Advanced Asymmetric Synthesis Strategies for High-Purity Pharmaceutical Intermediates
Introduction to Novel Asymmetric Synthesis Pathways
The pharmaceutical industry continuously seeks robust methodologies for producing chiral intermediates with high optical purity, particularly for complex therapeutic agents like elastase inhibitors used in treating cystic fibrosis. Patent CN1218463A introduces a groundbreaking approach to the asymmetric synthesis of R-alpha-propyl-piperonylamine and its analogs, addressing critical limitations found in prior art regarding stereoselectivity and scalability. This technology provides a reliable pharmaceutical intermediates supplier with the capability to deliver high-value chiral building blocks essential for the production of orally active, non-toxic selective elastase inhibitors. The disclosed methods utilize sophisticated catalytic hydrogenation techniques and optimized Grignard additions to ensure consistent quality and yield.
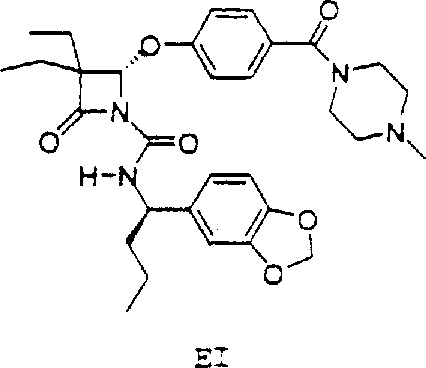
By leveraging these advanced synthetic routes, manufacturers can overcome the historical challenges associated with racemization and low diastereomeric excess that plagued earlier methodologies. The patent details two distinct yet complementary pathways: one originating from a ketone precursor via Raney Nickel catalysis, and another utilizing an aldehyde precursor through a cerium-free Grignard reaction. Both strategies are designed to facilitate cost reduction in pharmaceutical intermediates manufacturing by streamlining purification steps and utilizing commercially available starting materials. This represents a significant leap forward in process chemistry, enabling the commercial scale-up of complex pharmaceutical intermediates with enhanced supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral amines like R-alpha-propyl-piperonylamine has been fraught with technical hurdles that impede efficient large-scale production. Previous attempts, such as those described in US Patent 5,149,838, relied on Mitsunobu conditions which frequently resulted in significant side reactions and a detrimental loss of stereochemical integrity. Furthermore, methods involving hydrogenolysis of N-(1-phenylethyl)-1-arylethylamines often suffered from extremely slow reaction kinetics or unavoidable racemization of the desired product. Other literature approaches, such as those by Eleveld or Ukaji, demonstrated poor diastereoselectivity specifically for meta-substituted substrates or required the use of prohibited reagents like cerium chloride, which complicates waste management and increases operational costs. These inefficiencies create substantial bottlenecks for any reliable agrochemical intermediate supplier or pharma partner attempting to secure consistent supply.
The Novel Approach
The methodology outlined in CN1218463A circumvents these obstacles through a meticulously engineered sequence of condensation and selective hydrogenation steps. The primary route involves the formation of a chiral imine from a ketone precursor, followed by a highly stereoselective hydrogenation using Raney Nickel catalyst under specific pressure and temperature regimes. Alternatively, a second route employs a Grignard addition to a chiral imine derived from an aldehyde, notably achieving high diastereomeric excess without the need for Lewis acid chelating agents. This dual-pathway flexibility allows for cost reduction in electronic chemical manufacturing and pharma sectors by selecting the most economically viable starting material. The elimination of problematic reagents and the optimization of reaction conditions ensure that the process is not only chemically superior but also environmentally more sustainable.
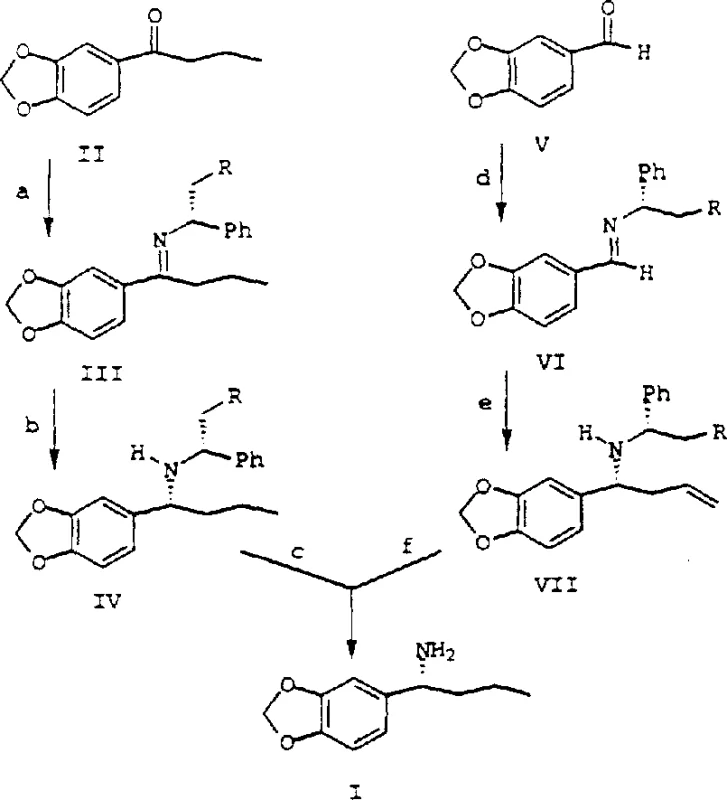
Mechanistic Insights into Raney Nickel Catalyzed Stereoselective Hydrogenation
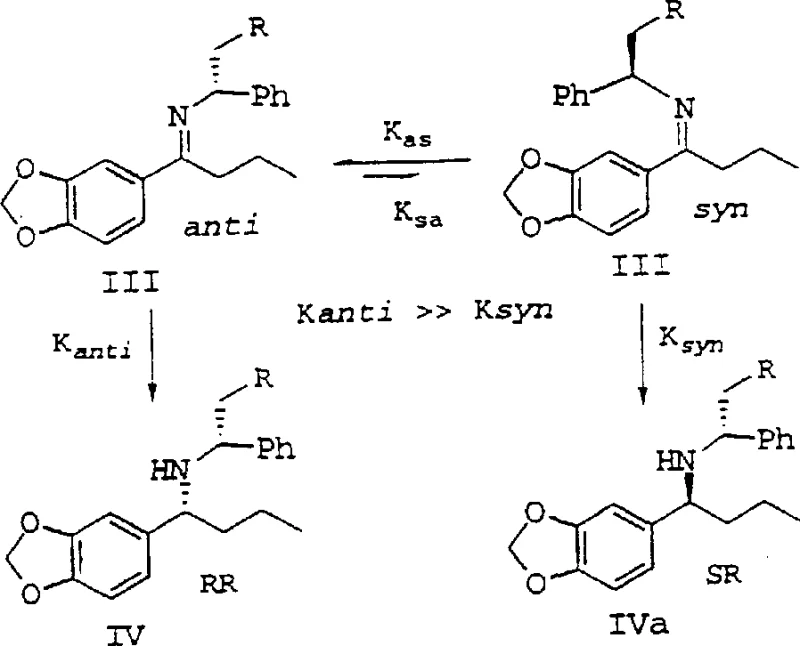
A core innovation of this patent lies in the profound understanding of the kinetic and thermodynamic factors governing the hydrogenation of chiral imines. The process utilizes Raney Nickel to reduce the imine intermediate, where the stereoselectivity is critically dependent on the interplay between hydrogen pressure and temperature. The inventors discovered that the reduction rate of the trans-imine isomer is significantly faster than that of the cis-imine isomer under specific conditions. By manipulating the reaction temperature, it is possible to influence the equilibrium constant (K_as) between the cis and trans isomers, thereby driving the reaction towards the desired stereoisomer. This mechanistic nuance allows for the achievement of diastereomeric excess values exceeding 90%, which is paramount for meeting the stringent purity specifications required in API synthesis.
Furthermore, the patent describes a sophisticated two-stage temperature protocol that maximizes this kinetic advantage. Initially, the reaction is conducted at a lower temperature to rapidly reduce the predominant trans-imine species. Subsequently, the temperature is elevated to promote the isomerization of the remaining cis-imine into the trans-form, which is then immediately reduced. This dynamic process prevents the accumulation of the unwanted stereoisomer and minimizes the risk of racemization. Such precise control over the catalytic cycle demonstrates a level of process sophistication that ensures reducing lead time for high-purity pharmaceutical intermediates while maintaining exceptional optical purity throughout the synthesis.
How to Synthesize R-alpha-Propyl-Piperonylamine Efficiently
The synthesis of this critical chiral amine involves a sequence of well-defined chemical transformations that balance reactivity with stereocontrol. The process begins with the condensation of a carbonyl compound with a chiral amine auxiliary to form an imine, followed by catalytic hydrogenation to set the stereocenter, and concludes with the removal of the auxiliary group. Detailed operational parameters regarding solvent systems, catalyst loading, and workup procedures are essential for replicating the high yields and selectivity reported in the patent. For a comprehensive understanding of the specific reagent quantities and safety protocols, please refer to the standardized synthesis guide below.
- Condense 1-(1,3-benzodioxol-5-yl)-1-butanone with chiral methylbenzylamine using TiCl4 to form the chiral imine intermediate.
- Perform selective hydrogenation of the imine using Raney Nickel catalyst under controlled temperature and pressure to achieve high diastereoselectivity.
- Execute catalytic hydrogenolysis using Pd/C in an alcohol-acid solvent system to remove the chiral auxiliary and yield the final amine salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented synthesis route offers transformative benefits for supply chain stability and cost efficiency. By eliminating the reliance on expensive and difficult-to-handle reagents such as cerium chloride or specialized phosphorus compounds required in Mitsunobu reactions, the overall raw material costs are substantially reduced. The use of common solvents like ethanol, methanol, and acetic acid further simplifies the logistics of solvent recovery and waste disposal, contributing to a greener and more cost-effective manufacturing profile. These factors collectively enhance the economic viability of producing elastase inhibitor intermediates on a multi-ton scale.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by utilizing Raney Nickel, a relatively inexpensive catalyst, instead of precious metal catalysts for the critical stereoselective step. Additionally, the avoidance of stoichiometric chelating agents reduces the material bill of goods. The streamlined workup procedures, which often involve simple filtration and crystallization rather than complex chromatographic separations, drastically lower labor and processing time expenses. This economic efficiency makes the technology highly attractive for large-scale commercial production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of hydrogen pressures and temperatures, ensures consistent batch-to-batch performance. The starting materials, such as piperonal and butyric anhydride, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The methodology is explicitly designed for industrial scale-up, with examples demonstrating successful execution from gram to kilogram scales. The absence of heavy metal contaminants in the final product simplifies the purification process and ensures compliance with strict regulatory limits for residual metals in APIs. Furthermore, the reduced generation of hazardous waste aligns with modern environmental sustainability goals, facilitating easier permitting and operation of manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this asymmetric synthesis technology. These insights are derived directly from the experimental data and embodiments described in the patent documentation, providing clarity on critical process parameters and potential troubleshooting scenarios. Understanding these nuances is vital for process chemists aiming to adapt this methodology for specific manufacturing environments.
Q: Why is Raney Nickel preferred over Palladium Carbon for the initial imine reduction?
A: According to patent CN1218463A, Raney Nickel provides significantly higher diastereoselectivity (up to 94% de) compared to Pd/C (67% de) during the hydrogenation of the chiral imine intermediate, ensuring superior optical purity.
Q: Does this process require hazardous cerium reagents for Grignard additions?
A: No, the novel route described eliminates the need for cerium chloride or other Lewis acid chelating agents, which simplifies waste treatment and reduces raw material costs while maintaining high diastereomeric excess.
Q: What are the critical parameters for maintaining stereochemistry during hydrogenation?
A: The patent highlights a two-stage temperature protocol where initial low-temperature hydrogenation reduces the trans-imine, followed by elevated temperatures to isomerize the cis-imine, preventing racemization and maximizing yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-alpha-Propyl-Piperonylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of R-alpha-propyl-piperonylamine meets the highest standards of optical and chemical purity required for clinical and commercial applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your drug development timeline and optimize your supply chain efficiency.
