Advanced Synthesis of Condensed Pyridine Mevalonolactone Intermediates for Statin Production
Introduction to Advanced Intermediate Synthesis
The pharmaceutical industry's relentless pursuit of more effective lipid-lowering agents has placed significant emphasis on the development of robust synthetic routes for HMG-CoA reductase inhibitors, commonly known as statins. Patent CN1102644A discloses a groundbreaking methodology for producing condensed pyridine type mevalonolactone intermediates, which are critical precursors in the manufacture of these life-saving medications. Unlike traditional approaches that often struggle with stereochemical control and hazardous reagent handling, this novel process introduces a highly efficient condensation strategy. By leveraging specific phosphonate or phosphonium derivatives of condensed pyridine rings, manufacturers can achieve exceptional E-isomer selectivity, a crucial factor for biological activity. This technological leap not only addresses the complex chemical challenges associated with constructing the side chain but also aligns with modern demands for greener, more scalable pharmaceutical manufacturing processes suitable for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of condensed pyridine type monohydroxy valeric acid lactone derivatives relied on stepwise chain extension methods, as referenced in earlier European and Japanese patents. These conventional pathways were fraught with significant operational difficulties, including the necessity for extremely low temperature controls and the use of dangerous borane reagents for reduction steps. Furthermore, maintaining the precise cis-structure of the two hydroxyl groups required highly sophisticated asymmetric synthetic methods or tedious optical resolution of racemic mixtures, which drastically reduced overall yield and increased production costs. The progressive extension of the side chain often led to cumulative impurities, making the purification of the final optically active material, specifically the (3R, 5S)-structure known for high activity, an arduous and economically inefficient task for large-scale operations.
The Novel Approach
The innovative method presented in the patent data revolutionizes this landscape by introducing a direct condensation reaction between a functionalized condensed pyridine derivative and a specific aldehyde compound. This approach bypasses the need for multiple chain-extension steps and eliminates the reliance on hazardous borane chemistry. Instead, it utilizes a phosphonate or phosphonium salt intermediate that reacts with a protected aldehyde under basic conditions to form the carbon-carbon double bond with remarkable precision. 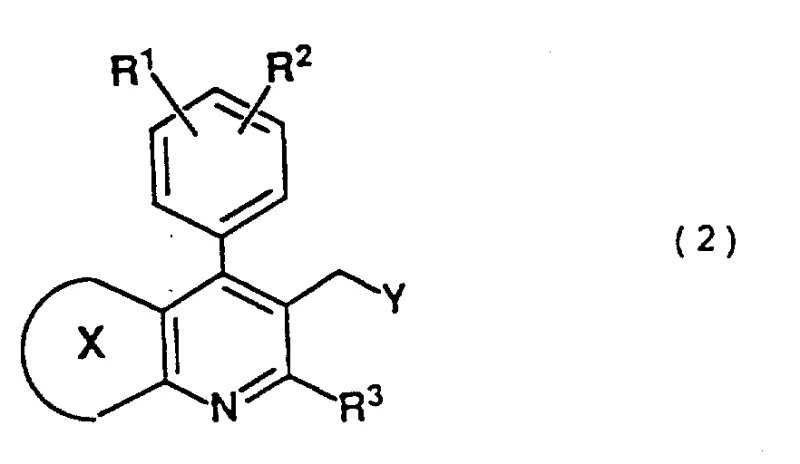 As illustrated in the reaction scheme, this convergent synthesis allows for the direct assembly of the core structure, significantly streamlining the workflow. The ability to control the stereochemistry during the bond-forming step ensures that the desired trans-configuration is obtained predominantly, thereby simplifying downstream purification and enhancing the overall economic viability of producing high-purity pharmaceutical intermediates.
As illustrated in the reaction scheme, this convergent synthesis allows for the direct assembly of the core structure, significantly streamlining the workflow. The ability to control the stereochemistry during the bond-forming step ensures that the desired trans-configuration is obtained predominantly, thereby simplifying downstream purification and enhancing the overall economic viability of producing high-purity pharmaceutical intermediates.
Mechanistic Insights into Phosphonate-Mediated Condensation
The core of this synthetic breakthrough lies in the mechanistic elegance of the Horner-Wadsworth-Emmons (HWE) or Wittig-type reaction employed to link the pyridine core with the mevalonate side chain. The process begins with the generation of a stabilized carbanion adjacent to the phosphorus group on the condensed pyridine ring. This is achieved by treating the precursor with a strong base, such as tert-butyllithium or lithium 2,2,6,6-tetramethylpiperidide (LTMP), in an anhydrous inert solvent like tetrahydrofuran (THF) at temperatures ranging from -78°C to 30°C. The resulting anion is highly nucleophilic yet sufficiently stabilized by the electron-withdrawing phosphonate group to prevent unwanted side reactions. Upon addition of the aldehyde component, specifically a protected 6-oxo-heptenoic acid ester, the anion attacks the carbonyl carbon to form a betaine or oxaphosphetane intermediate, which subsequently collapses to release the phosphate byproduct and form the desired alkene.
Crucially, the choice of base and reaction conditions profoundly influences the stereoselectivity of the resulting double bond. The patent data highlights that when using diphenyl phosphonate derivatives in conjunction with bulky bases like LTMP, the reaction exhibits fantastic trans-selectivity (E-isomer), with ratios reaching as high as 99:1. 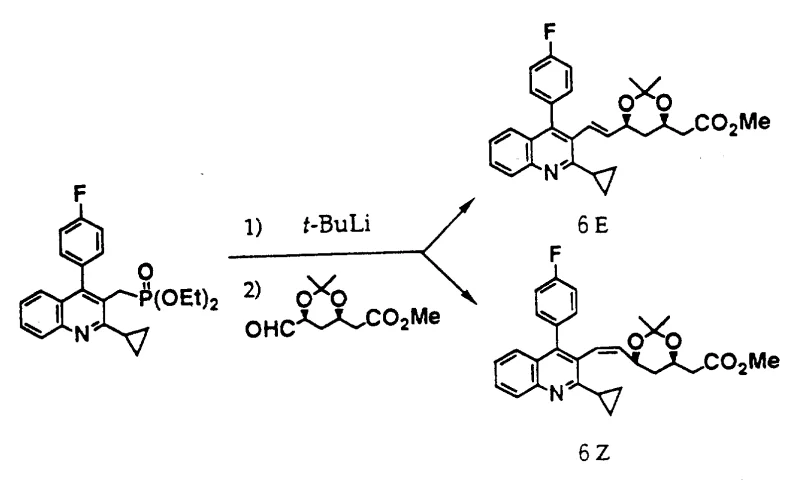 This high level of stereocontrol is vital because the biological activity of the final HMG-CoA inhibitor is strictly dependent on the spatial arrangement of the hydroxyl groups and the side chain geometry. By optimizing the electronic properties of the phosphorus substituent and the steric environment of the base, the process minimizes the formation of the inactive Z-isomer, thereby reducing the burden on chromatographic purification and ensuring a consistent, high-quality impurity profile that meets stringent regulatory standards for active pharmaceutical ingredients.
This high level of stereocontrol is vital because the biological activity of the final HMG-CoA inhibitor is strictly dependent on the spatial arrangement of the hydroxyl groups and the side chain geometry. By optimizing the electronic properties of the phosphorus substituent and the steric environment of the base, the process minimizes the formation of the inactive Z-isomer, thereby reducing the burden on chromatographic purification and ensuring a consistent, high-quality impurity profile that meets stringent regulatory standards for active pharmaceutical ingredients.
How to Synthesize Condensed Pyridine Mevalonolactone Efficiently
To implement this synthesis effectively, operators must adhere to strict anhydrous conditions and precise temperature protocols to maximize yield and selectivity. The process involves preparing the phosphonate-substituted quinoline or pyridine derivative, generating the lithiated species at cryogenic temperatures, and then coupling it with the chiral aldehyde fragment. Detailed standardized synthesis steps see the guide below.
- Prepare the condensed pyridine derivative containing a phosphonate or phosphonium group at the 3-position side chain.
- Generate the carbanion by treating the derivative with a strong base such as tert-butyllithium or lithium tetramethylpiperidide at low temperatures (-78°C).
- Condense the resulting anion with a protected 6-oxo-heptenoic acid ester aldehyde to form the desired E-isomer with high stereoselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple chemical efficiency. By replacing multi-step, low-yielding sequences with a convergent condensation strategy, the overall manufacturing timeline is drastically compressed, leading to substantial cost savings in labor and facility usage. The elimination of expensive and hazardous reagents like borane complexes not only reduces raw material costs but also mitigates the safety risks associated with storage and handling, thereby lowering insurance and compliance overheads. Furthermore, the robustness of the reaction conditions allows for easier technology transfer between sites, ensuring a more resilient and flexible supply network capable of meeting fluctuating market demands for statin intermediates without compromising on quality or delivery reliability.
- Cost Reduction in Manufacturing: The streamlined nature of this process significantly lowers the cost of goods sold by reducing the total number of unit operations required. By avoiding the need for complex asymmetric synthesis or resolution steps early in the pathway, manufacturers can achieve higher throughput with existing equipment. The high stereoselectivity means less material is wasted on inactive isomers, directly improving the mass balance and reducing the volume of solvent and silica gel needed for purification. Additionally, the use of commercially available phosphonate reagents instead of custom-synthesized organometallics further drives down the input costs, making the final API more price-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of stable, shelf-stable intermediates that do not require specialized cold-chain logistics for long-term storage. The reagents involved, such as substituted quinolines and protected aldehydes, are sourced from established supply chains, reducing the risk of bottlenecks associated with exotic or single-source chemicals. The scalability of the reaction from gram to kilogram scales has been demonstrated with consistent results, assuring partners that volume increases can be accommodated without re-optimizing the entire process. This reliability is critical for maintaining uninterrupted production schedules for downstream formulation plants.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with green chemistry principles by minimizing waste generation and avoiding toxic heavy metals or pyrophoric reagents where possible. The simplified work-up procedures reduce the volume of aqueous and organic waste streams, easing the burden on wastewater treatment facilities and ensuring compliance with increasingly strict environmental regulations. The ability to run the reaction at manageable temperatures (-78°C to room temperature) using standard industrial reactors means that scale-up to multi-ton production is feasible without requiring specialized cryogenic infrastructure, facilitating rapid commercialization and market entry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized intermediates. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing portfolios. The answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: The primary advantage is the superior stereoselectivity (E/Z ratio up to 99:1) and the elimination of harsh borane reagents, leading to higher purity and safer processing conditions.
Q: Which bases are recommended for the condensation reaction?
A: The patent specifies the use of strong bases such as tert-butyllithium, n-butyllithium, or lithium 2,2,6,6-tetramethylpiperidide (LTMP) in anhydrous ether solvents like THF.
Q: What is the final application of these intermediates?
A: These intermediates are precursors for condensed pyridine type mevalonolactone derivatives, which serve as potent HMG-CoA reductase inhibitors for treating hypercholesterolemia and arteriosclerosis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Condensed Pyridine Mevalonolactone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation cardiovascular therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the stereochemical integrity and chemical purity of every batch. Our commitment to excellence ensures that the condensed pyridine mevalonolactone intermediates we supply meet the exacting standards required for global regulatory filings.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall time to market.
