Scalable Synthesis of Polyenoid Dialdehyde Monoacetals for High-Value Carotenoid Production
The pharmaceutical and fine chemical industries constantly seek robust methodologies for constructing complex polyene chains, which serve as the backbone for high-value carotenoids such as capsanthin and cryptocapsin. Patent CN1197837C, filed in 2005, introduces a sophisticated and technically scalable process for preparing polyenoid dialdehyde monoacetals, specifically targeting C15 and C20 structural units. This innovation addresses critical bottlenecks in the synthesis of unsymmetrical carotenoids by providing a reliable pathway to protected dialdehyde intermediates that were previously difficult to access with high purity. The core of this technology lies in the strategic manipulation of acetal protecting groups combined with efficient carbon-chain elongation techniques. 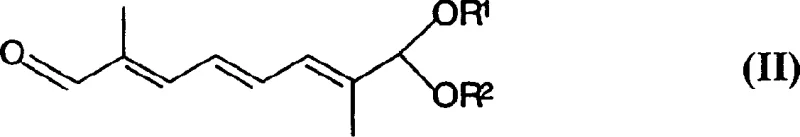 As illustrated by the starting material structure, the process begins with a protected polyenoid dialdehyde, setting the stage for a highly controlled sequence of Wittig condensations and functional group transformations that ensure the integrity of the sensitive conjugated double bond system throughout the synthesis.
As illustrated by the starting material structure, the process begins with a protected polyenoid dialdehyde, setting the stage for a highly controlled sequence of Wittig condensations and functional group transformations that ensure the integrity of the sensitive conjugated double bond system throughout the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of unsymmetrical C40 carotenoids has been plagued by significant technical hurdles, particularly regarding the availability and stability of the requisite building blocks. Prior art, such as DE-A2851051, described methods relying on asymmetric C10 dialdehydes which are not commonly available synthons on a technical scale, leading to supply chain fragility and high raw material costs. Furthermore, alternative approaches involving the partial hydrolysis of diacetals, as noted in synthetic literature from the early 1990s, often suffered from inadequate selectivity. This lack of selectivity necessitated rigorous and costly chromatographic purification steps to isolate the desired monoacetal from a mixture of diacetal and dialdehyde byproducts. Such purification requirements are economically prohibitive for commercial-scale manufacturing, as they drastically reduce overall throughput and increase solvent consumption, rendering these older methods unsuitable for the production of bulk fine chemical intermediates required by the global nutraceutical and pigment markets.
The Novel Approach
The methodology disclosed in CN1197837C represents a paradigm shift by decoupling the chain extension steps from the protection group strategy, thereby enhancing overall process control. Instead of relying on difficult-to-source asymmetric dialdehydes, this novel approach utilizes a modular construction where a C15 structural unit is first established and then optionally extended to a C20 unit through a sequential Wittig or Wittig-Horner reaction. A key breakthrough is the implementation of specific hydrolysis conditions, utilizing citric acid catalysis, which allows for the selective deprotection of the acetal function under mild conditions. This selectivity minimizes the formation of side products and significantly reduces the burden on downstream purification processes. By converting the intermediate ester directly into the final aldehyde through a two-stage reduction-oxidation sequence, the process avoids the instability associated with handling free polyenoid aldehydes for extended periods, ensuring higher yields and a more consistent impurity profile suitable for GMP-compliant manufacturing environments.
Mechanistic Insights into Wittig-Horner Condensation and Catalytic Oxidation
The chemical elegance of this process is rooted in its precise control over carbon-carbon bond formation and functional group interconversion. The initial chain elongation relies on the reaction of the Formula II aldehyde with phosphonium salts or phosphonates (Formula III) under standard Wittig or Wittig-Horner conditions. The choice of base is critical; the patent highlights the use of alkali metal alcoholates or organolithium compounds in inert solvents like toluene or THF to generate the necessary ylide or anion species. This step constructs the extended polyene backbone while maintaining the stereochemistry of the double bonds, which is essential for the biological activity and color properties of the final carotenoid product. Following chain extension, the ester functionality is preserved, acting as a stable masked aldehyde that protects the sensitive terminal position during subsequent synthetic manipulations, thereby preventing premature polymerization or degradation of the conjugated system.
The transformation of the terminal ester into the final aldehyde functionality is achieved through a sophisticated two-step sequence that prioritizes safety and environmental sustainability. First, the ester is reduced to the corresponding allylic alcohol (Formula VII) using hydride reagents. The patent specifically advocates for the use of sodium bis(2-methoxyethoxy)aluminum hydride (Vitride) in toluene. Unlike pyrophoric solid lithium aluminum hydride, Vitride is a liquid solution that is insensitive to oxygen and easier to handle on a multi-kilogram scale, significantly mitigating safety risks in plant operations. Subsequently, the alcohol is oxidized to the aldehyde (Formula I) using a catalytic system. 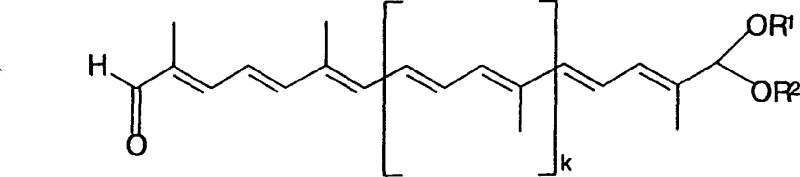 Rather than employing stoichiometric amounts of toxic chromium or manganese oxidants, the process utilizes a catalytic amount of copper(I) chloride and TEMPO radicals with molecular oxygen as the terminal oxidant. This green chemistry approach not only reduces heavy metal waste but also simplifies the workup procedure, as the catalyst loading is minimal (2-4 mol%), aligning perfectly with modern regulatory demands for low residual metal content in pharmaceutical intermediates.
Rather than employing stoichiometric amounts of toxic chromium or manganese oxidants, the process utilizes a catalytic amount of copper(I) chloride and TEMPO radicals with molecular oxygen as the terminal oxidant. This green chemistry approach not only reduces heavy metal waste but also simplifies the workup procedure, as the catalyst loading is minimal (2-4 mol%), aligning perfectly with modern regulatory demands for low residual metal content in pharmaceutical intermediates.
How to Synthesize Polyenoid Dialdehyde Monoacetals Efficiently
The synthesis of these complex intermediates requires strict adherence to the reaction parameters outlined in the patent to ensure optimal yield and purity. The process involves a sequence of condensation, optional hydrolysis, reduction, and oxidation steps, each demanding precise temperature control and reagent stoichiometry. For R&D teams looking to replicate or adapt this chemistry, it is crucial to note the specific advantages of using Vitride for the reduction step and the Cu/TEMPO system for oxidation, as these choices define the scalability of the route. The detailed standardized synthesis steps, including exact molar ratios, solvent volumes, and temperature profiles for each transformation, are provided in the guide below to facilitate immediate technology transfer and pilot plant trials.
- Perform Wittig or Wittig-Horner reaction on Formula II compound with Formula III reagent to yield Formula IV.
- Optionally hydrolyze the acetal group and react with Formula V reagent to extend the chain to Formula VI.
- Reduce the ester group to alcohol (Formula VII) using Vitride, followed by catalytic oxidation to the final aldehyde (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1197837C offers substantial strategic benefits beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly translates to improved cost structures and supply reliability. By eliminating the need for complex chromatographic purifications that were characteristic of older monoacetal synthesis methods, the process significantly reduces solvent usage and processing time. This efficiency gain allows for larger batch sizes and shorter campaign cycles, enabling suppliers to respond more agilely to market demand fluctuations. Furthermore, the use of stable ester intermediates rather than reactive aldehydes for storage and transport minimizes the risk of material degradation during logistics, ensuring that the quality of the raw material remains intact from the manufacturer to the customer's production facility.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive and hazardous reagents with more economical and safer alternatives. The substitution of stoichiometric heavy metal oxidants with a catalytic copper/oxygen system removes the cost associated with purchasing large quantities of oxidizing agents and, more importantly, the downstream costs of treating and disposing of toxic heavy metal waste. Additionally, the use of Vitride, while a specialized reagent, offers operational savings through improved safety profiles and reduced need for specialized containment equipment required for pyrophoric solids. The overall reduction in purification steps means lower energy consumption for solvent recovery and distillation, contributing to a leaner and more cost-effective manufacturing operation that enhances margin potential for high-volume carotenoid production.
- Enhanced Supply Chain Reliability: From a sourcing perspective, this methodology relies on readily available starting materials and reagents that are produced by multiple global chemical manufacturers, reducing the risk of single-source dependency. The robustness of the acetal protecting group strategy ensures that the intermediates have a longer shelf-life and are less susceptible to environmental factors such as humidity and light compared to unprotected polyenoid aldehydes. This stability allows for the maintenance of strategic inventory buffers without significant loss of potency, thereby securing the supply chain against unexpected disruptions. The scalability of the Wittig-Horner reaction, a well-understood industrial process, further guarantees that production can be ramped up from pilot scale to multi-ton commercial quantities without encountering unforeseen engineering bottlenecks.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than traditional routes, aligning with the increasingly stringent ESG (Environmental, Social, and Governance) criteria imposed by multinational corporations. The catalytic oxidation step generates water as the primary byproduct rather than heavy metal sludge, simplifying wastewater treatment and reducing the regulatory burden on the manufacturing site. The process is designed to be executed in common organic solvents like toluene and ethanol, which are easily recycled and managed within standard solvent recovery systems. This compatibility with existing infrastructure facilitates rapid technology transfer to contract manufacturing organizations (CMOs) and ensures that the production of these high-value intermediates meets the rigorous environmental standards required for supplying the global pharmaceutical and food additive markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within CN1197837C, providing clarity on the specific advantages of the catalytic oxidation system and the handling of sensitive polyenoid structures. Understanding these nuances is essential for R&D directors evaluating the feasibility of integrating this chemistry into their existing pipelines for carotenoid and vitamin synthesis.
Q: What are the advantages of using Vitride over Lithium Aluminum Hydride in this process?
A: Vitride (sodium bis(2-methoxyethoxy)aluminum hydride) is supplied as a toluene solution, making it non-pyrophoric and significantly safer to handle on a technical scale compared to solid LiAlH4, while maintaining high reduction efficiency for polyenoid esters.
Q: How does this patent address the issue of selectivity in monoacetal formation?
A: The process utilizes specific hydrolysis conditions, such as citric acid catalysis in aqueous ethanol, to selectively deprotect one acetal group without affecting the other, avoiding the need for complex chromatographic purification often required in older methods.
Q: Is the oxidation step suitable for large-scale manufacturing?
A: Yes, the patent prefers a catalytic oxidation system using copper(I) chloride and TEMPO radicals with molecular oxygen, which eliminates the need for stoichiometric amounts of expensive and toxic heavy metal oxidants, enhancing environmental compliance and cost-efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyenoid Dialdehyde Monoacetal Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex academic and patented chemistry into commercial reality. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of reaction conditions required for polyenoid synthesis is maintained even at the largest scales. We understand that the integrity of the conjugated double bond system is paramount, and our stringent purity specifications and rigorous QC labs are equipped to monitor and control trace impurities that could affect the color or stability of the final carotenoid product. Our commitment to quality assurance ensures that every batch of intermediate meets the exacting standards required for downstream synthesis of high-value active ingredients.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced synthesis technology for your specific applications. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how implementing this catalytic route can optimize your specific bill of materials. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project timelines. Let us demonstrate how our expertise in fine chemical intermediates can drive efficiency and reliability in your supply chain.
