Advanced Synthesis of Cyclosporin A Analogues: Scalable Diene Formation for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust methodologies for synthesizing complex immunosuppressive agents, particularly Cyclosporin A analogues which exhibit potent therapeutic profiles. Patent CN1798759A discloses a groundbreaking process for the preparation of Cyclosporin A analogues featuring a terminal diene group, specifically targeting compounds like (E)-ISA247. This technology represents a significant leap forward in medicinal chemistry, addressing long-standing challenges in stereoselective carbon-carbon bond formation. By leveraging a sophisticated allylation strategy followed by a Peterson elimination sequence, this method ensures high isomeric purity and operational efficiency. For global procurement teams and R&D directors, understanding the nuances of this patented route is essential for securing reliable sources of high-purity pharmaceutical intermediates. The ability to consistently produce these complex structures with minimal impurity profiles directly impacts the downstream success of drug development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of modified Cyclosporin A analogues relied on less efficient pathways that often suffered from poor stereocontrol and harsh reaction conditions. Prior art, such as methods described in earlier literature involving trimethylsilyl-propylene-boric acid pinacol esters, frequently required excessive reagent loading to drive reactions to completion. These conventional approaches often resulted in mixtures of double bond isomers, necessitating costly and time-consuming purification steps like preparative HPLC to isolate the desired therapeutic agent. Furthermore, the use of unstable organometallic reagents without proper activation led to inconsistent batch-to-batch reproducibility, posing significant risks for commercial scale-up. The formation of unwanted byproducts, such as vinyl silanes under basic conditions, further complicated the workup procedures and reduced overall process mass intensity. These inefficiencies translate directly into higher manufacturing costs and extended lead times for critical pharmaceutical intermediates.
The Novel Approach
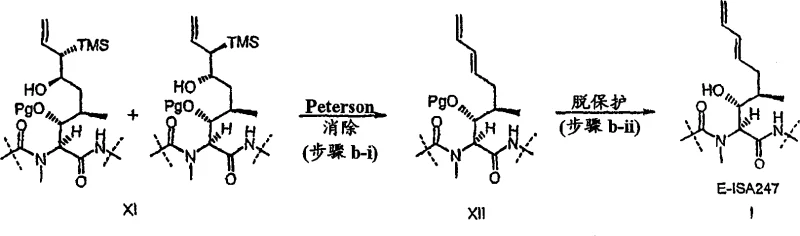
In contrast, the novel approach outlined in the patent data introduces a highly controlled allylation-Peterson elimination sequence that overcomes these historical bottlenecks. By utilizing gamma-silylated allyl metal reagents, specifically boron-based species, the process achieves remarkable stereoselectivity through a well-defined Zimmerman-Traxler transition state. The innovation lies in the ability to tune the reaction environment, using activators like tartrate esters or specific Lewis acids to enhance reactivity without compromising selectivity. This method allows for the direct formation of the trans-beta-silyl alcohol intermediate, which is the precise precursor needed for the subsequent elimination step. The result is a streamlined synthetic route that minimizes waste and maximizes the yield of the target diene structure. This technological advancement provides a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates, ensuring a steady supply of high-quality materials for drug formulation.
Mechanistic Insights into Allyl Borane-Mediated Cyclization
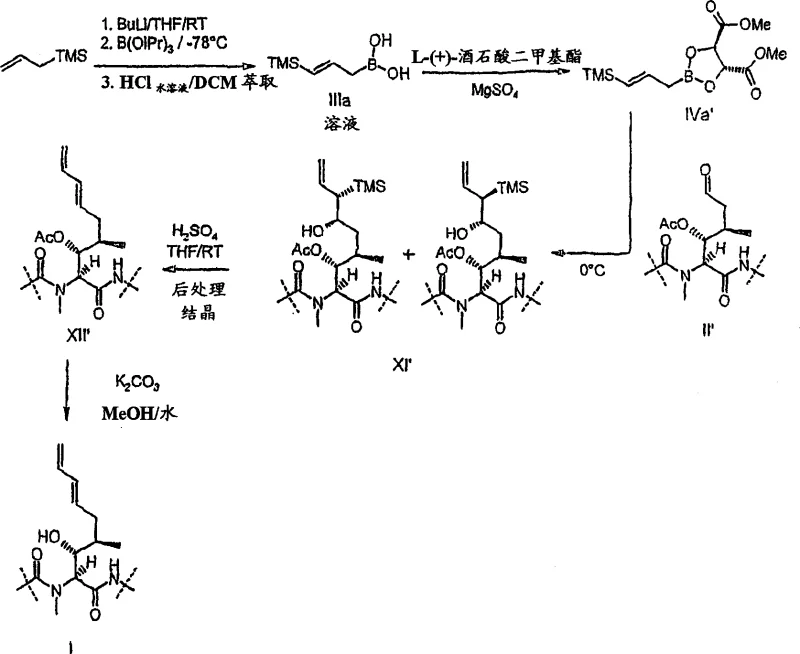
The core of this synthetic breakthrough relies on the intricate mechanistic interplay between the allyl borane reagent and the protected Cyclosporin A aldehyde. The reaction proceeds through a six-membered chair-like transition state where the spatial arrangement of substituents dictates the stereochemical outcome. When trans-gamma-silylated allyl metal reagents are employed, the preferred conformation minimizes 1,3-diaxial interactions, leading predominantly to the trans-beta-silyl alcohol diastereomer. This level of control is critical because the geometry of this alcohol intermediate directly determines the geometry of the final double bond after elimination. The patent details how varying the metal center, from boron to titanium or aluminum, can influence the reaction kinetics and selectivity, offering chemists multiple levers to optimize the process for specific manufacturing constraints. Understanding these mechanistic details allows R&D teams to troubleshoot potential deviations and maintain rigorous quality standards throughout production.
Following the allylation, the Peterson elimination step serves as the decisive transformation that installs the terminal diene functionality. This elimination is highly stereospecific, meaning the configuration of the starting alcohol dictates the configuration of the resulting alkene. Under acidic conditions, the trans-isomer yields the desired trans-double bond, which is essential for the biological activity of analogues like ISA247. The patent highlights the importance of pH control, noting that basic conditions can lead to the formation of undesired vinyl silane byproducts. By carefully selecting acidic promoters such as sulfuric acid or boron trifluoride etherate, the process ensures clean conversion to the target olefin. This mechanistic precision eliminates the need for extensive chromatographic purification, significantly simplifying the downstream processing and enhancing the overall economic viability of the manufacturing route.
How to Synthesize (E)-ISA247 Efficiently
Implementing this synthesis requires careful attention to reagent preparation and reaction conditions to maximize efficiency and yield. The process begins with the generation of the active allyl borane species, which can be prepared in situ or used as a stable complex depending on the specific variation of the method chosen. The subsequent allylation of the protected aldehyde must be monitored closely, typically using HPLC, to ensure complete conversion before proceeding to the elimination step. Detailed standardized synthesis steps are provided below to guide technical teams in replicating this high-performance route within their own facilities. Adhering to these protocols ensures that the critical quality attributes of the intermediate are maintained, facilitating a smooth technology transfer from laboratory to pilot plant.
- Prepare protected Cyclosporin A aldehyde intermediates by protecting the beta-hydroxyl group and oxidizing the terminal olefin.
- Perform stereoselective allylation using gamma-silylated allyl borane reagents, optionally activated by tartrate esters for enhanced reactivity.
- Execute acid-catalyzed Peterson elimination to form the trans-double bond, followed by deprotection to yield the final (E)-ISA247 analogue.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that resonate deeply with procurement managers and supply chain leaders. The primary benefit lies in the drastic simplification of the synthetic route, which removes the need for multiple isolation and purification stages that traditionally inflate manufacturing costs. By achieving high conversion rates and selectivity in fewer steps, the process reduces the consumption of expensive solvents and reagents, leading to significant cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the use of stable and commercially available starting materials mitigates supply chain risks associated with sourcing exotic or hazardous chemicals. This reliability ensures consistent production schedules and reduces the likelihood of delays caused by raw material shortages. The robustness of the chemistry also supports scalable operations, allowing manufacturers to respond flexibly to fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction in purification steps directly lower the operational expenditure associated with producing these complex molecules. By avoiding expensive heavy metal removal processes and minimizing solvent usage through one-pot sequences, the overall cost of goods sold is substantially decreased. This economic efficiency makes the final API more competitive in the global market while maintaining healthy margins for suppliers. Additionally, the high yield of the reaction means less raw material is wasted, further contributing to a leaner and more sustainable manufacturing model that aligns with modern green chemistry principles.
- Enhanced Supply Chain Reliability: The reliance on stable reagents like allyl trimethylsilane and triisopropylborate ensures a secure supply chain that is less vulnerable to disruptions. Unlike sensitive organometallic reagents that require special handling and storage, the reagents used in this process are robust and easily sourced from multiple global vendors. This diversity in supply options empowers procurement teams to negotiate better terms and secure long-term contracts with confidence. The predictability of the reaction outcome also reduces the risk of batch failures, ensuring that delivery commitments to downstream pharmaceutical clients are met consistently without unexpected interruptions.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents and standard reaction conditions that are easily adapted to large-scale reactors. The reduction in waste generation, particularly through the avoidance of heavy metal contaminants, simplifies environmental compliance and waste disposal procedures. This alignment with regulatory standards reduces the administrative burden on manufacturing sites and lowers the costs associated with environmental health and safety management. Consequently, the technology supports sustainable growth and enables the rapid expansion of production capacity to meet increasing global demand for these vital therapeutic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific pain points identified in the background art and the beneficial effects highlighted in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this advanced manufacturing route. By understanding these key aspects, stakeholders can make informed decisions about integrating this technology into their existing supply chains and R&D pipelines.
Q: What is the primary advantage of using tartrate-activated allyl borane reagents?
A: Tartrate activation significantly increases the reaction rate and conversion efficiency of the allylation step, allowing for high yields even with stoichiometric amounts of reagent, thereby reducing raw material waste.
Q: How is stereochemical control achieved in the diene formation?
A: Stereocontrol is managed through the specific configuration of the beta-silyl alcohol intermediate formed during allylation. Subsequent acid-catalyzed Peterson elimination ensures the formation of the desired trans-double bond isomer with high purity.
Q: Can this process be scaled for commercial manufacturing?
A: Yes, the process utilizes stable reagents and standard organic solvents like methylene dichloride and THF. The one-pot nature of certain variations minimizes isolation steps, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclosporin A Analogues Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the allylation-Peterson elimination sequence are executed with precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Cyclosporin A analogues meets the highest international standards. Our commitment to technical excellence allows us to navigate the intricacies of stereoselective synthesis, delivering materials that facilitate successful drug development outcomes for our partners worldwide.
We invite you to engage with our technical procurement team to discuss how this innovative route can optimize your supply chain. Request a Customized Cost-Saving Analysis to understand the specific economic benefits applicable to your project volume. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to support your long-term manufacturing needs. Partnering with us ensures access to cutting-edge chemistry backed by a reliable and responsive supply infrastructure dedicated to your success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
