Advanced Catalytic Hydrogenation Strategy for High-Purity (S)-3-Hydroxy-Gamma-Butyrolactone Production
Introduction to Novel Catalytic Hydrogenation Technology
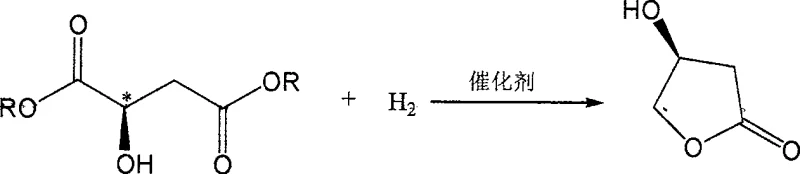
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective routes for producing chiral building blocks, particularly those serving as key intermediates for high-value active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is documented in Chinese Patent CN100503590C, which discloses a sophisticated method for synthesizing (S)-3-hydroxy-gamma-butyrolactone through the catalytic hydrogenation of L-malic acid diester. This specific chiral lactone is a pivotal precursor in the synthesis of lipid-lowering agents such as Lipitor (Atorvastatin) intermediates, neuroregulators like (R)-GABOB, and various other therapeutic compounds including AIDS treatments. The innovation lies not merely in the transformation itself but in the engineering of a unique homogeneous-heterogeneous composite catalyst system that bridges the gap between the high selectivity of homogeneous catalysis and the ease of separation associated with heterogeneous systems.
Traditionally, the production of such chiral lactones has been plagued by harsh reaction conditions, expensive stoichiometric reagents, and complex purification workflows that generate substantial chemical waste. The technology described in CN100503590C addresses these pain points by utilizing a rhodium complex anchored on metal-loaded silica supports. This approach enables the reaction to proceed at comparatively lower pressures and temperatures while achieving exceptional yields and optical purity. For R&D directors and process chemists evaluating potential manufacturing routes, this patent represents a compelling alternative to legacy methods, offering a pathway to greener chemistry without compromising on the stringent quality specifications required for pharmaceutical grade intermediates. The ability to tune the catalyst composition further enhances its versatility across different production scales.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (S)-3-hydroxy-gamma-butyrolactone has relied heavily on reduction strategies employing borane or metal borohydrides, as seen in earlier patents like US5808107. While effective in a laboratory setting, these stoichiometric reductions present severe drawbacks when translated to commercial manufacturing. The primary issue is the generation of large quantities of boron-containing waste, which necessitates complex and costly disposal procedures to meet environmental regulations. Furthermore, the purification of the final product from boron residues is often difficult, leading to potential contamination issues that can compromise the quality of downstream APIs. Another conventional route involves the oxidation of carbohydrates or sugar derivatives; however, these reactions typically occur in aqueous media where the reaction mixture becomes exceedingly complex, making the isolation of the target butyrolactone technically challenging and economically inefficient due to low recovery rates.
Even more recent attempts to improve this synthesis, such as those utilizing ruthenium heterogeneous catalysts in flow reactors, have required extreme operating conditions. For instance, some prior art methods necessitate pressures exceeding 230 atmospheres and temperatures above 120°C to achieve acceptable conversion. Such high-pressure requirements impose significant capital expenditure on reactor design and safety systems, increasing the overall barrier to entry for manufacturers. Additionally, the use of purely heterogeneous catalysts in these high-energy regimes can sometimes lead to compromised stereoselectivity or catalyst deactivation over time. These limitations collectively result in higher production costs, longer lead times, and a larger environmental footprint, creating an urgent demand for a more balanced and efficient catalytic solution that can operate under milder conditions.
The Novel Approach
The methodology outlined in CN100503590C introduces a paradigm shift by employing a homogeneous-heterogeneous composite catalyst, specifically RhLn/M-SiO2. This innovative catalyst system combines the high activity and selectivity of soluble rhodium complexes with the structural stability and recyclability of supported metal catalysts. By anchoring the rhodium complex onto a silica support that is itself loaded with transition metals like palladium, nickel, platinum, or ruthenium, the invention creates a synergistic effect that facilitates the hydrogenation of L-malic acid diester under much milder conditions. The reaction can be successfully conducted at pressures ranging from 50 to 150 atmospheres and temperatures between 50 and 100°C, which represents a substantial reduction in energy input and safety risk compared to the >230 atm required by previous methods.
Beyond the operational advantages, this novel approach delivers superior product quality. The composite catalyst ensures high conversion rates while maintaining excellent optical purity, with enantiomeric excess (ee) values reaching up to 98%. This level of stereocontrol is critical for pharmaceutical applications where the wrong enantiomer can be inactive or even toxic. Moreover, the heterogeneous nature of the supported catalyst allows for straightforward separation via simple filtration, eliminating the need for complex distillation or extraction steps often required to remove homogeneous catalysts. This simplification of the work-up procedure not only reduces processing time but also minimizes solvent consumption and waste generation, aligning perfectly with the principles of green chemistry and sustainable manufacturing practices demanded by modern supply chains.
Mechanistic Insights into RhLn/M-SiO2 Composite Catalysis
The core of this technological advancement lies in the precise molecular architecture of the catalyst. The active species is derived from a rhodium complex, typically represented as RhX[Ph2P(CH2)nSi(OR)3]3, where the phosphine ligands contain silyl groups capable of covalent bonding to the silica surface. This anchoring mechanism prevents the leaching of the precious rhodium metal into the product stream, a common failure mode in traditional homogeneous catalysis. Simultaneously, the silica support is pre-loaded with a secondary metal (M), such as Pd, Ni, Pt, or Ru. This dual-metal configuration is believed to facilitate hydrogen activation and transfer more efficiently than either component alone. The rhodium center likely coordinates with the carbonyl groups of the L-malic acid diester, activating the substrate for nucleophilic attack by hydride species generated on the adjacent metal sites within the porous silica matrix.
This cooperative catalysis mechanism explains the observed enhancement in both activity and selectivity. The rigid environment provided by the silica support imposes steric constraints that favor the formation of the desired (S)-enantiomer, thereby suppressing the formation of unwanted byproducts. Furthermore, the interaction between the anchored rhodium complex and the supported metal nanoparticles creates a unique electronic environment that lowers the activation energy for the hydrogenation step. This allows the reaction to proceed rapidly even at the moderate temperatures of 50-100°C. From an impurity control perspective, the stability of the anchored complex ensures that the catalyst does not decompose into inactive species that could promote side reactions, such as over-reduction or racemization. Consequently, the crude reaction mixture is cleaner, requiring less intensive purification to meet the rigorous specifications of a reliable pharmaceutical intermediates supplier.
How to Synthesize (S)-3-Hydroxy-Gamma-Butyrolactone Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction parameters to maximize yield and optical purity. The process begins with the synthesis of the composite catalyst, where a soluble rhodium-phosphine complex is refluxed with a metal-loaded silica support in an inert solvent like toluene. Once the catalyst is prepared and dried, it is introduced into a high-pressure reactor along with the L-malic acid diester substrate and a polar aprotic or alcoholic solvent such as tetrahydrofuran, methanol, or ethanol. The detailed standardized synthesis steps, including specific molar ratios, mixing sequences, and work-up procedures, are outlined below to ensure reproducibility and safety during scale-up operations.
- Prepare the homogeneous-heterogeneous composite catalyst by anchoring a rhodium complex onto metal-loaded silica (M-SiO2).
- Load the reactor with L-malic acid diester substrate, the prepared catalyst, and a suitable solvent such as tetrahydrofuran or ethanol.
- Conduct the hydrogenation reaction at 50-100°C and 50-150 atm pressure, followed by filtration to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic hydrogenation technology offers tangible strategic benefits that extend beyond simple chemical transformation. The shift from stoichiometric borane reductions to a catalytic hydrogenation process fundamentally alters the cost structure of manufacturing (S)-3-hydroxy-gamma-butyrolactone. By eliminating the need for expensive and hazardous reducing agents, the raw material costs are significantly reduced. Furthermore, the ability to recover and reuse the heterogeneous catalyst multiple times without significant loss of activity translates into drastic savings on precious metal consumption. This reduction in consumable costs directly improves the margin profile of the final API, allowing for more competitive pricing in the global market while maintaining high quality standards.
- Cost Reduction in Manufacturing: The transition to this catalytic method removes the financial burden associated with purchasing and disposing of large quantities of borane reagents. Since the catalyst is heterogeneous, it can be filtered and recycled, minimizing the ongoing expense of rhodium and support materials. Additionally, the milder reaction conditions (50-150 atm vs >230 atm) reduce energy consumption for heating and compression, leading to lower utility costs per kilogram of product. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Relying on stoichiometric reagents often exposes manufacturers to supply volatility and price fluctuations in the bulk chemical market. In contrast, hydrogen gas and robust solid catalysts are generally more stable and readily available commodities. The simplified purification process also shortens the overall production cycle time, enabling faster turnaround from order to delivery. This agility is crucial for meeting the just-in-time delivery requirements of major pharmaceutical clients and ensuring continuity of supply for critical medications.
- Scalability and Environmental Compliance: The reduction in hazardous waste generation simplifies regulatory compliance and lowers the costs associated with waste treatment and disposal. The process is inherently safer due to lower operating pressures and temperatures, reducing the risk of accidents and facilitating easier scale-up from pilot plant to commercial tonnage. This environmental and safety profile makes the technology highly attractive for manufacturing in regions with strict environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its integration into their supply chain. The following questions address common inquiries regarding catalyst performance, reaction conditions, and product quality, providing clarity on how this patented technology compares to existing alternatives. These insights are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: What are the advantages of the RhLn/M-SiO2 catalyst over traditional borane reduction?
A: The RhLn/M-SiO2 composite catalyst eliminates the need for expensive and hazardous borane or metal borohydride reagents, significantly reducing waste generation and simplifying downstream purification processes while maintaining high optical purity.
Q: What are the optimal reaction conditions for this hydrogenation process?
A: The process operates efficiently at relatively mild conditions compared to prior art, specifically requiring a hydrogen pressure of 50-150 atmospheres and a temperature range of 50-100°C in solvents like THF or alcohols.
Q: Is the catalyst system reusable for industrial scale-up?
A: Yes, the heterogeneous nature of the supported catalyst allows for easy separation via filtration and subsequent reuse, which is a critical factor for improving the economic viability and sustainability of large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Hydroxy-Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development and production of life-saving medicines. Our technical team has extensively analyzed advanced synthetic routes like the one described in CN100503590C to ensure we offer the most efficient and sustainable manufacturing solutions available. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to deliver products with stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify optical purity and impurity profiles, guaranteeing that every batch meets the exacting standards required by global regulatory bodies.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to optimize their supply chains for (S)-3-hydroxy-gamma-butyrolactone and related intermediates. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation and quality can drive value for your organization and accelerate your drug development timelines.
