Scalable Synthesis of Monoterpene Indole Alkaloids for Advanced Oncology Applications
Introduction to Novel Monoterpene Indole Alkaloid Scaffolds
The pharmaceutical industry is constantly seeking novel scaffolds that offer high therapeutic indices with manageable synthesis costs. Patent CN107400128B discloses a significant advancement in the field of oncology intermediates by presenting a series of pseudomonoterpene indole alkaloids characterized by the general structural formula (I). These compounds are designed to leverage the inherent biological activity of natural product frameworks while allowing for extensive structural diversification through semi-synthetic modification. The core innovation lies in the strategic coupling of a genipin-derived iridoid skeleton with various nitrogen-containing heterocycles, specifically indole derivatives. This approach not only mimics biosynthetic pathways found in nature but also provides a robust platform for generating libraries of compounds with potential antitumor properties. The structural versatility allows for substitutions at multiple positions, enabling fine-tuning of pharmacokinetic and pharmacodynamic profiles.
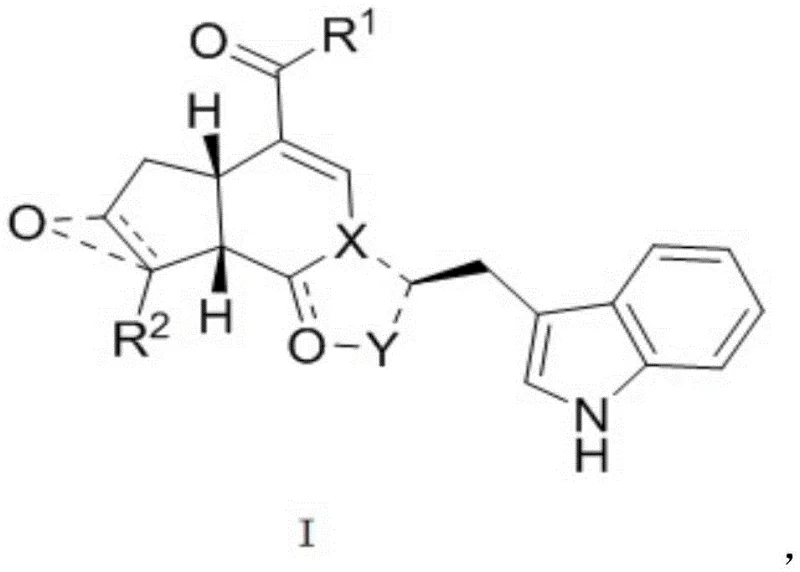
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of complex monoterpene indole alkaloids has been hindered by the reliance on total synthesis or extraction from natural sources. Total synthesis of these intricate polycyclic systems often demands numerous steps, utilizing expensive chiral catalysts and protecting group strategies that drastically reduce overall atom economy. Furthermore, many classical routes require extreme reaction conditions, such as high temperatures, high pressures, or the use of strong acids and bases, which pose significant safety risks and environmental burdens during scale-up. Extraction from plant materials, while seemingly direct, suffers from low yields, seasonal variability, and the difficult separation of closely related analogues, making it unsuitable for consistent commercial supply. These factors collectively create a bottleneck in the development of new antitumor agents based on this privileged scaffold.
The Novel Approach
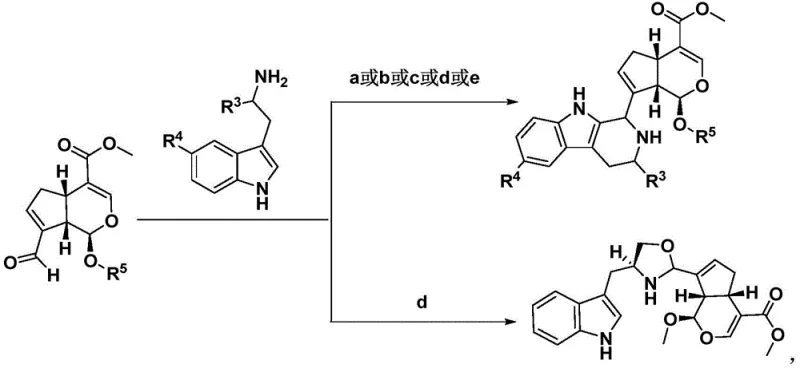
The methodology outlined in the patent overcomes these hurdles by employing a semi-synthetic strategy centered on genipin, a readily available hydrolysis product of geniposide from Gardenia jasminoides. This route bypasses the need for constructing the core cyclopenta[c]pyran skeleton from scratch, as it is already present in the starting material. The synthesis proceeds through mild condensation reactions, often catalyzed by trifluoroacetic acid or acetic acid in common organic solvents like dichloromethane or ethanol. By avoiding extreme conditions and utilizing simple, commercially available amine coupling partners such as tryptamine and its derivatives, the process achieves high operational simplicity. This shift from complex total synthesis to modular assembly allows for the rapid generation of diverse analogues, facilitating structure-activity relationship studies essential for drug discovery.
Mechanistic Insights into Genipin-Amine Condensation
The core chemical transformation driving this technology is a biomimetic condensation reaction, functionally similar to a Pictet-Spengler or Mannich-type cyclization. The process typically begins with the activation of the genipin scaffold, often involving the oxidation of a hydroxymethyl group to an aldehyde using reagents like activated manganese dioxide or Dess-Martin periodinane. This electrophilic aldehyde then reacts with the nucleophilic amine group of the indole derivative. Under acidic conditions, this forms an iminium ion intermediate which subsequently undergoes intramolecular cyclization or further nucleophilic attack to establish the rigid polycyclic framework. The stereochemistry is largely controlled by the existing chiral centers in the genipin molecule, ensuring high diastereoselectivity without the need for external chiral auxiliaries. This mechanistic efficiency is crucial for maintaining high purity standards required for pharmaceutical intermediates.
Impurity control in this synthesis is managed through the selection of specific reaction conditions and purification techniques. For instance, the use of mild reducing agents like sodium cyanoborohydride allows for the selective reduction of imine bonds without affecting other sensitive functional groups such as esters or double bonds within the iridoid ring. The patent details various workup procedures, including pH adjustments and solvent extractions, designed to remove unreacted amines and acidic byproducts effectively. Additionally, the modular nature of the synthesis means that impurities are often structurally distinct from the final product, facilitating their removal via standard silica gel column chromatography or preparative thin-layer chromatography. This level of control ensures that the final alkaloid derivatives meet the stringent quality specifications necessary for downstream biological screening and potential clinical development.
How to Synthesize Monoterpene Indole Alkaloids Efficiently
The synthesis of these high-value intermediates follows a logical sequence of oxidation, condensation, and functionalization steps that can be adapted for various substituents. The process begins with the preparation of the reactive genipin aldehyde, followed by coupling with the chosen indole amine under controlled acidic conditions. Subsequent modifications, such as acylation or reduction, allow for the fine-tuning of the molecular properties. The detailed standardized synthesis steps for specific analogues are provided in the guide below, ensuring reproducibility and consistency across batches.
- Oxidation of geniposide or genipin derivatives using activated manganese dioxide or Dess-Martin periodinane to generate reactive aldehyde intermediates.
- Condensation of the aldehyde intermediate with tryptamine or substituted indole amines under acidic conditions (e.g., TFA, AcOH) to form the core alkaloid skeleton.
- Post-synthetic modification including reduction with sodium cyanoborohydride or acylation to introduce diverse side chains for structure-activity optimization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial benefits by decoupling production from the volatility of natural extraction. Since the starting material, genipin, is derived from a widely cultivated medicinal plant, the supply chain is robust and less susceptible to the geopolitical or climatic risks associated with rare natural sources. The synthetic route utilizes commodity chemicals and solvents, such as ethanol, dichloromethane, and acetic acid, which are readily available in bulk quantities globally. This accessibility translates directly into cost stability and reduces the risk of production delays caused by specialized reagent shortages. Furthermore, the simplicity of the reaction conditions means that manufacturing can be performed in standard multipurpose reactors without the need for specialized high-pressure or corrosion-resistant equipment.
- Cost Reduction in Manufacturing: The elimination of complex asymmetric synthesis steps and expensive transition metal catalysts significantly lowers the bill of materials. By leveraging the chirality inherent in the natural genipin scaffold, the process avoids the costs associated with chiral resolution or enantioselective catalysis. Additionally, the mild reaction conditions reduce energy consumption, as there is no need for prolonged heating or cryogenic cooling. The high atom economy of the condensation reactions minimizes waste generation, thereby lowering disposal costs and improving the overall environmental footprint of the manufacturing process.
- Enhanced Supply Chain Reliability: The modular synthesis allows for the stocking of key intermediates, such as the oxidized genipin derivative, which can be rapidly converted into various final products based on demand. This flexibility enables a just-in-time manufacturing model, reducing inventory holding costs while ensuring quick turnaround times for custom orders. The use of stable, non-hazardous reagents simplifies logistics and storage requirements, further enhancing the reliability of the supply chain. Suppliers can confidently commit to long-term contracts knowing that the raw material base is secure and the synthesis pathway is resilient to minor fluctuations in reagent availability.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in batch sizes ranging from grams to potentially multi-kilogram scales without fundamental changes to the chemistry. The absence of heavy metals and the use of relatively benign solvents facilitate easier regulatory approval and compliance with increasingly strict environmental regulations. Waste streams are simpler to treat, and the potential for solvent recovery is high due to the use of common volatile organic compounds. This alignment with green chemistry principles not only reduces operational costs but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these monoterpene indole alkaloids. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners.
Q: What are the key advantages of using genipin as a starting material?
A: Genipin is a naturally occurring iridoid derived from Gardenia jasminoides, offering a chiral, renewable, and cost-effective scaffold that eliminates the need for complex asymmetric synthesis steps typically required in total synthesis.
Q: How does this method improve upon traditional total synthesis of indole alkaloids?
A: Unlike traditional total synthesis which often involves harsh conditions and low overall yields, this biomimetic approach utilizes mild reaction conditions (room temperature to 60°C) and avoids extreme pH or high pressure, significantly simplifying the operational complexity.
Q: What is the biological significance of the synthesized compounds?
A: The synthesized monoterpene indole alkaloids demonstrate potent in vitro inhibition against multiple human tumor cell lines including HL-60, A-549, and MCF-7, validating their potential as lead compounds for novel antitumor drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monoterpene Indole Alkaloids Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex academic research into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory curiosity to market-ready product. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of monoterpene indole alkaloids meets the highest international standards. We understand the critical nature of oncology intermediates and are committed to providing a supply chain that is both resilient and responsive to your evolving needs.
We invite you to collaborate with us to unlock the full potential of this innovative chemical space. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. Please contact our technical procurement team today to request specific COA data for our existing library or to discuss route feasibility assessments for your proprietary analogues. Together, we can accelerate the development of next-generation antitumor therapies.
