Optimized Synthesis of 1,3,2'-N,N,N-Triacetyl Gentamicin C1a for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways for complex aminoglycoside derivatives, particularly those serving as critical precursors for next-generation antibiotics. Patent CN111040007A introduces a significant advancement in the preparation of 1, 3, 2' -N, N, N-triacetyl gentamicin C1a, a pivotal intermediate in the production of Etimicin sulfate. This novel methodology addresses longstanding challenges regarding reaction selectivity and operational safety that have plagued traditional synthesis routes. By leveraging the intrinsic alkalinity of the gentamicin C1a substrate, the process eliminates the need for excessive external bases during the initial protection phase, thereby streamlining the workflow. For R&D directors and procurement specialists, this represents a tangible opportunity to enhance the reliability of their high-purity pharmaceutical intermediates supply chain while mitigating the risks associated with harsh chemical environments. The strategic implementation of this technology ensures a more consistent quality profile, which is essential for maintaining regulatory compliance in the highly scrutinized antibiotic market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of protected gentamicin derivatives has been fraught with inefficiencies stemming from the use of strong inorganic bases and non-selective protection strategies. Conventional protocols often rely on triethylamine or similar bases which, due to their strong basicity, fail to provide sufficient steric hindrance, resulting in poor reaction selectivity and a proliferation of unwanted side products. Furthermore, the post-reaction processing in these traditional methods typically involves complex liquid-liquid extractions using ethyl acetate, which adds unnecessary operational steps and increases the potential for product loss. A critical flaw in older methodologies is the use of sodium hydroxide for pH adjustment; during the concentration phase, this strong alkali can cause the pH to rise uncontrollably, triggering hydrolysis side reactions that degrade the valuable intermediate. These cumulative inefficiencies not only drive up the cost reduction in pharmaceutical manufacturing but also compromise the overall yield and purity required for downstream applications.
The Novel Approach
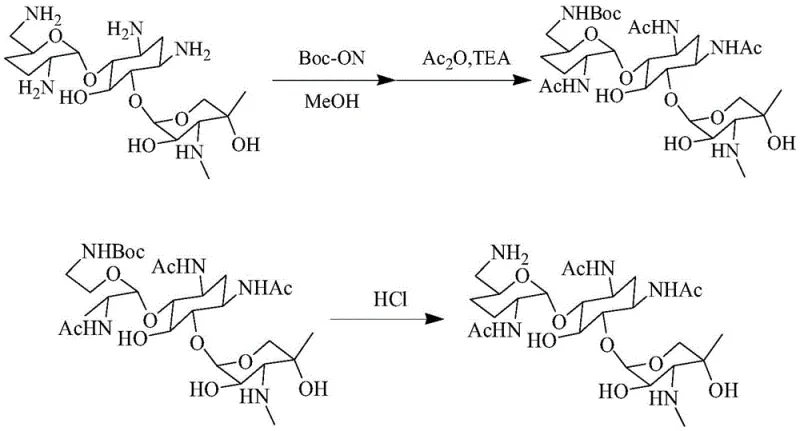
The innovative strategy outlined in the patent data fundamentally reengineers the synthesis by capitalizing on the self-alkalinity and steric properties of the starting material. Instead of forcing the reaction with external strong bases, the new method directly introduces the Boc chemical reagent into the solvent, allowing the 6'-N position to be protected selectively without additional alkaline agents. This simplification removes the need for complicated aftertreatment steps immediately following the protection phase, allowing the process to flow directly into acetylation. The subsequent neutralization step utilizes ammonia water rather than strong inorganic hydroxides, creating a mild environment that prevents the pH spikes responsible for hydrolysis. This refined approach not only simplifies the operation but also significantly enhances synthesis efficiency, offering a scalable solution for the commercial scale-up of complex polymer additives and pharmaceutical intermediates alike.
Mechanistic Insights into Selective Boc Protection and Acetylation
The core of this synthetic breakthrough lies in the precise manipulation of amino group reactivity within the gentamicin C1a structure. By dissolving the substrate in a polar solvent such as methanol and introducing Boc-ON (2-tert-butoxycarbonyloxyimino-2-phenylacetonitrile), the reaction leverages the inherent nucleophilicity differences between the various amine sites. The 6'-N position, being less sterically hindered and sufficiently alkaline, reacts preferentially with the Boc reagent without the need for catalytic bases that might activate other positions indiscriminately. This selectivity is crucial for preventing the formation of multi-protected byproducts that are difficult to separate later in the process. Following this selective protection, the addition of acetic anhydride and a controlled amount of organic base facilitates the acetylation of the remaining 1, 3, and 2' positions. The mechanistic elegance of this sequence ensures that the final deprotection step yields the desired triacetyl derivative with minimal structural degradation.
Controlling impurities in aminoglycoside synthesis is paramount, and this method employs a sophisticated pH management strategy to maintain product integrity. During the deprotection phase, hydrochloric acid is used to cleave the Boc group, but the critical control point occurs during neutralization. Unlike traditional methods where strong bases like NaOH are used, this protocol specifies the use of ammonia water to adjust the pH to a range of 7 to 10, preferably 8 to 9. This mild neutralization prevents the local excess of hydroxide ions that typically catalyze the hydrolysis of the glycosidic bonds or the acetyl groups. By maintaining these温和 conditions, the process ensures that the impurity profile remains clean, specifically minimizing the presence of hydrolyzed gentamicin species. This level of control is essential for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can invalidate entire batches.
How to Synthesize 1,3,2'-N,N,N-Triacetyl Gentamicin C1a Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process begins with the dissolution of gentamicin C1a in a suitable solvent, followed by the controlled addition of the Boc reagent and a prolonged stirring period to ensure complete selective protection. Once the protection is established, the reaction temperature is lowered, and the acetylation reagents are introduced to functionalize the remaining amino groups. The subsequent workup involves careful solvent removal and acid treatment, culminating in a neutralization step that demands precision to avoid product degradation. For detailed operational specifics, stoichiometry, and safety protocols, please refer to the standardized guide below which outlines the exact procedural steps for laboratory and pilot-scale execution.
- Dissolve gentamicin C1a in a solvent such as methanol and add a Boc chemical reagent like Boc-ON, stirring for 12 to 48 hours to utilize self-alkalinity for selective protection.
- Cool the reaction mixture and introduce acetic anhydride along with an organic base to acetylate the remaining amino positions, followed by solvent removal and concentration.
- Treat the concentrated solution with hydrochloric acid to remove the Boc group, neutralize carefully with ammonia water to prevent hydrolysis, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers substantial benefits that extend beyond mere chemical yield, directly impacting the bottom line and supply chain resilience. By eliminating the need for expensive transition metal catalysts and reducing the reliance on harsh inorganic bases, the process inherently lowers the cost of raw materials and waste disposal. The simplification of the workflow, characterized by the removal of complex extraction steps and the use of milder reagents, translates to reduced labor hours and lower energy consumption per kilogram of product. For procurement managers, this means a more predictable cost structure and the ability to secure reliable agrochemical intermediate supplier partnerships that are less vulnerable to fluctuations in reagent pricing. Furthermore, the enhanced safety profile of using ammonia water instead of caustic sodas reduces regulatory burdens and insurance costs associated with hazardous chemical handling.
- Cost Reduction in Manufacturing: The elimination of extra alkali during the Boc protection phase and the removal of complex liquid separation steps drastically simplify the production workflow. This reduction in unit operations leads to significant savings in solvent usage and labor time, effectively lowering the overall manufacturing cost without compromising quality. Additionally, the higher selectivity of the reaction minimizes the formation of byproducts, which reduces the load on purification systems and increases the effective throughput of the facility. These efficiencies collectively contribute to a more competitive pricing model for the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply chains for downstream antibiotic production. By avoiding reagents that are prone to causing variable side reactions, manufacturers can reduce the incidence of failed batches and the associated delays in delivery schedules. This reliability makes the supplier a more attractive partner for long-term contracts, as it mitigates the risk of production stoppages due to quality deviations. Consequently, clients can plan their inventory with greater confidence, knowing that the lead time for high-purity pharmaceutical intermediates will remain stable.
- Scalability and Environmental Compliance: The use of milder conditions and the avoidance of strong inorganic bases make this process highly amenable to scale-up from laboratory to industrial production volumes. The reduced generation of saline waste from neutralization steps aligns with increasingly stringent environmental regulations, facilitating easier permitting and lower waste treatment costs. This environmental compatibility ensures that the manufacturing process remains sustainable and compliant with global green chemistry initiatives, future-proofing the supply chain against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this specialized intermediate. These insights are derived directly from the patented methodology and are intended to clarify the operational advantages and quality implications for potential partners. Understanding these details is crucial for making informed decisions about integrating this technology into your existing production frameworks or sourcing strategies.
Q: Why is ammonia water preferred over sodium hydroxide for neutralization in this synthesis?
A: Using strong alkali like sodium hydroxine can increase the pH too drastically during concentration, leading to hydrolysis side reactions that degrade the product quality, whereas ammonia water offers a milder, safer pH adjustment.
Q: What is the primary advantage of the Boc-ON reagent in this specific pathway?
A: Boc-ON allows for selective protection of the 6'-N position by leveraging the substrate's own alkalinity and steric hindrance, eliminating the need for excessive external bases that cause poor selectivity.
Q: How does this method impact the purity profile of the final Etimicin sulfate intermediate?
A: By minimizing side reactions through controlled pH and selective protection, this method consistently achieves purity levels exceeding 98%, which is critical for meeting stringent pharmacopoeia standards for antibiotic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,2'-N,N,N-Triacetyl Gentamicin C1a Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antibiotics like Etimicin sulfate. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trials and full-scale market launch. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify every batch. Our capability to adapt complex synthetic routes like the one described in CN111040007A allows us to offer a level of technical support and supply security that few competitors can match in the fine chemical sector.
We invite you to collaborate with us to optimize your supply chain and reduce your overall production costs through our advanced manufacturing capabilities. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can enhance your project's success and ensure a steady flow of high-quality materials.
