Advanced Solid-Phase Synthesis of Cell-Permeable Peptide-Oligonucleotide Conjugates for Drug Development
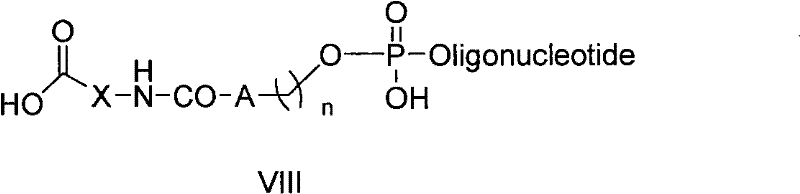
The pharmaceutical and biotechnology sectors are constantly seeking robust solutions to overcome the inherent delivery challenges of nucleic acid therapeutics. Patent CN101899092A introduces a groundbreaking approach to this persistent problem by disclosing a novel class of peptide-linker-oligonucleotide conjugates and their efficient solid-phase synthesis method. This technology addresses the critical bottleneck of cell membrane impermeability that plagues antisense oligonucleotides and siRNA molecules, offering a chemically elegant solution that covalently attaches cell-penetrating peptides directly to the therapeutic cargo. The core innovation lies in the general formula (VIII) structure, which utilizes a specialized bifunctional linker to bridge the peptide and oligonucleotide domains without compromising the integrity of either component during synthesis. This strategic design not only enhances the biological activity of the conjugate by facilitating intracellular transport but also streamlines the manufacturing process, making it a highly attractive candidate for industrial adoption.
For research directors and process chemists, understanding the limitations of conventional bioconjugation strategies is essential to appreciating the value of this new methodology. Traditionally, synthesizing peptide-oligonucleotide conjugates has been fraught with difficulties arising from the incompatibility of reaction conditions required for peptide synthesis versus oligonucleotide assembly. Conventional liquid-phase fragment condensation often suffers from low yields due to solubility issues and the necessity for extensive purification between steps. Furthermore, attempting to synthesize the peptide first requires side-chain protecting groups that are stable under oligonucleotide synthesis conditions, which severely limits the choice of amino acids and protecting group strategies. Conversely, synthesizing the oligonucleotide first restricts the peptide synthesis to Fmoc chemistry with acid-labile linkers, creating a narrow window of operational feasibility that hinders the development of diverse conjugate libraries.
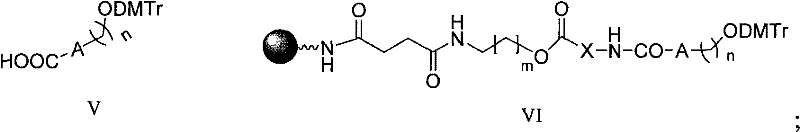
The novel approach presented in this patent circumvents these historical obstacles through a unified solid-phase step-by-step synthesis strategy. By anchoring the synthesis on a controlled pore glass (CPG) resin extended with a specific linker arm, the method allows for the sequential construction of the peptide, the linker, and finally the oligonucleotide in a single continuous flow. This eliminates the need to isolate unstable intermediates and significantly reduces the risk of racemization or degradation. The use of a versatile linker, defined by formula V where A can be a substituted phenyl ring or carbon atom, provides the necessary chemical orthogonality to tolerate both the basic conditions of Fmoc peptide deprotection and the acidic/oxidative conditions of oligonucleotide coupling. This seamless integration of two distinct chemistries on a single solid support represents a significant leap forward in process efficiency and product purity.
Mechanistic Insights into Solid-Phase Bioconjugation Strategy
The mechanistic elegance of this synthesis lies in the precise orchestration of solid-phase peptide synthesis (SPPS) and phosphoramidite chemistry. The process initiates with the functionalization of aminated CPG resin using a linker arm precursor, typically activated by uronium salts like HBTU in the presence of HOBt and DIPEA. This initial step creates a stable anchor that is crucial for the subsequent elongation of the peptide chain. The peptide sequence, often rich in cationic residues like lysine or hydrophobic residues like leucine to enhance membrane interaction, is built up residue by residue. Each coupling cycle involves deprotection of the N-terminal amine, activation of the incoming amino acid, and thorough washing to remove excess reagents, ensuring high fidelity in the peptide sequence before the linker is even introduced.
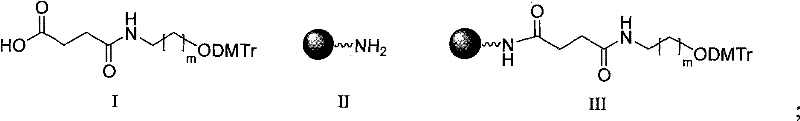
Once the peptide domain is complete, the critical ligation step occurs where the bifunctional linker (Formula V) is coupled to the N-terminus of the resin-bound peptide. This linker acts as a chemical bridge, possessing a carboxylic acid group for amide bond formation with the peptide and a hydroxyl group protected by DMTr for the subsequent initiation of oligonucleotide synthesis. The compatibility of this linker with standard DNA synthesizers is a key feature, allowing the automated addition of nucleoside phosphoramidites directly onto the peptide-linker construct. The final cleavage from the resin is achieved using strong acids like TFA or ammonia, depending on the specific protecting group strategy employed, simultaneously removing side-chain protecting groups and releasing the final conjugate into solution. This consolidated workflow minimizes handling losses and ensures that the final product retains the structural integrity required for biological function.
How to Synthesize Peptide-Linker-Oligonucleotide Conjugates Efficiently
The synthesis of these high-value bioconjugates requires strict adherence to the solid-phase protocol outlined in the patent to ensure optimal coupling efficiency and purity. The process leverages standard laboratory equipment such as automated DNA synthesizers alongside manual or automated peptide synthesis vessels, making it accessible for most well-equipped chemical biology laboratories. The key to success lies in the rigorous monitoring of each coupling step, often utilizing colorimetric tests like the Kaiser test to confirm reaction completion before proceeding to the next amino acid or nucleotide addition. Detailed standardized synthetic steps for preparing the resin, elongating the peptide, attaching the linker, and synthesizing the oligonucleotide are provided in the technical guide below.
- Functionalize aminated CPG resin with a linker arm using coupling reagents like HBTU and HOBt to create the solid support foundation.
- Synthesize the cell-penetrating peptide sequence on the resin using standard Fmoc or Boc solid-phase peptide synthesis protocols.
- Couple the specific bifunctional linker to the peptide N-terminus, followed by automated solid-phase oligonucleotide synthesis and final cleavage.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers substantial advantages by fundamentally simplifying the manufacturing workflow of complex biologics. The shift from multi-step liquid-phase convergent synthesis to a linear solid-phase process drastically reduces the number of unit operations required. In traditional methods, each fragment synthesis and subsequent conjugation would require separate reaction vessels, workup procedures, and purification columns, all of which consume significant amounts of solvents, resins, and labor hours. By consolidating these steps onto a single solid support, the material footprint of the process is significantly reduced, leading to a leaner manufacturing operation that is less susceptible to supply chain disruptions caused by the shortage of specific purification media or solvents.
- Cost Reduction in Manufacturing: The economic benefits of this solid-phase approach are driven by the elimination of intermediate isolation and purification steps. In conventional bioconjugation, purifying peptide fragments and oligonucleotide fragments separately before coupling them often results in significant yield loss, sometimes discarding more than half of the valuable material. By keeping the molecule attached to the resin until the very end, the process minimizes mechanical losses and reduces the consumption of expensive chromatography media. Furthermore, the ability to use excess reagents to drive reactions to completion without complicating downstream purification allows for higher overall throughput, effectively lowering the cost per gram of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The reliance on standard, commercially available building blocks enhances the resilience of the supply chain. The synthesis utilizes common Fmoc-amino acids and standard phosphoramidites, which are commodity chemicals produced by multiple global suppliers. This diversification of raw material sources mitigates the risk of single-source dependency that often plagues proprietary linker technologies. Additionally, the robustness of the solid-phase method means that production can be easily scaled or shifted between different manufacturing sites without requiring specialized custom equipment, ensuring consistent availability of these critical research and therapeutic tools even during periods of high market demand.
- Scalability and Environmental Compliance: While solid-phase synthesis is traditionally associated with small-scale library generation, the streamlined nature of this specific protocol facilitates easier scale-up compared to complex liquid-phase bioconjugations. The reduction in solvent usage per mole of product, due to the absence of multiple extraction and precipitation steps, aligns well with modern green chemistry initiatives. Waste generation is more predictable and contained, primarily consisting of spent resin and wash solvents which can be managed through established waste streams. This environmental efficiency not only reduces disposal costs but also simplifies regulatory compliance regarding solvent residues and impurity profiles in the final drug substance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this peptide-linker-oligonucleotide technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of this approach for your specific research or production needs. Understanding these nuances is critical for project managers planning the integration of cell-permeable oligonucleotides into their development pipelines.
Q: What is the primary advantage of the solid-phase method described in CN101899092A?
A: The primary advantage is the avoidance of compatibility issues between peptide and oligonucleotide synthesis conditions. By using a specialized linker and a unified solid-phase approach, the method eliminates the need for complex intermediate purifications and protects sensitive functional groups throughout the process.
Q: How does this conjugate improve oligonucleotide delivery?
A: The conjugate incorporates specific cell-penetrating peptide sequences (such as poly-lysine or leucine-rich motifs) covalently linked to the oligonucleotide. This structural modification significantly enhances membrane permeability, allowing the therapeutic cargo to enter cells more efficiently without the need for lipid vectors or electroporation.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the solid-phase step-by-step synthesis is designed for high-throughput preparation. It simplifies operations by keeping the growing molecule attached to the resin, reducing substrate consumption and facilitating the automation required for commercial scale-up of complex bioconjugates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Peptide-Oligonucleotide Conjugate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the solid-phase synthesis technology described in CN101899092A for advancing nucleic acid therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless. Our state-of-the-art facilities are equipped to handle the stringent purity specifications required for oligonucleotide drugs, supported by rigorous QC labs that utilize advanced HPLC and MS techniques to verify the integrity of every peptide-linker bond and nucleotide sequence.
We invite you to collaborate with us to leverage this innovative synthesis route for your next-generation therapeutics. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific target sequence, demonstrating how our optimized solid-phase processes can reduce your overall development budget. Please contact our technical procurement team today to request specific COA data for similar conjugates and to discuss route feasibility assessments for your proprietary candidates, ensuring a secure and efficient supply chain for your critical molecular biology tools and drug candidates.
