Scalable Synthesis of C10 Dialdehyde Monoacetals for Advanced Carotenoid Manufacturing
Scalable Synthesis of C10 Dialdehyde Monoacetals for Advanced Carotenoid Manufacturing
The production of high-value asymmetric carotenoids, such as xanthophylls and meso-zeaxanthin, relies heavily on the availability of precise C10 building blocks that allow for selective chain elongation. Patent CN1241890C discloses a robust and industrially viable process for preparing 2,7-dimethyl-2,4,6-octatrienal monoacetals, a critical intermediate class often referred to as C10-dialdehyde monoacetals. This technology addresses long-standing challenges in carotenoid synthesis by providing a route that avoids the formation of symmetric byproducts and eliminates the need for hazardous cryogenic conditions. By utilizing a strategic combination of Wittig-Horner olefination, safe hydride reduction, and catalytic oxidation, this method ensures high regioselectivity and purity, making it an essential asset for manufacturers aiming to produce complex nutraceutical and pharmaceutical ingredients.
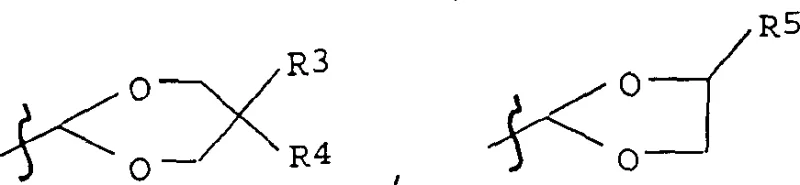
The significance of this intermediate lies in its ability to facilitate the construction of unsymmetric carotenoid structures where different end groups are required. Traditional approaches often struggle with the differentiation of the two carbonyl groups in a C10 dialdehyde, leading to mixtures that are difficult and costly to separate. The methodology outlined in CN1241890C provides a solution by introducing the acetal protecting group early in the synthesis on the C5 precursor, thereby locking in the asymmetry before the final carbon-carbon bond-forming events. This strategic foresight not only simplifies the downstream purification processes but also significantly enhances the overall yield of the desired mono-protected species, which is crucial for the economic feasibility of producing high-margin carotenoids.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing C10-dialdehyde monoacetals have been plagued by significant technical and economic drawbacks that hinder large-scale implementation. For instance, historical literature describes the direct acetalization of C10-dialdehyde using methylal and acid catalysts, a process that suffers from poor selectivity and necessitates complicated low-temperature crystallization steps to isolate the monoacetal from symmetric diacetal byproducts. Furthermore, alternative routes involving the reduction of nitrile precursors to aldehydes using diisobutyl aluminium hydride (DIBAL-H) require extreme cryogenic conditions, typically around -70°C, which imposes a heavy energy burden and limits reactor throughput. These conventional pathways often result in insufficient productive rates, sometimes as low as 37% after tedious purification, and generate substantial chemical waste, making them unattractive for modern green chemistry standards and cost-sensitive supply chains.
The Novel Approach
In stark contrast to these legacy methods, the process defined in CN1241890C introduces a streamlined three-step sequence that begins with readily available C5 precursors. The innovation lies in the condensation of an ester phosphonium salt or ester phosphonate with a protected C5-acetal aldehyde via a Wittig or Wittig-Horner reaction. This initial step constructs the polyene chain with the protecting group already in place, effectively bypassing the selectivity issues associated with protecting a symmetrical dialdehyde post-synthesis. The subsequent transformation of the ester functionality into the aldehyde is achieved through a mild reduction-oxidation sequence that avoids hazardous reagents and extreme temperatures. This approach not only guarantees the structural integrity of the sensitive polyene chain but also allows for the isolation of intermediates with high purity, thereby reducing the need for resource-intensive chromatographic separations.
Mechanistic Insights into Wittig-Horner Condensation and Catalytic Oxidation
The core of this synthetic strategy involves a highly controlled Wittig or Wittig-Horner olefination followed by a sophisticated functional group interconversion. In the first stage, the reaction between the phosphorus ylide or phosphonate and the C5-acetal aldehyde is conducted in inert organic solvents such as toluene, THF, or DMSO. The choice of base is critical; the patent highlights the efficacy of alkali metal alcoholates or hexamethyldisilazane bases, which facilitate the formation of the desired E-isomer with high stereoselectivity. This step is pivotal because it establishes the conjugated triene system characteristic of carotenoid intermediates while maintaining the orthogonality of the acetal protecting group, ensuring it remains intact under the basic reaction conditions.
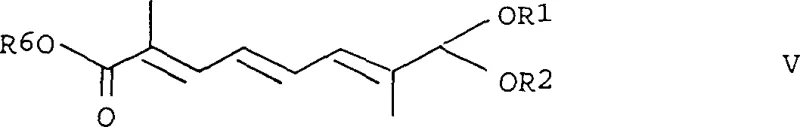
The conversion of the intermediate ester to the final aldehyde represents a significant mechanistic advancement over prior art. Instead of using stoichiometric and hazardous hydride reagents, the process employs a two-step protocol: reduction to the alcohol followed by catalytic oxidation. The reduction utilizes dihydro bis(2-methoxyethoxy) sodium aluminate (commercially known as Vitride), a reagent that is non-pyrophoric and stable in air, allowing the reaction to proceed safely at temperatures between -20°C and 30°C. The subsequent oxidation step leverages a catalytic system involving TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) and copper salts under an oxygen atmosphere. This catalytic cycle efficiently converts the allylic alcohol to the aldehyde without over-oxidation to the carboxylic acid, a common side reaction in traditional manganese dioxide oxidations, thus preserving the valuable carbon skeleton.
How to Synthesize 2,7-Dimethyl-2,4,6-octatrienal Monoacetals Efficiently
The synthesis of these critical intermediates is designed for operational simplicity and high throughput, making it ideal for contract development and manufacturing organizations. The process begins with the condensation of the phosphorus component and the protected aldehyde, followed by an aqueous workup that yields the crude acetal ester in quantitative amounts. This crude material is sufficiently pure to be telescoped directly into the reduction step without further purification, minimizing solvent usage and processing time. The reduction with Vitride is performed in a toluene-THF mixture, and the resulting alcohol is isolated via phase separation and washing, again yielding a crude product suitable for immediate use in the final oxidation. This telescoping capability is a key feature that drives down manufacturing costs and reduces the physical footprint required for production.
- Condense an ester phosphonium salt or ester phosphonate with a C5-acetal aldehyde via Wittig or Wittig-Horner reaction to form an acetal ester intermediate.
- Reduce the resulting acetal ester to an acetal alcohol using a safe hydride reagent such as dihydro bis(2-methoxyethoxy) sodium aluminate (Vitride) at mild temperatures.
- Oxidize the acetal alcohol to the final aldehyde monoacetal using a catalytic system such as TEMPO/CuCl under oxygen atmosphere.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers tangible benefits in terms of risk mitigation and cost efficiency. The elimination of cryogenic conditions (-70°C) removes the need for specialized low-temperature reactors and the associated high energy consumption, leading to significant operational expenditure savings. Furthermore, the replacement of pyrophoric reagents like lithium aluminum hydride or DIBAL-H with the safer Vitride reagent reduces the safety risks associated with storage and handling, potentially lowering insurance premiums and safety compliance costs. The robustness of the process ensures consistent batch-to-batch quality, which is essential for maintaining uninterrupted supply lines to downstream customers in the nutraceutical and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the use of catalytic oxidation rather than stoichiometric heavy metal oxidants. Traditional methods using manganese dioxide require large molar excesses of the oxidant and generate tons of solid manganese waste that must be disposed of, incurring high environmental fees. By switching to a TEMPO/copper catalytic system with oxygen as the terminal oxidant, the process drastically reduces raw material costs and waste disposal liabilities. Additionally, the ability to telescope intermediates without purification reduces solvent consumption and labor hours, further driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents enhances supply chain resilience. Reagents like Vitride are supplied as stable toluene solutions, eliminating the logistics challenges associated with shipping moisture-sensitive solids that require special packaging and handling. The mild reaction conditions also mean that the process can be executed in a wider range of manufacturing facilities, including those without specialized cryogenic infrastructure, thereby diversifying the potential supplier base and reducing the risk of single-source bottlenecks.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns well with modern green chemistry principles. The avoidance of chlorinated solvents in favor of toluene, ethers, and esters, combined with the reduction of heavy metal waste, simplifies the permitting process for scale-up. The high selectivity of the reaction minimizes the formation of isomeric impurities, which reduces the load on wastewater treatment plants and lowers the volume of organic waste generated per kilogram of product. This makes the technology highly scalable from pilot plant to multi-ton commercial production without encountering the environmental hurdles that often plague older carotenoid synthesis routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a clear picture of the process capabilities and limitations.
Q: Why is the Vitride reduction method preferred over traditional hydride reagents for this synthesis?
A: Traditional reagents like DIBAL-H require cryogenic conditions (-70°C) which are energy-intensive and difficult to scale. The patented method uses Vitride, which is non-pyrophoric, oxygen-insensitive, and operates effectively at mild temperatures between -20°C and 30°C, significantly enhancing operational safety and scalability.
Q: How does this process improve the purity profile compared to prior art methylal protection methods?
A: Prior art methods involving direct acetalization of dialdehydes often result in insufficient yields and require complex low-temperature crystallizations to separate symmetric byproducts. This novel route builds the carbon skeleton selectively via Wittig coupling first, ensuring high regioselectivity and allowing for high-purity isolation (up to 98.2% GC purity) through standard crystallization.
Q: What are the environmental advantages of the catalytic oxidation step described in the patent?
A: Conventional oxidation using manganese dioxide requires stoichiometric amounts of heavy metal oxidant and generates substantial solid waste, along with requiring extremely long reaction times (up to 60 hours). The patented catalytic oxidation utilizes TEMPO/copper systems with oxygen as the terminal oxidant, drastically reducing waste generation and reaction time while eliminating heavy metal disposal issues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-Dimethyl-2,4,6-octatrienal Monoacetal Supplier
At NINGBO INNO PHARMCHEM, we understand the critical role that high-purity intermediates play in the successful commercialization of complex carotenoids. Our technical team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We utilize stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,7-dimethyl-2,4,6-octatrienal monoacetal meets the exacting standards required for pharmaceutical and nutraceutical applications. Our commitment to quality assurance means that you can rely on us for consistent material that facilitates smooth downstream synthesis.
We invite you to discuss how our manufacturing capabilities can support your specific project requirements. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall production costs.
