cost structure that allows suppliers to offer better pricing without compromising on quality or margin.
- Cost Reduction in Manufacturing: The removal of organic solvents from the process flow eliminates the need for complex solvent recovery systems and reduces the volume of hazardous waste requiring specialized treatment. This simplification of the infrastructure leads to lower operational overheads and reduced maintenance costs for production equipment. Additionally, the high yield of over 92% means that less raw material is wasted, directly improving the material cost per kilogram of the final product. The use of inexpensive and readily available catalysts like iron or copper salts further contributes to the economic viability of the process compared to routes requiring precious metal catalysts.
- Enhanced Supply Chain Reliability: By relying on water as the primary reaction medium for the second step, the process reduces dependency on volatile organic solvent markets which can be subject to supply disruptions and price spikes. The robustness of the reaction conditions, which tolerate a range of catalysts and temperatures, ensures that production can continue even if specific reagent grades are temporarily unavailable. This flexibility enhances the resilience of the supply chain, guaranteeing consistent delivery schedules for downstream pharmaceutical manufacturers who rely on just-in-time inventory models. The high purity of the product also reduces the risk of batch rejections, ensuring a smooth flow of materials through the value chain.
- Scalability and Environmental Compliance: The solvent-free nature of this synthesis makes it inherently safer and easier to scale from pilot plants to full commercial production without the exponential increase in safety risks associated with large volumes of flammable solvents. The process generates minimal waste, primarily consisting of recyclable water and manageable salt byproducts, which simplifies compliance with increasingly strict environmental regulations. This 'zero emission' characteristic positions the manufacturer as a preferred partner for global pharmaceutical companies that have committed to aggressive sustainability goals. The ease of scale-up ensures that supply can be rapidly expanded to meet surging market demand for antibiotics and other derivative drugs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of P-Methylsulfonylbenzaldehyde using this advanced methodology. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply networks. The answers are derived directly from the technical specifications and experimental data provided in the underlying patent documentation, ensuring accuracy and relevance for industry professionals.
Q: What are the key advantages of the solvent-free synthesis method for P-Methylsulfonylbenzaldehyde?
A: The primary advantage is the elimination of organic solvents, which drastically reduces environmental pollution and waste treatment costs. Additionally, the process achieves a total yield exceeding 92% with product purity above 97%, ensuring high efficiency for downstream antibiotic production.
Q: Can the hydrolysis wastewater be recycled in this manufacturing process?
A: Yes, the patent explicitly describes a closed-loop system where the hydrolysis liquid is recycled for subsequent batches. This significantly reduces water consumption and minimizes the discharge of acidic wastewater containing hydrogen bromide.
Q: What catalysts are suitable for the bromination step in this synthesis route?
A: The process allows for flexibility in catalyst selection, including copper, copper salts, iodine, iron, or iron salts. This versatility allows manufacturers to optimize costs based on raw material availability while maintaining reaction control between 100°C and 200°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable P-Methylsulfonylbenzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final pharmaceutical product depends heavily on the quality and consistency of your starting materials. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of market fluctuations. Our commitment to stringent purity specifications and the operation of rigorous QC labs guarantees that every batch of P-Methylsulfonylbenzaldehyde we deliver meets the highest international standards. We leverage advanced green chemistry technologies, such as the solvent-free synthesis route discussed, to provide our partners with a sustainable and economically advantageous supply solution.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall procurement costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your development timeline. Partnering with us ensures not only a reliable source of high-purity intermediates but also a collaborative relationship focused on innovation and long-term supply chain stability.
