Optimized Industrial Synthesis of Imipenem Monohydrate Crystals for Global Pharmaceutical Supply Chains
Introduction to Advanced Imipenem Manufacturing Technology
The global demand for potent carbapenem antibiotics continues to rise, driven by the critical need for effective treatments against resistant bacterial strains. Imipenem, recognized as one of the highest-evaluated antibiotic varieties since its market introduction, remains a cornerstone in clinical therapy, particularly when co-administered with cilastatin sodium to inhibit renal dehydropeptidase. However, the complexity of its molecular structure has historically posed significant challenges for manufacturers seeking efficient, high-yield production methods. A pivotal advancement in this field is detailed in patent CN106083859B, which discloses a novel preparation method for Imipenem monohydrate crystals. This technology leverages a double-protection mother nucleus compound as a strategic starting material, employing a streamlined sequence of acylation, condensation, and catalytic hydrogenation. By optimizing reaction conditions and eliminating unnecessary isolation steps, this approach addresses long-standing issues regarding impurity profiles and process yield, offering a robust solution for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Imipenem has been plagued by operational complexities that hinder large-scale industrial application. Early routes, such as those disclosed in US4374772, involved reacting thienamycin derivatives with benzylformamidinium hydrochloride. While conceptually straightforward, these methods imposed severe requirements on reaction conditions, particularly regarding pH control, leading to cumbersome operations and consistently low yields. Furthermore, alternative strategies involving the phosphorylation of a carbapenem bicyclic nucleus often resulted in unstable intermediates. For instance, routes utilizing p-nitrobenzyl protected side chains frequently encountered issues where the double-protection imipenem was prone to losing the imine methyl group during the hydrogenation process, reverting to thienamycin and generating significant impurities. Other industrial processes attempted to mitigate this by isolating intermediates as solids and washing them with solvents to remove clathrates, but these additional purification steps invariably impacted the overall process yield and increased production costs.

The Novel Approach
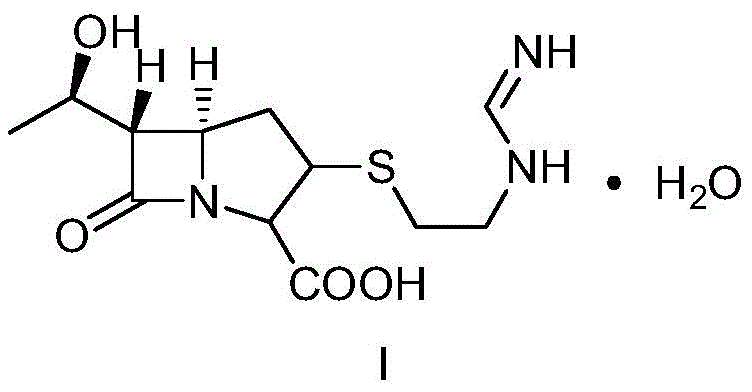
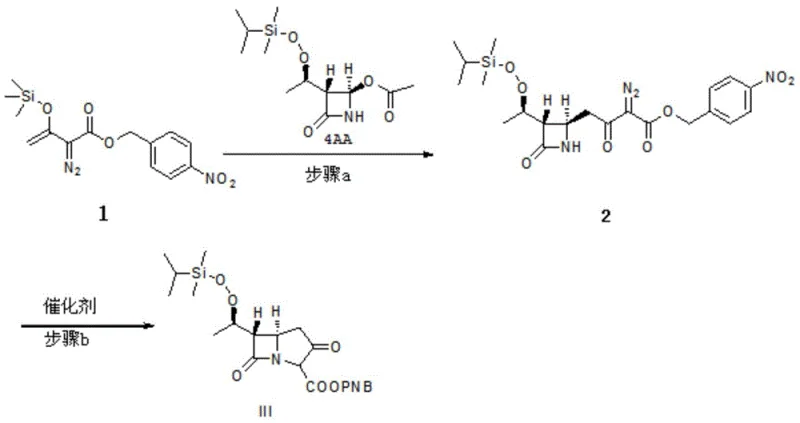
The methodology outlined in patent CN106083859B represents a significant departure from these inefficient legacy processes. By selecting a specific double-protection mother nucleus compound III, the new route initiates with an acylation reaction using diphenoxyl phosphoryl chloride to generate intermediate IV. Crucially, this intermediate is not isolated; instead, it reacts directly with cysteamine hydrochloride to form a solvate intermediate VI. This "one-pot" style progression minimizes material loss and exposure to degradation. Subsequent condensation with benzylformamidinium hydrochloride yields intermediate VII, which is then subjected to catalytic hydrogenation. The process concludes with a controlled crystallization step to obtain the Imipenem monohydrate crystal. This integrated approach not only simplifies the equipment requirements but also drastically reduces the generation of by-products, making it highly suitable for the commercial scale-up of complex antibiotic intermediates.
Mechanistic Insights into Phosphorylation and Catalytic Hydrogenation
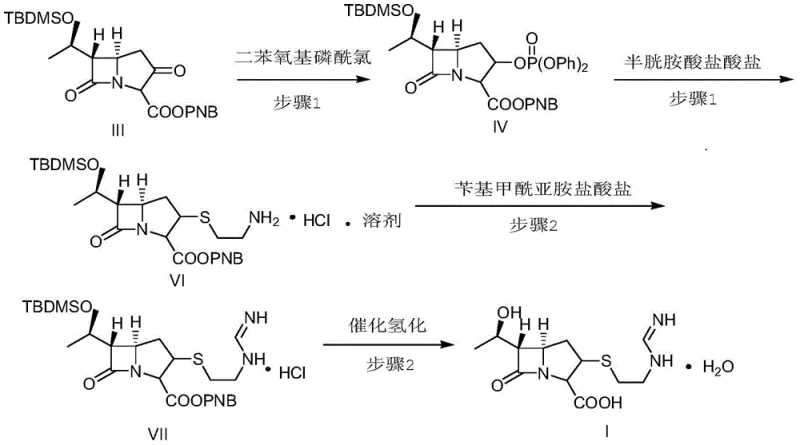
The success of this synthesis relies heavily on the precise control of the phosphorylation and subsequent condensation mechanisms. In the initial step, the reaction is conducted in an organic base environment, preferably using N,N-diisopropylethylamine or diisopropylamine, within a solvent system such as a mixture of N-methylpyrrolidone and methanol. The temperature is rigorously maintained between -70°C and -10°C to ensure selectivity. The use of diphenoxyl phosphoryl chloride is strategic; it activates the beta-lactam ring effectively without introducing the instability associated with other protecting groups. Following this, the addition of cysteamine hydrochloride allows for the nucleophilic attack on the activated intermediate, forming the critical sulfur-containing side chain. The formation of the solvate intermediate VI is a key feature, as it stabilizes the molecule prior to the final condensation, preventing the premature degradation often seen in unprotected species.
Furthermore, the final hydrogenation step is engineered to maximize purity through a sophisticated buffer system. The aqueous solution of intermediate VII is transferred to a hydrogenation kettle where a buffer solution—comprising purified water, isopropanol, N-methylmorpholine, and hydrochloric acid—is added to adjust the pH to a narrow window of 7.2 to 7.5. This specific pH range is vital for the activity of the palladium carbon or platinum carbon catalyst while simultaneously suppressing hydrolytic degradation of the sensitive beta-lactam ring. The reaction proceeds at moderate temperatures (25°C) and pressures (2.5 MPa), ensuring complete removal of the protecting groups without compromising the structural integrity of the final Imipenem molecule. This mechanistic precision ensures that the final product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize Imipenem Monohydrate Efficiently
The synthesis of Imipenem monohydrate via this patented route involves a sequence of highly optimized chemical transformations designed for reproducibility and yield. The process begins with the preparation of the double-protected mother nucleus, followed by the critical phosphorylation and thiolation sequence, and concludes with condensation and hydrogenation. Each step requires strict adherence to temperature controls and stoichiometric ratios to minimize impurity formation. For R&D teams looking to implement this technology, understanding the interplay between the solvent systems and the specific organic bases used is paramount. The following guide outlines the standardized operational framework derived from the patent data, providing a clear pathway for laboratory validation and subsequent process scaling.
- Perform acylation on double-protected mother nucleus compound III with diphenoxyl phosphoryl chloride in an organic base environment, followed by direct reaction with cysteamine hydrochloride to obtain solvate intermediate VI.
- Conduct a condensation reaction between intermediate VI and benzylformamidinium hydrochloride in a solvent system, followed by aqueous workup to isolate the aqueous solution of intermediate VII.
- Execute catalytic hydrogenation on the aqueous solution of intermediate VII using a palladium or platinum catalyst in a buffered system, followed by crystallization to obtain the final Imipenem monohydrate crystal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediates manufacturing achieved through process intensification. By eliminating the isolation of the phosphorylated intermediate and proceeding directly to the thiolation step, the process reduces solvent consumption, energy usage for drying, and labor hours associated with multiple filtration and washing cycles. This streamlining translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market without sacrificing margin.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps significantly lowers operational expenditures. Traditional methods often require extensive solvent washing to remove clathrates or impurities, which not only consumes vast quantities of expensive solvents like dichloromethane and methanol but also results in product loss during each transfer. By maintaining the intermediate in a solvated form and utilizing a direct condensation pathway, this method minimizes material handling and waste generation. Furthermore, the use of robust, commercially available reagents such as diphenoxyl phosphoryl chloride and cysteamine hydrochloride ensures that raw material costs remain stable and predictable, avoiding the volatility associated with specialized or custom-synthesized reagents.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route directly contributes to enhanced supply chain reliability and reduced lead time for high-purity pharmaceutical intermediates. Because the process tolerates a wider range of operating conditions compared to the pH-sensitive legacy routes, the risk of batch failure due to minor process deviations is significantly mitigated. This reliability ensures consistent delivery schedules, which is critical for downstream API manufacturers who operate on tight production timelines. Additionally, the simplicity of the equipment requirements—standard reactors capable of low-temperature control and hydrogenation—means that production can be easily distributed across multiple manufacturing sites if necessary, further de-risking the supply chain against localized disruptions.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method supports sustainable manufacturing practices. The reduction in solvent usage and the avoidance of complex purification steps inherently lower the volume of hazardous waste generated per kilogram of product. This aligns with increasingly stringent global environmental regulations regarding solvent emissions and waste disposal. Moreover, the process is inherently scalable; the crystallization and filtration steps described are standard unit operations that translate seamlessly from pilot plant to multi-ton commercial production. This scalability ensures that suppliers can rapidly ramp up capacity to meet surges in demand, such as those driven by public health emergencies, without requiring prohibitive capital investment in new technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Imipenem synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific manufacturing capabilities.
Q: How does this new preparation method improve upon conventional Imipenem synthesis routes?
A: Unlike conventional routes that suffer from severe pH control requirements, unstable side chains, or complex solvent washing steps that reduce yield, this method utilizes a direct phosphorylation and condensation strategy. It avoids the generation of unnecessary impurities during the phosphorylation step and eliminates the need for isolating intermediates between phosphorylation and thiolation, resulting in a higher overall yield and simpler operation suitable for industrial mass production.
Q: What are the critical reaction conditions for ensuring high purity in the hydrogenation step?
A: The hydrogenation step requires precise control of the buffer system to maintain a pH between 7.2 and 7.5. The process utilizes a specific buffer solution composed of purified water, a solvent like isopropanol, an organic base such as N-methylmorpholine, and an acid like hydrochloric acid. Maintaining this pH range during the catalytic hydrogenation with palladium carbon or platinum carbon is essential to prevent degradation and ensure the formation of high-purity Imipenem monohydrate crystals.
Q: Is this synthesis route scalable for commercial manufacturing of antibiotic intermediates?
A: Yes, the method is explicitly designed for industrial scalability. It employs robust starting materials and standard unit operations such as stirring, crystallization, and filtration. The elimination of complex isolation steps and the use of common solvents like N-methylpyrrolidone, methanol, and dichloromethane facilitate easy scale-up from laboratory to multi-ton production without compromising product quality or safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imipenem Monohydrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthesis routes like CN106083859B are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch of Imipenem monohydrate or related carbapenem intermediate meets the highest international standards for safety and efficacy.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your product quality and your bottom line.
