Advanced Base-Catalyzed Synthesis of Spiropyrazoline Pyrrolidone Derivatives for Commercial Scale-Up
The development of efficient synthetic routes for spiroheterocyclic compounds remains a critical frontier in modern organic chemistry, particularly for the creation of bioactive molecules used in agrochemical and pharmaceutical applications. Patent CN113717179A discloses a groundbreaking methodology for the preparation of spiropyrazoline pyrrolidone derivatives, addressing the longstanding challenge of constructing five-membered nitrogen-containing rings with high precision. This innovation leverages a base-mediated cycloaddition strategy that bypasses the limitations of traditional organocatalytic methods, offering a streamlined pathway to structurally diverse spiro scaffolds. The significance of this technology lies in its ability to generate complex molecular architectures found in potent antibacterial, anti-inflammatory, and anti-tumor agents through a operationally simple process. By utilizing readily available substituted 3-arylmethylenemaleimides and chlorinated hydrazones, this approach not only enhances reaction efficiency but also ensures high regioselectivity, making it an invaluable asset for R&D teams focused on novel drug discovery and pesticide development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiropyrazoline derivatives has relied heavily on organocatalytic cycloaddition reactions, which, while effective in certain contexts, often suffer from significant drawbacks when applied to the construction of specific five-membered nitrogen-containing heterocycles. Traditional methods frequently require expensive and sensitive catalysts that demand rigorous exclusion of moisture and oxygen, thereby increasing the complexity and cost of the manufacturing process. Furthermore, many existing protocols struggle with poor regioselectivity, leading to the formation of unwanted isomeric byproducts that complicate downstream purification and reduce overall yield. The reliance on transition metals or specialized organocatalysts also introduces potential contamination issues, necessitating additional purification steps to meet the stringent purity specifications required for pharmaceutical and agrochemical intermediates. These inefficiencies create bottlenecks in the supply chain, extending lead times and inflating the cost of goods sold for high-value spiro compounds.
The Novel Approach
In stark contrast, the methodology outlined in the patent data introduces a robust, base-promoted strategy that fundamentally simplifies the synthetic landscape for spiropyrazoline pyrrolidones. By reacting substituted 3-arylmethylenemaleimide compounds with substituted chlorinated hydrazone compounds in the presence of a suitable base, this novel approach achieves rapid cyclization under mild conditions ranging from 0°C to 100°C. The elimination of complex catalytic systems allows for the use of inexpensive inorganic bases such as cesium carbonate or potassium carbonate, drastically reducing raw material costs. This method exhibits exceptional substrate tolerance, accommodating a vast array of electronic and steric environments on the aromatic rings without sacrificing yield or selectivity. The result is a highly efficient process that delivers target products with high purity, minimizing the need for extensive chromatographic separation and enabling a more sustainable and cost-effective manufacturing workflow for industrial applications.
Mechanistic Insights into Base-Mediated [3+2] Cycloaddition
The core of this synthetic breakthrough lies in the generation of a reactive nitrile imine dipole through the base-mediated dehydrohalogenation of the substituted chlorinated hydrazone. Upon treatment with a base, the chlorinated hydrazone undergoes elimination of hydrogen chloride to form the 1,3-dipolar nitrile imine species in situ. This highly reactive intermediate then engages in a concerted [3+2] cycloaddition with the electron-deficient double bond of the substituted 3-arylmethylenemaleimide, which acts as the dipolarophile. The reaction proceeds through a transition state that favors the formation of the spiropyrazoline ring system with excellent regiocontrol, driven by the electronic complementarity between the dipole and the dipolarophile. This mechanistic pathway avoids the formation of radical intermediates often seen in metal-catalyzed processes, thereby reducing the risk of side reactions and polymerization that can plague less controlled systems.
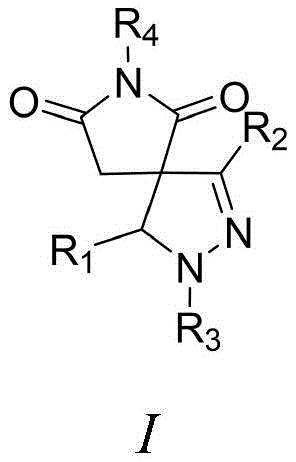
Furthermore, the choice of base and solvent plays a pivotal role in modulating the reaction kinetics and ensuring high conversion rates. The patent data indicates that a wide variety of solvents, including dichloromethane, acetonitrile, and toluene, are compatible with this transformation, providing flexibility for process optimization. The use of mild bases prevents the degradation of sensitive functional groups on the substrate, such as esters or nitro groups, which might otherwise be compromised under harsher acidic or strongly nucleophilic conditions. This chemoselectivity is crucial for maintaining the integrity of complex molecular structures intended for biological testing. Additionally, the straightforward workup procedure involving aqueous quenching and organic extraction effectively removes inorganic salts and unreacted starting materials, resulting in a crude product that is amenable to simple purification techniques, thus enhancing the overall practicality of the method for large-scale operations.
How to Synthesize Spiropyrazoline Pyrrolidone Derivatives Efficiently
To implement this high-efficiency synthesis in a laboratory or pilot plant setting, operators should adhere to the optimized molar ratios and reaction conditions specified in the patent to ensure maximum yield and reproducibility. The process involves combining the maleimide and hydrazone precursors with a stoichiometric amount of base in a suitable organic solvent, followed by stirring at ambient or slightly elevated temperatures. Detailed standard operating procedures regarding specific solvent volumes, addition rates, and safety precautions for handling chlorinated hydrazones are critical for successful execution. For a comprehensive guide on the standardized synthesis steps, please refer to the technical protocol section below.
- Combine substituted 3-arylmethylenemaleimide, substituted chlorinated hydrazone, and a suitable base (e.g., Cs2CO3) in an organic solvent like DCM at a molar ratio of 1.0: 1.5:1.5.
- Stir the reaction mixture at a controlled temperature between 0°C and 100°C until the reaction is complete, monitoring progress via TLC or HPLC.
- Quench with water or saturated saline, extract with organic solvent, dry, concentrate under reduced pressure, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers transformative benefits for procurement managers and supply chain directors seeking to optimize the sourcing of complex heterocyclic intermediates. The shift away from precious metal catalysts and exotic reagents towards commodity chemicals significantly de-risks the supply chain, ensuring consistent availability of raw materials even during market fluctuations. The simplicity of the reaction setup reduces the capital expenditure required for specialized reactor equipment, as standard glass-lined or stainless steel vessels are sufficient for handling the mild reaction conditions. Moreover, the high atom economy and reduced waste generation align with increasingly stringent environmental regulations, lowering the costs associated with waste disposal and environmental compliance. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-value agrochemical and pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and complex ligands results in a direct reduction in raw material costs, while the use of inexpensive inorganic bases further drives down the cost of goods. The mild reaction conditions minimize energy consumption for heating or cooling, and the simplified workup procedure reduces labor hours and solvent usage during purification. By avoiding the need for rigorous anhydrous conditions or inert atmosphere gloveboxes, the operational overhead is substantially lowered, making the production of these spiro compounds economically viable on a multi-ton scale.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as substituted maleimides and hydrazones ensures a stable supply chain that is less susceptible to geopolitical disruptions or single-source supplier bottlenecks. The robustness of the reaction across a broad range of substrates means that supply chain managers can qualify multiple vendors for raw materials without fearing significant variations in process performance. This flexibility allows for strategic stockpiling and just-in-time manufacturing strategies, reducing inventory holding costs and improving cash flow. The consistency of the method also facilitates easier technology transfer between manufacturing sites, ensuring uninterrupted supply to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with various substituents that mimic the complexity of commercial targets. The absence of heavy metal residues simplifies the regulatory approval process for pharmaceutical intermediates, as there is no need for extensive metal scavenging steps or validation of residual metal limits. The use of common organic solvents allows for established recycling protocols, minimizing the environmental footprint of the manufacturing process. This alignment with green chemistry principles not only enhances the corporate sustainability profile but also future-proofs the manufacturing process against tightening global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spiropyrazoline pyrrolidone synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the method's versatility and operational parameters. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific pipeline candidates and for procurement teams assessing the long-term viability of the supply source.
Q: What are the primary advantages of this synthesis method over traditional organocatalytic approaches?
A: This method eliminates the need for expensive transition metal catalysts or complex organocatalysts, utilizing cheap and readily available inorganic or organic bases instead. It offers superior regioselectivity and operates under mild conditions (0-100°C), significantly simplifying the workup process and reducing overall production costs.
Q: What is the substrate scope for the R groups in this spiropyrazoline pyrrolidone synthesis?
A: The process demonstrates exceptional substrate adaptability, tolerating a wide range of substituents including alkyl, cycloalkyl, aryl, heteroaryl, and various functional groups such as halogens, nitro, cyano, and methoxy groups on both the maleimide and hydrazone components without compromising yield.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the protocol is highly scalable due to its use of common solvents, inexpensive reagents, and straightforward purification methods like recrystallization or column chromatography. The high yields and lack of sensitive catalytic systems make it robust for commercial scale-up in agrochemical and pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiropyrazoline Pyrrolidone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust synthetic methodologies like the one described in CN11371717179A for advancing your drug discovery and agrochemical development programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of spiropyrazoline pyrrolidone derivatives delivered meets the highest industry standards for quality and consistency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how adopting this base-mediated route can optimize your budget without compromising on quality. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that accelerate your time to market and strengthen your competitive position in the global marketplace.
