Advanced Manufacturing of High-Optical-Purity Esomeprazole via Alkaline Oxidation Technology
Advanced Manufacturing of High-Optical-Purity Esomeprazole via Alkaline Oxidation Technology
The pharmaceutical industry continuously seeks robust methodologies for producing chiral proton pump inhibitors, specifically focusing on the efficient synthesis of Esomeprazole. Patent CN102964337A introduces a transformative production method that addresses the longstanding stability issues associated with traditional oxidation agents. By substituting the conventional acidic meta-chloroperbenzoic acid (m-CPBA) with its alkaline sodium salt counterpart, this innovation resolves the incompatibility between the oxidant and the sensitive titanium-based chiral catalyst. This technical breakthrough not only simplifies the operational workflow but also significantly enhances the optical purity of the final product while minimizing impurity profiles. For global procurement teams and R&D directors, this represents a pivotal shift towards more reliable and cost-effective manufacturing strategies for high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of single-isomer Esomeprazole from racemic Omeprazole or its sulfide precursors has been fraught with chemical challenges. Traditional approaches often relied on chiral chromatographic separation, a technique that, while effective for laboratory-scale purification, lacks the economic viability and throughput necessary for industrial-scale production. Furthermore, earlier chemical resolution methods utilizing resolving agents suffered from significant material waste, particularly the discarding of the unwanted R-isomer, leading to poor atom economy and inflated raw material costs. Another prevalent issue involved the use of acidic oxidants like m-CPBA in conjunction with titanium complexes; the inherent acidity of m-CPBA destabilizes the chiral titanium-tartrate complex, resulting in reduced enantioselectivity and the formation of undesirable by-products. These limitations collectively hindered the ability to achieve consistent high-purity outputs required for stringent regulatory compliance in the pharmaceutical sector.
The Novel Approach
The methodology disclosed in the patent circumvents these historical bottlenecks by employing sodium m-chloroperbenzoate as the oxidizing agent within a water-miscible organic solvent system. This strategic substitution creates an alkaline reaction environment that preserves the integrity of the chiral titanium catalyst throughout the oxidation process. The use of tetrahydrofuran (THF) as a co-solvent facilitates the dissolution of both organic substrates and the aqueous oxidant solution, ensuring homogeneous reaction conditions. By maintaining the stability of the chiral auxiliary, the process achieves superior stereocontrol, yielding Esomeprazole with exceptional optical purity. This approach eliminates the need for complex downstream purification steps such as chiral HPLC, thereby streamlining the production workflow and enhancing the overall feasibility of commercial scale-up for complex pharmaceutical intermediates.
Mechanistic Insights into Titanium-Catalyzed Asymmetric Oxidation
The core of this synthetic strategy lies in the formation and preservation of the active chiral catalyst species. The reaction initiates with the coordination of titanium isopropylate and D-diethyl tartrate to form a chiral titanium complex. In conventional acidic conditions, protons from the oxidant would compete for coordination sites or protonate the tartrate ligand, leading to catalyst decomposition and loss of chirality. However, the introduction of the sodium salt of the peracid ensures that the reaction medium remains sufficiently basic to prevent this degradation. The sulfide substrate coordinates to the titanium center, positioning the sulfur atom for stereoselective oxygen transfer. The precise temperature control, typically ranging from -10°C to 15°C during the oxidation phase, further suppresses non-selective background reactions, ensuring that the oxygen atom is delivered exclusively to one face of the sulfur atom.
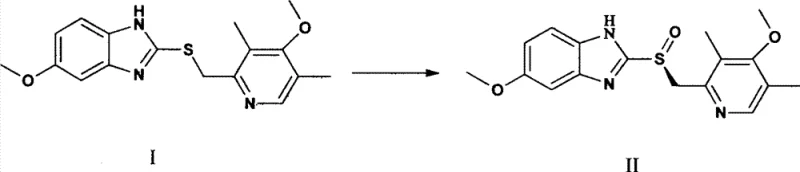
Following the oxygen transfer, the reaction mixture contains the desired sulfoxide along with reduced by-products. The quenching step using aqueous ammonia or sodium hydroxide serves a dual purpose: it neutralizes any remaining oxidant and facilitates the decomposition of the titanium complex, releasing the free Esomeprazole molecule. The subsequent extraction and crystallization steps are designed to remove inorganic salts and residual organic impurities. The use of acetone for crystallization is particularly effective in precipitating the high-purity solid while keeping soluble impurities in the mother liquor. This mechanistic understanding underscores the importance of pH control and temperature management in achieving the reported impurity levels of less than 0.3% and content purity exceeding 99%, which are critical metrics for regulatory approval and patient safety.
How to Synthesize Esomeprazole Efficiently
The practical implementation of this synthesis route requires careful attention to reagent addition rates and thermal management to maximize yield and selectivity. The process begins with the activation of the catalyst system in THF, followed by the controlled addition of the aqueous oxidant solution at low temperatures to maintain kinetic control over the reaction. Detailed standard operating procedures regarding stoichiometry, mixing speeds, and workup protocols are essential for reproducibility. For a comprehensive breakdown of the specific operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Dissolve the sulfide precursor in tetrahydrofuran (THF) and add titanium isopropylate and D-diethyl tartrate, stirring at 35-65°C to form the chiral catalyst complex.
- Cool the reaction mixture to -10 to 15°C and slowly add an aqueous solution of sodium m-chloroperbenzoate to effect asymmetric oxidation.
- Quench the reaction with aqueous ammonia or sodium hydroxide, extract with organic solvents like dichloromethane, and crystallize the product from acetone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented method offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By replacing expensive and operationally intensive chiral separation techniques with a direct asymmetric synthesis, the process drastically reduces the consumption of solvents and stationary phases. The elimination of acid-sensitive steps means that standard stainless steel reactors can be utilized without the need for specialized glass-lined equipment solely for corrosion resistance, further lowering capital expenditure. Additionally, the use of commodity chemicals like sodium m-chloroperbenzoate and titanium isopropylate ensures a stable and predictable supply of raw materials, mitigating the risks associated with sourcing specialty reagents. These factors combine to create a manufacturing process that is not only chemically robust but also economically superior for long-term production.
- Cost Reduction in Manufacturing: The transition to an alkaline oxidation system eliminates the need for costly chiral chromatography columns and the associated solvent volumes required for recycling and disposal. By avoiding the degradation of the chiral catalyst, the process achieves higher effective yields per batch, reducing the cost of goods sold (COGS). Furthermore, the simplified workup procedure involving standard liquid-liquid extraction and crystallization reduces labor hours and energy consumption compared to multi-step resolution processes. This logical deduction of cost savings stems from the fundamental simplification of the unit operations involved in the synthesis.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial chemicals such as THF, titanium isopropylate, and sodium peroxides ensures that the supply chain is not vulnerable to the bottlenecks often seen with proprietary or niche reagents. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond quickly to fluctuations in market demand. The high purity of the crude product reduces the burden on quality control laboratories, accelerating the release of batches for downstream formulation. This reliability is crucial for maintaining continuous supply lines for essential medications like proton pump inhibitors.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of multi-kilogram batches in the patent examples without loss of efficiency. The use of aqueous workups and standard organic solvents facilitates easier waste treatment and solvent recovery, aligning with modern green chemistry principles. By minimizing the generation of acidic waste streams associated with traditional m-CPBA usage, the environmental footprint of the manufacturing process is significantly reduced. This compliance with environmental standards simplifies the permitting process for new production facilities and reduces the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-optical-purity Esomeprazole production method. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, aiming to clarify the operational benefits and chemical rationale for potential partners and stakeholders.
Q: Why is sodium m-chloroperbenzoate preferred over m-CPBA for Esomeprazole synthesis?
A: Traditional m-CPBA is acidic and destabilizes the critical titanium-tartrate chiral catalyst. The sodium salt provides an alkaline environment compatible with the catalyst, ensuring high enantioselectivity.
Q: What are the primary impurities controlled in this novel oxidation method?
A: The method effectively minimizes oxidation impurities to below 0.3% and ensures high chemical content (>99%) by preventing acid-catalyzed degradation pathways common in older methods.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes readily available reagents and standard solvent systems like THF and dichloromethane, avoiding complex chromatographic separations, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Esomeprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to implement complex asymmetric oxidation technologies positions us as a strategic partner for companies seeking to optimize their API supply chains.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operation. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
