Advanced Synthesis of Biapenem Ester: Overcoming Stability Challenges with Dimer Intermediates
The pharmaceutical landscape for carbapenem antibiotics demands rigorous control over intermediate stability and process scalability. Patent CN101891756A introduces a transformative synthesis method for biapenem ester, a critical precursor in the manufacturing of broad-spectrum antibiotics. This technology addresses a longstanding bottleneck in the industry: the instability of the traditional side-chain precursor. By shifting from a hygroscopic monomeric thiol to a stable dimeric structure, this process significantly enhances the robustness of the supply chain for high-purity pharmaceutical intermediates. The innovation lies not merely in a new reagent but in a fundamental rethinking of how complex beta-lactam structures are assembled, offering a pathway that reduces operational complexity while maintaining stringent purity standards required for parenteral medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
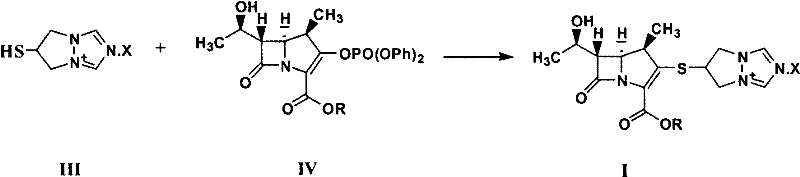
Historically, the synthesis of biapenem ester has relied heavily on the direct coupling of a carbapenem core with a specific thiol-containing side chain, often designated as Compound III in technical literature. This conventional route presents severe logistical and chemical challenges for large-scale manufacturing. Compound III is characterized by extreme hygroscopicity, meaning it readily absorbs moisture from the atmosphere, which complicates weighing, storage, and reaction stoichiometry. This sensitivity often leads to batch-to-batch variability and necessitates specialized storage conditions, driving up inventory costs. Furthermore, the preparation of this monomeric side chain is inherently difficult, often requiring multi-step sequences that yield unstable products prone to degradation. These factors collectively create a fragile supply chain where raw material quality is a constant variable, posing risks to the final API purity and overall production timelines.
The Novel Approach
The methodology disclosed in CN101891756A circumvents these instability issues by utilizing a dimeric form of the side chain, referred to as Compound II. This dimer serves as a stable, non-hygroscopic surrogate that effectively protects the reactive thiol functionality until the moment of coupling. In this novel approach, the dimer reacts with the carbapenem phosphate intermediate (Compound IV) in the presence of a phosphine catalyst and a base. This strategy eliminates the need to handle the troublesome monomeric thiol entirely. The reaction proceeds smoothly under mild conditions, typically between -40°C and 50°C, and allows for direct crystallization of the product without intermediate purification steps. This represents a significant leap in process chemistry, transforming a delicate, moisture-sensitive operation into a robust, scalable industrial process suitable for reliable pharmaceutical intermediate supplier operations.
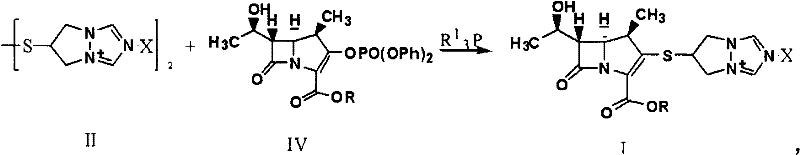
Mechanistic Insights into Phosphine-Mediated Dimer Coupling
The core of this synthetic advancement relies on the unique reactivity of trivalent phosphorus compounds, such as tributylphosphine, in mediating the cleavage of the disulfide or dimeric bond in Compound II. Mechanistically, the phosphine acts as a nucleophile that attacks the sulfur-sulfur linkage or facilitates the generation of the active thiolate species in situ. This activation occurs concurrently with the displacement of the phosphate leaving group on the carbapenem core (Compound IV). The presence of a base, such as diisopropylethylamine or lutidine, is critical to neutralize the acidic byproducts and maintain the nucleophilicity of the sulfur species. This concerted mechanism ensures that the reactive thiol is generated only in the immediate vicinity of the electrophilic carbapenem core, minimizing side reactions such as disulfide reformation or hydrolysis of the sensitive beta-lactam ring. The result is a highly efficient coupling that preserves the stereochemical integrity of the 1-beta-methyl group, which is essential for the biological activity of the final antibiotic.
Impurity control in this process is inherently superior due to the stability of the starting materials and the simplicity of the workup. In conventional routes, the degradation of the hygroscopic side chain often introduces sulfur-containing impurities that are difficult to separate from the product. By using the stable dimer, the impurity profile is significantly cleaner from the outset. Furthermore, the protocol specifies an acidification step post-reaction, adjusting the pH to a range of 3.0 to 6.5. This precise pH control is vital for protonating any remaining basic impurities and facilitating the precipitation of the target ester. The subsequent crystallization step, induced by cooling and the addition of anti-solvents like methylene dichloride, acts as a powerful purification engine. This ability to achieve high purity through crystallization rather than chromatography is a hallmark of a mature, cost-effective manufacturing process, ensuring that the final biapenem ester meets the rigorous specifications demanded by global regulatory bodies.
How to Synthesize Biapenem Ester Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a GMP environment. The process begins with the careful selection of solvents, with acetonitrile and tetrahydrofuran identified as optimal media for balancing solubility and reaction rate. Operators must ensure strict air isolation throughout the reaction phase to prevent oxidation of the phosphine catalyst and the sulfur intermediates. Following the coupling reaction, which typically proceeds for 15 minutes to several hours depending on temperature, the mixture is quenched with acid. The subsequent crystallization is driven by thermodynamic control, where cooling the solution to temperatures as low as -30°C maximizes yield. For a detailed breakdown of the specific molar ratios, solvent volumes, and agitation speeds required for scale-up, please refer to the standardized synthesis guide below.
- Under air isolation, combine organic solvent, stable dimer compound (II), carbapenem core (IV), tributylphosphine, and a base in a reactor, maintaining temperature between -40°C and 50°C for over 15 minutes.
- Monitor reaction progress via HPLC; upon completion, adjust the system to acidity (pH 3.0–6.5) using mineral or organic acid.
- Cool the reaction mixture, add a crystallization solvent such as methylene dichloride, stir to induce crystallization, and filter to obtain the target biapenem ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this dimer-based synthesis offers tangible strategic benefits beyond simple chemical elegance. The primary advantage lies in the drastic simplification of raw material logistics. By replacing a difficult-to-store, hygroscopic reagent with a stable solid dimer, companies can reduce warehousing costs and minimize the risk of raw material spoilage. This stability translates directly into supply chain resilience, ensuring that production schedules are not disrupted by the degradation of key starting materials. Additionally, the elimination of complex purification steps reduces the consumption of silica gel and solvents, contributing to a leaner, more environmentally compliant manufacturing footprint. These factors combine to create a more predictable and cost-efficient supply model for this critical antibiotic intermediate.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of unit operations. Traditional methods often require extensive purification to remove impurities derived from the unstable side chain. By enabling direct crystallization, this method significantly lowers operational expenditures related to labor, energy, and consumables. The avoidance of expensive chromatography columns and the reduction in solvent usage for purification further drive down the cost of goods sold. Moreover, the higher stability of the dimer intermediate reduces waste associated with expired or degraded raw materials, optimizing the overall material balance of the production line.
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical sector. The hygroscopic nature of the conventional side chain (Compound III) makes it susceptible to environmental fluctuations during transport and storage, creating a single point of failure in the supply chain. The stable dimer (Compound II) mitigates this risk entirely, offering a shelf-stable alternative that can be sourced and stocked with confidence. This robustness allows for larger batch sizes and longer production runs without the fear of raw material variability, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges. This synthesis route is inherently safer due to the reduced handling of reactive, moisture-sensitive thiols. The ability to operate at near-ambient temperatures (-30°C to 30°C) reduces the energy load compared to cryogenic processes. Furthermore, the simplified workup generates less hazardous waste, aligning with modern green chemistry principles. This ease of scale-up means that transitioning from pilot plant quantities to multi-ton commercial production can be achieved with minimal process re-engineering, accelerating time-to-market for generic versions of biapenem.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN101891756A, providing a factual basis for evaluating the process feasibility. Understanding these nuances is essential for R&D teams planning technology transfer and procurement teams assessing vendor capabilities. The focus remains on the practical implications of using the dimer intermediate versus the traditional monomer.
Q: Why is the dimer intermediate (Compound II) preferred over the monomer (Compound III) in biapenem synthesis?
A: Compound III is highly hygroscopic and difficult to store, leading to quality variability. The dimer (Compound II) is chemically stable, simplifying storage and handling while ensuring consistent reaction outcomes.
Q: Does this synthesis method require complex purification steps?
A: No, the process is designed for efficiency. The target compound can be obtained directly through crystallization after acidification, eliminating the need for column chromatography or complex purification.
Q: What are the critical reaction conditions for this coupling?
A: The reaction requires strict air isolation and the presence of a phosphine catalyst (e.g., tributylphosphine) and a base. Temperature control between -30°C and 30°C is preferred for optimal results.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biapenem Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capacity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this dimer-based synthesis are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze complex carbapenem impurity profiles. Our commitment to quality assurance means that every batch of biapenem ester we produce adheres to the highest international standards, providing our clients with the confidence needed to navigate regulatory filings and market entry.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis method can optimize your supply chain. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance metrics and our dedication to delivering high-value pharmaceutical solutions.
