Advanced Asymmetric Synthesis of Taxol Side Chains for Commercial Scale-up of Complex Pharmaceutical Intermediates
Advanced Asymmetric Synthesis of Taxol Side Chains for Commercial Scale-up of Complex Pharmaceutical Intermediates
The global demand for paclitaxel (Taxol) and its analogues, such as docetaxel, continues to drive innovation in semi-synthetic pathways, particularly focusing on the efficient production of the chiral C-13 side chain. Patent CN1709864A introduces a groundbreaking synthetic methodology that addresses the critical bottlenecks of cost and stereocontrol in this domain. By leveraging low-cost alpha-glycolic acid as the foundational raw material and employing chirality tert-butyl sulfinamide as a powerful stereochemical director, this invention establishes a robust route to optically pure side chains. The core innovation lies in the asymmetric addition reaction induced by the chiral auxiliary, which serves as the committed step to generate the crucial beta-lactam or straight-chain precursors. This approach not only ensures high chemical product quality but also streamlines the protection group strategy, making the synthesis process significantly shorter and more operationally feasible for industrial applications.
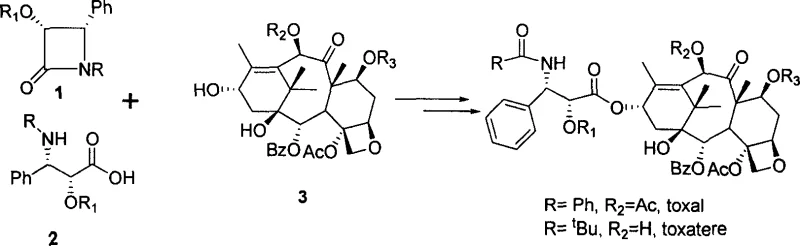
The limitations of conventional methods for synthesizing Taxol side chains have long plagued the pharmaceutical supply chain, primarily due to reliance on expensive chiral starting materials and complex multi-step sequences. Traditional approaches often utilize optical purity phenylethylamine or anisole ethylamine as initiators to prepare Schiff bases, followed by 2+2 cycloaddition reactions. While effective, these routes frequently suffer from moderate yields and require rigorous purification to remove diastereomeric impurities. Other methods involving alpha-hydroxy acetates protected with triisopropyl silicon groups or threonine methyl esters introduce additional complexity regarding protecting group manipulation and atom economy. These legacy processes often result in substantial waste generation and higher operational expenditures, creating friction for procurement teams aiming to reduce the cost of goods sold (COGS) for final API production.
In stark contrast, the novel approach detailed in this patent utilizes a highly efficient asymmetric induction strategy centered on tert-butyl sulfinyl amine chemistry. This method transforms the synthesis landscape by enabling the direct construction of the chiral center with exceptional fidelity. The process begins with the formation of an enol intermediate from a protected alpha-hydroxy acetic ester under highly basic conditions, which then reacts with an optically pure tert-butyl sulfinyl amine Schiff base. This key transformation proceeds with remarkable cis-selectivity, yielding 2-hydroxyl-3-anilino ester intermediates with defined (2R, 3S) or (2S, 3R) configurations. The elegance of this route is further enhanced by the versatility of the protecting groups, which can be easily regulated and removed under mild acidic conditions, thereby simplifying the downstream processing and enhancing the overall throughput of the manufacturing line.
Mechanistic Insights into Tert-Butyl Sulfinamide Induced Asymmetric Reduction
The mechanistic cornerstone of this synthesis is the nucleophilic attack of the enolate derived from the protected glycolic ester onto the chiral imine nitrogen of the tert-butyl sulfinyl Schiff base. Under the influence of strong bases such as lithium hexamethyldisilazide (LHMDS) or lithium diisopropylamide (LDA) at cryogenic temperatures (typically -78°C), the alpha-hydrogen of the ester is abstracted to form a reactive enolate species. This enolate then approaches the electrophilic carbon of the Schiff base in a highly organized transition state dictated by the bulky tert-butyl group on the sulfur atom. This steric bulk effectively shields one face of the imine, forcing the nucleophile to attack from the less hindered trajectory, thus securing the high diastereoselectivity observed in the experimental data. The resulting adduct possesses the precise stereochemistry required for the biological activity of the final Taxol molecule, eliminating the need for difficult resolution steps later in the synthesis.
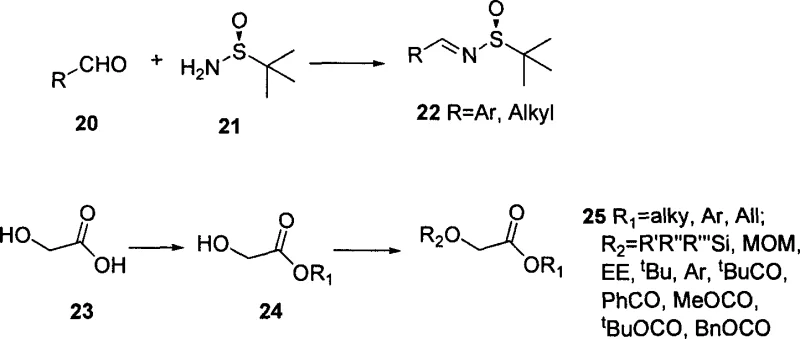
Impurity control in this process is intrinsically managed through the high selectivity of the chiral auxiliary. The patent data indicates that by optimizing the R2 substituent on the ester and the type of base used, the formation of unwanted isomers can be minimized to negligible levels. For instance, the use of carbonate-based protecting groups (like Boc) in conjunction with LHMDS has been shown to yield the desired (2R, 3S) isomer with nearly 100% diastereomeric excess in certain embodiments. This level of purity is critical for R&D directors who must ensure that the impurity profile of the intermediate meets stringent regulatory standards before it is coupled with the baccatin III core. Furthermore, the stability of the tert-butyl sulfinyl group allows it to function dually as a chiral inducer and a protecting group for the amine, which can be cleanly removed via acid hydrolysis without affecting other sensitive functionalities in the molecule.
How to Synthesize Taxol Side Chain Intermediates Efficiently
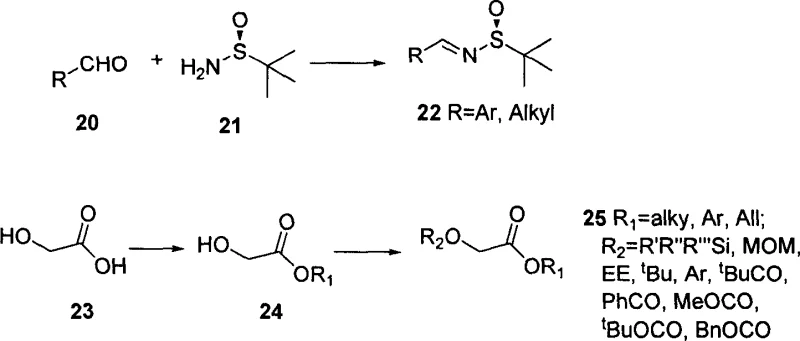
The synthesis of these high-value intermediates follows a logical progression starting from commodity chemicals. The process initiates with the preparation of the chiral Schiff base and the protected glycolic ester, followed by the key coupling reaction. The operational simplicity of this route makes it highly attractive for technology transfer teams looking to implement new capabilities. Detailed standardized synthetic steps for the preparation of the key intermediates and their subsequent conversion into the final side chain precursors are outlined below, providing a clear roadmap for laboratory and pilot plant execution.
- Preparation of chiral tert-butyl sulfinyl amine Schiff base (Compound 22) via condensation of aldehyde and optically pure tert-butyl sulfinamide.
- Generation of enolate intermediate from protected alpha-hydroxy acetic ester (Compound 25) using strong bases like LHMDS at low temperature (-78°C).
- Asymmetric nucleophilic addition of the enolate to the Schiff base to form the beta-amino ester intermediate (Compound 26) with high cis-selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits regarding cost structure and supply security. The primary driver of cost reduction is the substitution of expensive chiral pool materials with low-cost alpha-glycolic acid, a widely available commodity chemical. This shift drastically lowers the raw material entry cost, which cascades through the entire value chain. Additionally, the shortened synthesis sequence means fewer unit operations, reduced solvent consumption, and lower energy requirements per kilogram of product. The high yield of the committed step further amplifies these savings by maximizing the throughput of the reactor train, ensuring that capital assets are utilized more efficiently.
- Cost Reduction in Manufacturing: The elimination of expensive chiral initiators and the reduction in total step count directly translate to a lower cost of goods. By avoiding complex resolution processes and utilizing robust, high-yielding reactions, the manufacturing overhead is significantly decreased. The ability to use standard protecting groups that are easy to remove also reduces the consumption of specialized reagents and minimizes waste disposal costs associated with hazardous byproducts.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks like glycolic acid and benzyl alcohol derivatives mitigates the risk of supply disruptions often associated with niche natural product extracts. The synthetic nature of this route ensures a consistent and scalable supply of the side chain intermediate, independent of agricultural variables or seasonal fluctuations that affect plant-based extraction methods. This reliability is crucial for maintaining continuous API production schedules and meeting global market demand without interruption.
- Scalability and Environmental Compliance: The reaction conditions described, such as the use of THF and dichloromethane at low temperatures, are well-established in industrial organic synthesis and can be safely scaled from kilograms to metric tons. The high selectivity of the process reduces the burden on downstream purification units, leading to a smaller environmental footprint. Furthermore, the removal of the sulfinyl group generates benign byproducts, aligning with modern green chemistry principles and facilitating easier regulatory approval for the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on the feasibility and advantages of this synthetic strategy for potential partners and licensees.
Q: What is the primary advantage of using tert-butyl sulfinamide in Taxol side chain synthesis?
A: The use of chiral tert-butyl sulfinamide provides exceptional asymmetric induction, achieving high chemical yields and greater than 95% diastereoselectivity (d.e.) in the key C-C bond forming step, which simplifies downstream purification.
Q: How does this method improve cost efficiency compared to traditional routes?
A: This route utilizes low-cost alpha-glycolic acid as the starting material and features a shorter synthetic sequence with easily removable protecting groups, significantly reducing raw material costs and processing time compared to methods relying on expensive chiral pool precursors.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process employs robust reaction conditions using common solvents like THF and dichloromethane, and the key intermediates are stable solids or liquids that can be purified by standard column chromatography or crystallization, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Taxol Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable and cost-effective supply of critical oncology intermediates. Our technical team has extensively analyzed the pathway described in CN1709864A and validated its potential for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Taxol side chain intermediate meets the exacting standards required for GMP API synthesis.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in asymmetric synthesis can drive value and security for your paclitaxel production programs.
