Scalable Synthesis of Anticancer Tetrahydro-beta-carboline Indole Derivatives via Copper Catalysis
Scalable Synthesis of Anticancer Tetrahydro-beta-carboline Indole Derivatives via Copper Catalysis
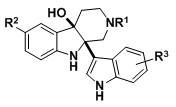
The pharmaceutical industry is constantly seeking robust and scalable pathways to access complex heterocyclic scaffolds that possess potent biological activities. A significant breakthrough in this domain is detailed in Chinese Patent CN112574198B, which discloses a novel class of indolized derivatives of tetrahydro-beta-carboline. These compounds are synthesized through a highly efficient copper-ion catalyzed oxidative coupling reaction between tetrahydro-beta-carboline precursors and various indole derivatives. This methodology represents a paradigm shift in late-stage functionalization, utilizing molecular oxygen as a green oxidant under remarkably mild conditions. For research directors and procurement specialists alike, this patent offers a compelling route to generate diverse libraries of anticancer agents without the need for extreme temperatures or pressures, thereby streamlining the supply chain for critical oncology intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of C-C bonds between electron-rich heterocycles like carboline and indole systems has been fraught with synthetic challenges. Conventional approaches often rely on pre-functionalized starting materials, such as halogenated substrates requiring stoichiometric organometallic reagents, or harsh acidic conditions that can degrade sensitive functional groups. These legacy methods frequently necessitate rigorous exclusion of moisture and oxygen, demanding specialized equipment and inert gas manifolds that increase capital expenditure. Furthermore, the use of precious metal catalysts like palladium or rhodium in cross-coupling reactions introduces significant cost burdens and complicates downstream purification due to strict limits on residual heavy metals in pharmaceutical products. The inability to directly utilize simple, unactivated indoles and tetrahydro-beta-carbolines often results in longer synthetic sequences, lower overall atom economy, and increased generation of hazardous waste streams.
The Novel Approach
In stark contrast, the technology described in CN112574198B leverages a direct oxidative coupling strategy that bypasses the need for pre-activation. By employing a catalytic amount of inexpensive copper chloride (0.2 equivalents) and pyridine as a base, the reaction proceeds smoothly in DMF solvent at room temperature. This approach eliminates the requirement for expensive ligands or noble metals, drastically reducing the raw material costs associated with catalyst loading. The use of ambient oxygen as the terminal oxidant not only enhances the environmental profile of the process but also simplifies the operational setup, as it avoids the handling of hazardous chemical oxidants. The versatility of this method is demonstrated by its tolerance to a wide range of substituents on the indole ring, including fluoro, chloro, bromo, methyl, and methoxy groups, allowing for rapid diversification of the chemical library for structure-activity relationship studies.
Mechanistic Insights into CuCl2-Catalyzed Oxidative Coupling
The core of this synthetic innovation lies in the copper-mediated activation of the C-H bonds at the reactive positions of both the tetrahydro-beta-carboline and the indole moieties. Mechanistically, the copper(II) species likely facilitates a single-electron transfer (SET) process or coordinates with the nitrogen atoms to enhance the nucleophilicity of the indole C3 position while activating the C1 position of the tetrahydro-beta-carboline framework. The presence of molecular oxygen serves to regenerate the active copper catalyst from its reduced state, closing the catalytic cycle and driving the equilibrium towards product formation. This redox-neutral pathway ensures that the reaction does not produce stoichiometric amounts of inorganic salt byproducts, which simplifies the workup procedure significantly. The mildness of the conditions preserves the stereochemical integrity of the chiral centers present in the tetrahydro-beta-carboline scaffold, which is crucial for maintaining the specific biological activity required for receptor binding in oncology applications.
From an impurity control perspective, this mechanism offers distinct advantages over radical initiators or strong Lewis acids that often lead to polymerization or non-selective substitution. The specific coordination environment provided by the pyridine base helps to moderate the reactivity of the copper center, preventing over-oxidation of the sensitive indole ring which could otherwise lead to tar formation. The resulting crude reaction mixtures are generally cleaner, facilitating easier purification via standard silica gel column chromatography using common eluent systems like petroleum ether and ethyl acetate. This predictability in impurity profiles is vital for GMP manufacturing, as it allows for the establishment of robust control strategies to ensure the final active pharmaceutical ingredient meets stringent purity specifications without requiring complex recrystallization protocols or preparative HPLC.
How to Synthesize Tetrahydro-beta-carboline Indole Derivatives Efficiently
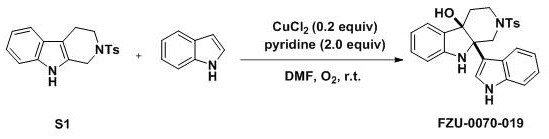
The practical implementation of this synthesis is straightforward and amenable to standard laboratory glassware, making it accessible for both small-scale discovery and larger pilot plant operations. The protocol involves dissolving the tetrahydro-beta-carboline starting material in DMF, followed by the sequential addition of the indole derivative, copper chloride catalyst, and pyridine base. The reaction is then stirred under an oxygen atmosphere at room temperature for a period ranging from 12 to 48 hours, depending on the electronic nature of the indole substituent. Upon completion, the mixture is diluted with ethyl acetate and washed with water to remove inorganic salts and polar impurities, followed by drying and concentration. The detailed standardized synthesis steps for specific analogues are outlined in the guide below.
- Dissolve the tetrahydro-beta-carboline starting material (e.g., S1) in N,N-dimethylformamide (DMF) solvent within a reaction vessel.
- Add 4.0 equivalents of the desired indole derivative, 0.2 equivalents of anhydrous copper chloride (CuCl2) catalyst, and 2.0 equivalents of pyridine base to the mixture.
- Stir the reaction mixture at room temperature under an oxygen atmosphere for 12 to 48 hours, then extract with ethyl acetate and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered on cost efficiency and operational simplicity. The elimination of precious metal catalysts removes a major volatility factor from the raw material budget, as copper salts are abundant and price-stable compared to palladium or platinum complexes. Additionally, the ability to run reactions at room temperature significantly reduces energy consumption costs associated with heating or cooling large reactor vessels, contributing to a lower carbon footprint and reduced utility overheads. The use of common solvents like DMF and ethyl acetate ensures that supply chains remain resilient, as these chemicals are commodity items with multiple global suppliers, mitigating the risk of production delays due to solvent shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the low catalyst loading and the avoidance of expensive protecting group strategies. Since the reaction tolerates various functional groups directly, synthetic steps are saved, which cumulatively lowers the cost of goods sold (COGS). The simplified workup procedure, which relies on liquid-liquid extraction rather than complex quenching or filtration of metal scavengers, further reduces labor time and consumable usage. By minimizing the number of unit operations required to isolate the pure product, manufacturers can achieve substantial cost savings in both material and processing time, enhancing the overall margin potential for the final drug substance.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions translates directly into higher reliability for supply continuity. Because the process does not require specialized high-pressure equipment or cryogenic cooling, it can be executed in a wider range of manufacturing facilities, increasing the pool of qualified contract manufacturing organizations (CMOs). The stability of the reagents allows for flexible inventory management, as there is no need for just-in-time delivery of air-sensitive or thermally unstable intermediates. This flexibility empowers supply chain planners to build strategic stockpiles of key starting materials without fear of rapid degradation, ensuring that production schedules can be maintained even during periods of logistical disruption or raw material scarcity.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to kilogram scale is facilitated by the exothermic nature of the oxidation being manageable at room temperature, reducing the risk of thermal runaway incidents. The use of oxygen as an oxidant generates water as the primary byproduct, aligning with green chemistry principles and simplifying waste treatment protocols. This environmental compatibility reduces the regulatory burden associated with hazardous waste disposal and helps companies meet increasingly strict sustainability targets. The straightforward purification via silica gel chromatography is a well-established unit operation in the industry, ensuring that scale-up does not introduce unforeseen bottlenecks in downstream processing, thus guaranteeing a smooth transition from R&D to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these tetrahydro-beta-carboline derivatives, based on the data provided in the patent literature. Understanding these specifics is crucial for evaluating the feasibility of integrating this technology into existing drug development pipelines. The answers reflect the experimental conditions and results observed during the optimization of the oxidative coupling process.
Q: What are the optimal reaction conditions for this oxidative coupling?
A: The process utilizes 0.2 equivalents of CuCl2 and 2.0 equivalents of pyridine in DMF solvent at room temperature under an oxygen atmosphere, avoiding harsh thermal conditions.
Q: What is the typical yield range for these derivatives?
A: According to the patent data, the general reaction yield is approximately 50%, with optimized examples achieving yields greater than 65% after silica gel purification.
Q: What biological activity do these compounds exhibit?
A: The synthesized indolized derivatives demonstrate significant inhibitory activity against MCF-7 (breast cancer), A549 (lung cancer), and Hela (cervical cancer) cell lines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-beta-carboline Derivative Supplier
As the demand for novel oncology therapeutics continues to rise, the ability to rapidly access high-quality intermediates is a critical competitive advantage. NINGBO INNO PHARMCHEM stands ready to support your drug discovery and development efforts with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in transition metal catalysis and heterocyclic chemistry, ensuring that we can replicate and optimize the copper-catalyzed oxidative coupling described in CN112574198B with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of tetrahydro-beta-carboline derivative meets the highest standards of quality and consistency required for clinical trials.
We invite you to collaborate with us to leverage this innovative synthetic route for your next-generation anticancer programs. Our commercial team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments for your proprietary targets. Let us be your partner in transforming promising chemical entities into life-saving medicines through efficient and scalable manufacturing solutions.
