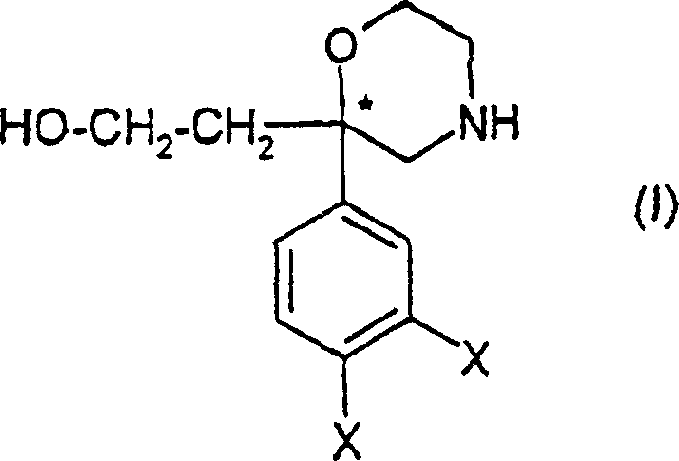
Advanced Synthesis of Enantiomerically Pure 2-(2-Arylmorpholin-2-yl)-Ethanol Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable pathways for synthesizing complex heterocyclic intermediates, particularly those serving as key scaffolds for receptor antagonists. Patent CN1266115C discloses a groundbreaking methodology for the preparation of substituted 2-(2-arylmorpholin-2-yl) ethanol derivatives in the form of pure enantiomers. These compounds are critical precursors for tachykinin receptor antagonists, which hold significant therapeutic potential for treating disorders in the respiratory, gastrointestinal, and central nervous systems. The disclosed technology addresses long-standing inefficiencies in prior art by introducing a streamlined sequence that leverages hydroboration-oxidation chemistry to functionalize the morpholine ring with high stereochemical fidelity. This innovation not only enhances the chemical elegance of the synthesis but also provides a viable route for the commercial scale-up of complex pharmaceutical intermediates, ensuring a reliable supply chain for downstream drug development.
Historically, the synthesis of these morpholine-based architectures has been plagued by prohibitive costs and dismal yields, creating bottlenecks for reliable pharmaceutical intermediate suppliers. Conventional methods, such as those described in international application WO96/23787, often rely on convoluted sequences that result in overall yields as low as 1-2% when calculated from basic starting materials like benzaldehyde. Furthermore, alternative enantioselective approaches described in academic literature frequently necessitate the use of prohibitively expensive reagents, including dichloro-bis(triphenylphosphine)palladium(II) and AD-mix formulations. These legacy processes impose a heavy financial burden on cost reduction in API manufacturing, rendering them economically unfeasible for large-scale industrial application. The reliance on precious metal catalysts also introduces significant challenges regarding heavy metal removal and environmental compliance, further complicating the supply chain logistics for high-volume production.
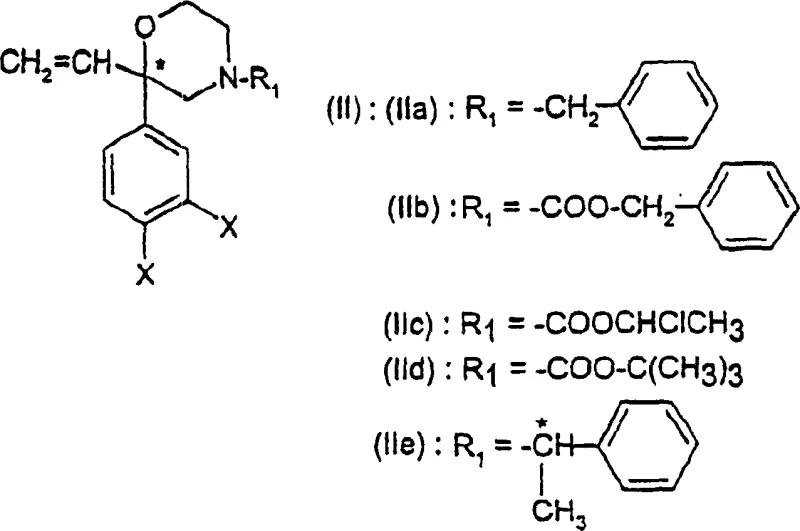
In stark contrast, the novel approach detailed in CN1266115C utilizes a sophisticated yet practical strategy centered on the transformation of vinylmorpholine intermediates. By employing readily available borane complexes, such as 9-borabicyclo[3.3.1]nonane (9-BBN) or borane-tetrahydrofuran, the process achieves a remarkable improvement in efficiency, with overall yields reaching approximately 5-25%. This method bypasses the need for costly transition metal catalysts, substituting them with inexpensive and abundant boron reagents. The core of this innovation lies in the regioselective hydroboration of the vinyl group followed by oxidative workup, which installs the crucial hydroxyethyl side chain with excellent control. This shift in chemical strategy represents a paradigm change, offering a pathway that is not only chemically superior but also aligns perfectly with the economic imperatives of modern pharmaceutical intermediates production.
Mechanistic Insights into Hydroboration-Oxidation and Morpholine Cyclization
The mechanistic elegance of this synthesis is rooted in the precise manipulation of the morpholine ring's substituents. The process begins with the formation of a vinylmorpholine scaffold, which serves as the pivotal intermediate for subsequent functionalization. The conversion of this vinyl group into a 2-hydroxyethyl moiety is achieved through a hydroboration-oxidation sequence that proceeds with high regioselectivity. The use of bulky borane reagents like 9-BBN ensures that the boron atom adds to the less hindered terminal carbon of the vinyl group, setting the stage for the correct placement of the hydroxyl functionality upon oxidation. This step is critical for maintaining the structural integrity required for biological activity in the final tachykinin antagonist. Furthermore, the reaction conditions are meticulously optimized to prevent racemization, preserving the stereochemical information established in earlier steps or introduced via chiral resolution.
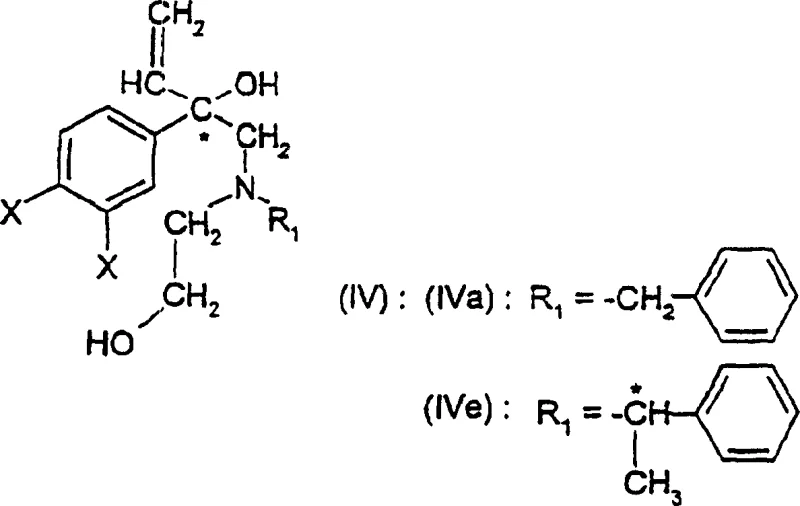
Another key mechanistic feature is the cyclization strategy used to construct the morpholine ring itself. The patent describes a phase-transfer catalyzed cyclization of diol precursors using alkyl or aryl sulfonyl halides. This reaction proceeds through the formation of a sulfonate ester intermediate, which undergoes intramolecular nucleophilic substitution to close the ring. The use of phase transfer catalysts, such as tetrabutylammonium bisulfate, facilitates this transformation in a biphasic system, enhancing reaction rates and simplifying workup procedures. Importantly, when applied to enantiomerically pure diols, this cyclization proceeds with retention of configuration at the asymmetric carbon center, ensuring that the optical purity of the starting material is faithfully transmitted to the morpholine product. This mechanistic robustness is essential for producing high-purity pharmaceutical intermediates that meet stringent regulatory standards.
How to Synthesize 2-(2-Arylmorpholin-2-yl)-Ethanol Efficiently
The synthesis of these valuable intermediates involves a multi-step sequence that integrates classical organic transformations with modern stereochemical control techniques. The process typically initiates with the preparation of a halogenated ketone via Friedel-Crafts acylation, followed by Grignard addition to introduce the vinyl functionality. Subsequent alkylation with amino-alcohol derivatives and cyclization yields the key vinylmorpholine intermediate. The final and most critical transformation involves the hydroboration-oxidation of the vinyl group to generate the target hydroxyethyl side chain. Detailed operational parameters, including solvent exchanges to mitigate safety risks associated with peroxide-THF mixtures and specific temperature profiles for exothermic oxidation steps, are crucial for success. For a comprehensive guide on executing these transformations with maximum efficiency and safety, please refer to the standardized protocol below.
- Prepare the vinylmorpholine intermediate via cyclization of the corresponding amino-alcohol precursor using phase transfer catalysis.
- Perform hydroboration on the vinyl group using 9-BBN or borane-THF complexes followed by oxidation with hydrogen peroxide.
- Resolve the racemic mixture using optically active organic acids such as di-p-toluoyl tartaric acid to obtain pure enantiomers.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple yield improvements. The elimination of expensive palladium catalysts and specialized chiral reagents like AD-mix drastically reduces the raw material cost base, leading to substantial cost savings in the overall manufacturing budget. By relying on commodity chemicals such as borane complexes and common sulfonyl chlorides, the process mitigates the risk of supply disruptions associated with scarce or geographically concentrated reagents. This shift enhances supply chain resilience, ensuring reducing lead time for high-purity intermediates and providing a more predictable production schedule for downstream API manufacturers. The simplified reagent profile also translates to easier sourcing and inventory management, allowing procurement teams to negotiate more favorable terms with vendors.
Furthermore, the process design inherently supports environmental compliance and operational safety, which are increasingly critical factors in global supply chains. The avoidance of heavy metal catalysts eliminates the need for complex and costly metal scavenging steps, reducing waste generation and simplifying effluent treatment. The patent explicitly addresses safety concerns by recommending solvent swaps from THF to toluene before the oxidation step, thereby minimizing the risk of hazardous peroxide accumulation. These features facilitate smoother regulatory approvals and reduce the environmental footprint of the manufacturing process. Consequently, this technology enables commercial scale-up with greater confidence, offering a sustainable and economically viable solution for the long-term production of these essential pharmaceutical building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these morpholine derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production workflows.
Q: What is the primary advantage of the new synthesis method over conventional routes?
A: The new method significantly improves overall yield from approximately 1-2% in conventional processes to 5-25%, while eliminating the need for expensive palladium catalysts and complex reagents like AD-mix.
Q: How is enantiomeric purity achieved in this process?
A: Enantiomeric purity is secured through resolution using optically active organic acids such as L-(-)-di-p-toluoyl tartaric acid or by employing chiral auxiliaries during the asymmetric synthesis steps.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes robust reagents like borane complexes and standard phase transfer catalysts, avoiding hazardous peroxide-THF mixtures by switching to aromatic solvents, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Arylmorpholin-2-yl)-Ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-(2-arylmorpholin-2-yl)-ethanol intermediate adheres to the highest quality standards required by global regulatory bodies. Our expertise in handling complex heterocyclic chemistry allows us to optimize these novel processes for maximum yield and minimal environmental impact.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this improved synthesis can enhance your project's economics. Please contact us to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your path to market with reliable, high-quality intermediates.
