Advanced Synthesis of 5-Substituted Pyrrolo[2,3-d]pyrimidine Antifolates via Stable Sulfonic Acid Intermediates
Advanced Synthesis of 5-Substituted Pyrrolo[2,3-d]pyrimidine Antifolates via Stable Sulfonic Acid Intermediates
The pharmaceutical industry's relentless pursuit of potent antifolate agents for oncology applications has long been hindered by the synthetic challenges associated with their key intermediates. Patent CN1213998C, filed in the mid-2000s, introduces a transformative methodology that addresses the critical instability issues plaguing the production of 5-substituted pyrrolo[2,3-d]pyrimidines. This intellectual property outlines a novel series of sulfonic acid metal cation salts that serve as robust, storable precursors to the notoriously fragile alpha-halo aldehydes required for antifolate assembly. For R&D directors and process chemists, this represents a paradigm shift from handling unstable oils to managing crystalline solids, thereby enhancing the reproducibility and safety of the synthetic workflow. The invention not only secures a reliable supply chain for these high-value pharmaceutical intermediates but also offers a scalable pathway that mitigates the risks associated with traditional aldehyde chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical alpha-halo aldehyde intermediates, such as those of general formula XIV, relied on direct oxidative methods or palladium-catalyzed couplings that yielded complex mixtures. As illustrated in the prior art routes, such as the Taylor and Harrington synthesis, the generation of these aldehydes often resulted in product mixtures containing undesirable byproducts that were extremely difficult to separate. 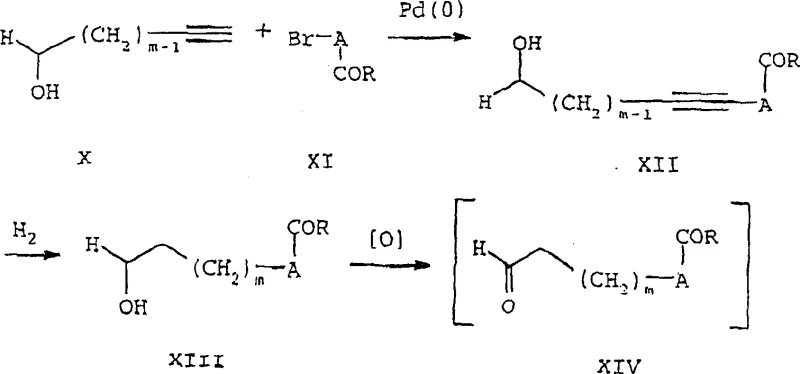 Furthermore, methods disclosed by Larock involving palladium(0) catalysis frequently produced desired products alongside significant impurities, complicating purification efforts. The inherent instability of the free aldehyde form meant that these compounds could not be easily isolated, purified, or stored in bulk, forcing manufacturers to generate them in situ and immediately consume them, which severely limits process flexibility and increases the risk of batch failure due to decomposition.
Furthermore, methods disclosed by Larock involving palladium(0) catalysis frequently produced desired products alongside significant impurities, complicating purification efforts. The inherent instability of the free aldehyde form meant that these compounds could not be easily isolated, purified, or stored in bulk, forcing manufacturers to generate them in situ and immediately consume them, which severely limits process flexibility and increases the risk of batch failure due to decomposition.
The Novel Approach
The breakthrough detailed in CN1213998C lies in the interception of the unstable aldehyde as a stable sulfonic acid metal cation salt, depicted generally as Formula IV. This approach fundamentally alters the physical state of the intermediate from a reactive, degradable liquid to a stable, crystalline solid that can be manufactured in large quantities, purified via recrystallization, and stored for extended periods without degradation. 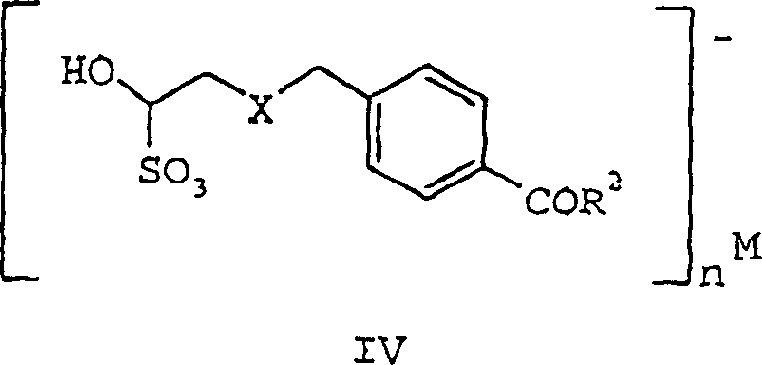 By converting the transient aldehyde into a bisulfite adduct, the process effectively "locks" the molecule in a safe configuration. This allows for rigorous quality control testing prior to the final activation step, ensuring that only high-purity material proceeds to the costly cyclization stage. This strategic modification provides a reliable pharmaceutical intermediate supplier with the ability to stockpile key precursors, decoupling the production of the aldehyde from its immediate consumption and smoothing out supply chain bottlenecks.
By converting the transient aldehyde into a bisulfite adduct, the process effectively "locks" the molecule in a safe configuration. This allows for rigorous quality control testing prior to the final activation step, ensuring that only high-purity material proceeds to the costly cyclization stage. This strategic modification provides a reliable pharmaceutical intermediate supplier with the ability to stockpile key precursors, decoupling the production of the aldehyde from its immediate consumption and smoothing out supply chain bottlenecks.
Mechanistic Insights into Palladium-Catalyzed Coupling and Sulfonation
The core of this innovative process involves a sophisticated sequence beginning with a palladium-catalyzed coupling reaction between an aryl halide and an alkenol, such as 3-buten-1-ol. Under optimized conditions utilizing catalysts like palladium(0) bis(dibenzylideneacetone) and phase-transfer catalysts, the reaction proceeds to form the initial aldehyde precursor. However, instead of isolating this sensitive species, the protocol immediately introduces a metal bisulfite reagent, such as sodium bisulfite, into the reaction mixture. This nucleophilic addition to the carbonyl group generates the stable sulfonic acid salt. The mechanism ensures that the chiral integrity of the molecule is preserved, particularly when L-glutamic acid residues are involved, as the mild conditions prevent racemization. The resulting salt precipitates from the solution, allowing for simple filtration to remove palladium residues and other organic impurities, a significant advantage over the extractive workups required for free aldehydes.
Subsequent conversion of the stable salt back to the reactive alpha-halo aldehyde is achieved through a clever desulfonation-halogenation sequence using trialkylsilyl halides, such as trimethylsilyl chloride, followed by a halogenating agent like elemental bromine. This step regenerates the aldehyde functionality while simultaneously introducing the necessary halogen at the alpha position, creating the electrophilic center required for the final ring closure. 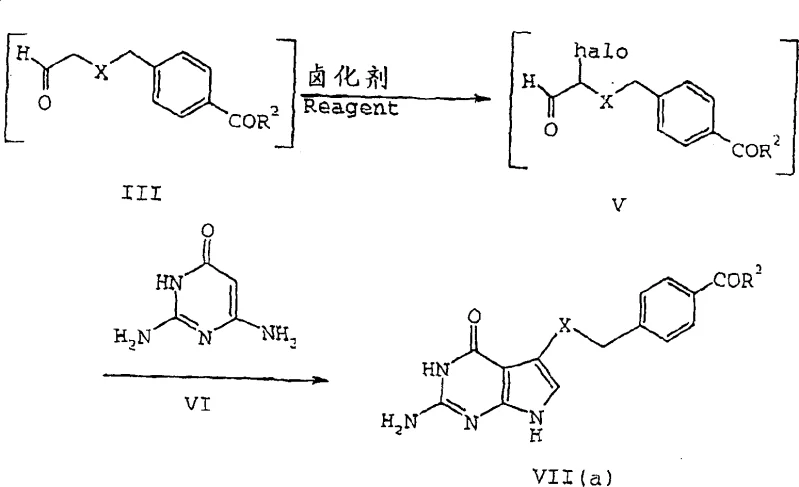 The final cyclization with 2,4-diamino-6-hydroxypyrimidine derivatives proceeds efficiently to yield the target 5-substituted pyrrolo[2,3-d]pyrimidine core. This mechanistic pathway minimizes side reactions because the reactive alpha-halo aldehyde is generated in a controlled manner and immediately consumed in the presence of the nucleophile, preventing polymerization or oxidation that typically plagues free aldehyde handling.
The final cyclization with 2,4-diamino-6-hydroxypyrimidine derivatives proceeds efficiently to yield the target 5-substituted pyrrolo[2,3-d]pyrimidine core. This mechanistic pathway minimizes side reactions because the reactive alpha-halo aldehyde is generated in a controlled manner and immediately consumed in the presence of the nucleophile, preventing polymerization or oxidation that typically plagues free aldehyde handling.
How to Synthesize Antifolate Intermediates Efficiently
The synthesis of these high-value antifolate precursors requires precise control over reaction parameters to maximize yield and purity. The process begins with the coupling of aryl halides with alkenols in polar aprotic solvents like dimethylformamide, followed by the critical stabilization step with bisulfites. Detailed operational procedures, including specific stoichiometric ratios of lithium acetate and phase-transfer catalysts, are essential for reproducibility. The subsequent transformation of the stable salt into the reactive aldehyde and its final cyclization demands careful temperature management, typically ranging from ambient to 70°C, to ensure complete conversion without degradation. For a comprehensive guide on executing this chemistry, please refer to the standardized synthesis steps provided below.
- Perform palladium-catalyzed coupling of aryl halides with alkenols to generate aldehyde precursors, followed by immediate stabilization with metal bisulfites to form crystalline sulfonic acid salts.
- Convert the stable sulfonic acid salt intermediates into reactive alpha-halo aldehydes using trialkylsilyl halides and halogenating agents under controlled thermal conditions.
- Execute the final cyclization reaction between the alpha-halo aldehyde and 2,4-diamino-6-hydroxypyrimidine derivatives to yield the target 5-substituted pyrrolo[2,3-d]pyrimidine antifolate core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sulfonic acid salt methodology translates into tangible operational efficiencies and risk mitigation. The ability to isolate stable, crystalline intermediates drastically simplifies inventory management, as these materials do not require the specialized cold-chain logistics or immediate usage protocols associated with unstable aldehydes. This stability allows for the aggregation of production batches, enabling manufacturers to produce larger volumes less frequently, which optimizes reactor utilization and reduces overall manufacturing overhead. Furthermore, the simplified purification process, which relies on crystallization rather than complex chromatography, significantly lowers the cost of goods sold by reducing solvent consumption and waste disposal costs.
- Cost Reduction in Manufacturing: The elimination of difficult purification steps for unstable oils leads to substantial cost savings. By avoiding the loss of material associated with decomposing intermediates and reducing the need for expensive chromatographic resins, the overall production cost is significantly lowered. The process utilizes readily available reagents like sodium bisulfite and common solvents, avoiding the need for exotic or prohibitively expensive catalysts that drive up the price of traditional routes.
- Enhanced Supply Chain Reliability: The stability of the sulfonic acid salt intermediates ensures a consistent supply of high-quality materials. Unlike free aldehydes which may degrade during transit or storage, these crystalline salts maintain their integrity over time, reducing the risk of supply disruptions caused by spoiled batches. This reliability allows for longer lead times in planning and reduces the pressure on just-in-time manufacturing schedules, providing a buffer against market volatility.
- Scalability and Environmental Compliance: The process is inherently scalable due to the robust nature of the intermediates and the simplicity of the workup procedures. The reduction in solvent usage and the avoidance of hazardous purification techniques align with green chemistry principles, facilitating easier regulatory approval and environmental compliance. The ability to filter and wash solid intermediates rather than performing multiple liquid-liquid extractions reduces the volume of aqueous waste generated, streamlining waste treatment processes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel synthetic route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical application of the technology for industrial scale-up.
Q: Why are sulfonic acid metal cation salts preferred over free aldehydes for antifolate synthesis?
A: Free alpha-halo aldehydes are inherently unstable and difficult to purify, often decomposing before use. The sulfonic acid metal cation salts described in CN1213998C are crystalline, stable solids that can be stored and transported safely, only converting to the reactive aldehyde form when needed.
Q: What are the key advantages of this process for large-scale manufacturing?
A: The process eliminates the need for difficult chromatographic separations of unstable oils. By isolating stable crystalline intermediates, manufacturers can achieve higher purity through simple filtration and recrystallization, significantly reducing waste and processing time.
Q: Can this methodology be adapted for different R-group substitutions?
A: Yes, the methodology is highly versatile. The patent demonstrates applicability across various carboxyl-protecting groups (methyl, ethyl) and linker lengths, making it suitable for synthesizing a broad library of 5-substituted pyrrolo[2,3-d]pyrimidine analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Antifolate Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving oncology therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of antifolate intermediate meets the highest global standards, providing our partners with the confidence needed to advance their clinical programs.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your production volume and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in high-purity pharmaceutical intermediates can accelerate your drug development timeline.
