Advanced Manufacturing of 4-(Dimethoxymethyl)-Piperidine for Global Pharmaceutical Supply Chains
Advanced Manufacturing of 4-(Dimethoxymethyl)-Piperidine for Global Pharmaceutical Supply Chains
The pharmaceutical industry constantly demands robust, scalable, and high-purity intermediates to support the production of complex active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN112661694B, which discloses an innovative preparation method for 4-(dimethoxymethyl)-piperidine, a critical building block in medicinal chemistry. This compound, characterized by its unique piperidine scaffold protected by a dimethoxymethyl group, serves as a versatile precursor for synthesizing various bioactive molecules, including those with anti-inflammatory, antibacterial, and antitumor properties. The patent outlines a streamlined two-step synthetic route that addresses historical challenges regarding yield, purity, and environmental impact. By leveraging specific acid-catalyzed acetalization followed by efficient noble metal-catalyzed hydrogenation, this technology offers a compelling solution for manufacturers seeking to optimize their supply chains for high-value heterocyclic intermediates.
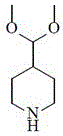
The structural integrity and chemical versatility of 4-(dimethoxymethyl)-piperidine make it an indispensable asset in the synthesis of marketed drugs such as donepezil, risperidone, and naratriptan. The ability to produce this intermediate with exceptional consistency is paramount for R&D directors who require reliable materials for process development. The method described ensures that the final product is a colorless to pale yellow transparent liquid with a molecular weight of 159.226, meeting stringent quality specifications required for downstream coupling reactions. As we delve deeper into the technical specifics, it becomes evident that this patent represents not just a laboratory curiosity, but a viable industrial protocol capable of supporting large-scale commercial production while adhering to modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of piperidine derivatives and their protected analogues has been fraught with inefficiencies that hinder large-scale adoption. Conventional literature often describes acetalization reactions of aromatic aldehydes that suffer from inconsistent yields and a lack of comprehensive data regarding substrate scope. For instance, prior studies on the reaction of aromatic aldehydes with methylating agents have indicated that electron-withdrawing groups facilitate the reaction, whereas electron-donating groups pose significant kinetic barriers, often leading to incomplete conversions. Furthermore, many traditional protocols fail to report critical metrics such as final product yield or purity, leaving manufacturers in the dark regarding the true economic feasibility of these routes. The reliance on harsh reaction conditions or stoichiometric reagents that cannot be recovered further exacerbates the problem, resulting in excessive waste generation and inflated production costs. These limitations create a bottleneck for procurement managers who struggle to source high-quality intermediates at competitive prices, forcing them to rely on fragmented supply chains with variable quality control standards.
The Novel Approach
In stark contrast to these legacy methods, the novel approach presented in patent CN112661694B introduces a highly optimized pathway that maximizes efficiency and minimizes waste. The process begins with the reaction of 4-pyridine formaldehyde and a methylating agent under the influence of a protonic or solid acid catalyst, operating at mild temperatures between 20 and 80°C. This initial step achieves a raw material conversion rate of greater than or equal to 99.5%, effectively eliminating the issue of unreacted starting materials that plague older techniques. The subsequent hydrogenation step utilizes a noble metal supported catalyst under controlled hydrogen pressure, ensuring the selective reduction of the pyridine ring to the piperidine structure without compromising the acetal protecting group. This strategic sequence allows for a final yield of greater than or equal to 96% and a purity exceeding 99%, setting a new benchmark for quality in this chemical class. By integrating catalyst recovery and solvent recycling directly into the workflow, this method transforms the production landscape, offering a sustainable and economically superior alternative for the global market.
Mechanistic Insights into Acid-Catalyzed Acetalization and Catalytic Hydrogenation
The core of this technological advancement lies in the precise orchestration of two distinct catalytic cycles. The first stage involves the acid-catalyzed protection of the aldehyde functionality. Whether utilizing a protonic acid like concentrated sulfuric acid or a solid acid such as phosphotungstic acid, the mechanism facilitates the nucleophilic attack of the methoxy species onto the carbonyl carbon of the 4-pyridinecarboxaldehyde. This reaction is carefully tuned to proceed at temperatures ranging from 20 to 80°C over a period of 4 to 12 hours, ensuring complete conversion to the 4-(dimethoxymethyl)-pyridine intermediate. The choice of catalyst is critical; solid acids offer the advantage of easier separation, while protonic acids provide rapid kinetics. The use of methylating agents such as trimethyl orthoformate or dimethyl carbonate ensures that the equilibrium is driven towards the acetal product, preventing the formation of hemiacetals or other unstable byproducts that could complicate downstream purification.
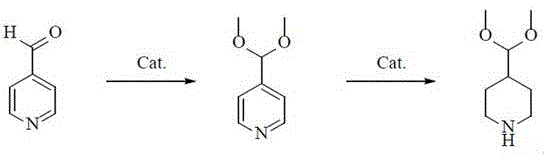
Following the acetalization, the second stage employs a heterogeneous hydrogenation mechanism to reduce the aromatic pyridine ring to the saturated piperidine ring. This step utilizes noble metals such as ruthenium or rhodium supported on carriers like activated carbon, silica, or titania. The reaction is conducted under a hydrogen pressure of 2 to 4 MPa and temperatures between 40 and 100°C. The supported nature of the catalyst is crucial for impurity control, as it allows for the physical removal of the metal via simple filtration post-reaction, thereby preventing metal contamination in the final API. The mild conditions prevent the hydrolysis of the dimethoxymethyl group, preserving the integrity of the protecting group throughout the reduction. This mechanistic precision ensures that the final product, 4-(dimethoxymethyl)-piperidine, is obtained with minimal side reactions, such as ring opening or over-reduction, which are common pitfalls in less optimized hydrogenation processes. The result is a clean reaction profile that simplifies the workup and enhances the overall safety of the manufacturing process.
How to Synthesize 4-(Dimethoxymethyl)-Piperidine Efficiently
Implementing this synthesis route requires careful attention to reaction parameters and material ratios to replicate the high success rates reported in the patent. The process is designed to be robust, accommodating variations in scale while maintaining consistent quality. Operators must ensure that the molar ratio of the aldehyde to the methylating agent is maintained between 1:2 and 1:4 to drive the acetalization to completion. Similarly, the loading of the noble metal catalyst in the second step should be optimized between 0.05 and 0.15 mass ratio relative to the substrate to balance reaction speed with cost efficiency. The detailed standardized synthesis steps, including specific solvent choices like toluene or methanol and precise distillation conditions under reduced pressure, are outlined below to guide technical teams in adopting this superior methodology.
- React 4-pyridinecarboxaldehyde with a methylating agent using a protonic or solid acid catalyst at 20-80°C to form 4-(dimethoxymethyl)-pyridine.
- Dissolve the intermediate in an organic solvent and add a noble metal supported catalyst (Ru or Rh).
- Perform hydrogenation at 2-4MPa and 40-100°C, then filter and distill to obtain the final high-purity piperidine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented method translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the purification process. Because the reaction achieves such high conversion rates and selectivity, the need for extensive chromatographic purification is eliminated, replaced by straightforward filtration and distillation. This reduction in processing steps directly correlates to lower energy consumption and reduced labor costs, making the final product more price-competitive in the global marketplace. Furthermore, the ability to recycle both the noble metal catalyst and the organic solvents creates a closed-loop system that significantly mitigates the volatility of raw material pricing. By recovering expensive ruthenium or rhodium catalysts, manufacturers can insulate themselves from fluctuations in precious metal markets, ensuring stable long-term pricing for their customers.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally sound due to the elimination of wasteful stoichiometric reagents and the implementation of catalytic cycles. The high atom economy of the acetalization step, combined with the recoverability of the hydrogenation catalyst, means that the cost per kilogram of the final intermediate is substantially lower than traditional methods. Additionally, the mild reaction conditions reduce the thermal load on manufacturing equipment, leading to lower utility costs and extended equipment lifespan. The avoidance of complex workup procedures further reduces the consumption of auxiliary chemicals and disposal fees associated with hazardous waste, contributing to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the scarcity of specialized reagents or the complexity of synthesis routes. This method utilizes readily available starting materials like 4-pyridinecarboxaldehyde and common methylating agents, reducing the risk of supply disruptions. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures without significant loss in yield, ensures that production schedules can be met consistently even under varying operational circumstances. This reliability is crucial for pharmaceutical companies that require just-in-time delivery of intermediates to maintain their own API production timelines, thereby strengthening the partnership between suppliers and end-users.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this process offers a clear pathway to compliance. The reduction in three wastes (waste water, waste gas, and solid waste) through solvent recycling and catalyst recovery aligns with modern green chemistry mandates. The scalability of the hydrogenation step, which can be safely performed in standard pressure reactors, allows for seamless transition from pilot plant to full commercial scale without the need for exotic equipment. This ease of scale-up ensures that suppliers can rapidly respond to increased market demand, providing a secure source of high-purity intermediates for the growing pharmaceutical sector.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its potential for integration into their supply chains. The following questions address common concerns regarding quality, safety, and operational feasibility, drawing directly from the data provided in the patent documentation. These insights are intended to clarify the advantages of this route over conventional alternatives and to demonstrate its readiness for industrial application.
Q: What is the purity level achievable with this new synthesis method?
A: According to patent CN112661694B, the described method achieves a product purity of greater than or equal to 99% as determined by gas chromatography, ensuring suitability for sensitive pharmaceutical applications.
Q: Can the catalysts used in this process be recycled?
A: Yes, the process utilizes noble metal supported catalysts which are recovered via filtration after the reaction. Both the catalyst residues and the distilled solvents can be recycled, significantly reducing material costs and environmental waste.
Q: What are the typical reaction conditions for the hydrogenation step?
A: The hydrogenation step is conducted under mild to moderate conditions, specifically at temperatures between 40-100°C and hydrogen pressures ranging from 2 to 4 MPa, allowing for safe and scalable industrial operation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(Dimethoxymethyl)-Piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team of expert chemists has thoroughly analyzed the technology disclosed in patent CN112661694B and is fully prepared to leverage this advanced synthesis route for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4-(dimethoxymethyl)-piperidine meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive your projects forward with confidence and reliability.
