Revolutionizing L-2-Aminopropanol Production: High-Selectivity Catalytic Hydrogenation for Commercial Scale
The pharmaceutical industry's relentless pursuit of efficient, green, and cost-effective synthetic routes has brought significant attention to the production of chiral amino alcohols, particularly L-2-aminopropanol. As a critical chiral building block for the synthesis of third-generation quinolone antibiotics like Ofloxacin, the quality and cost of L-2-aminopropanol directly impact the final drug's market viability. Recent advancements detailed in patent CN114950452A introduce a groundbreaking catalytic system that fundamentally shifts the paradigm from batch-wise chemical reduction to continuous catalytic hydrogenation. This technology leverages a sophisticated multi-component catalyst comprising CoO, Y2O3, and CeO2 supported on boric acid-immobilized activated carbon. By addressing long-standing challenges such as racemization, low atom economy, and hazardous reagent usage, this innovation offers a robust solution for reliable L-2-aminopropanol supplier networks seeking to modernize their manufacturing capabilities. The patent outlines a method that not only achieves near-perfect conversion rates but also ensures optical purity levels that meet the stringent requirements of global regulatory bodies, marking a significant leap forward in fine chemical engineering.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-2-aminopropanol has been plagued by significant technical and economic inefficiencies inherent to traditional reduction methodologies. The most common legacy approach involves the use of powerful hydride reducing agents such as lithium aluminum hydride (LiAlH4) or sodium borohydride (NaBH4), often preceded by an esterification step to activate the carboxylic acid group of L-2-alanine. These processes are fraught with dangers; LiAlH4 is highly pyrophoric and requires strictly anhydrous conditions, posing severe safety risks during large-scale handling and storage. Furthermore, the stoichiometric nature of these reductions generates substantial amounts of inorganic salt waste, creating a heavy burden on downstream wastewater treatment facilities and increasing the overall environmental footprint of the production line. From a purity perspective, these methods often struggle to maintain high optical integrity, frequently yielding products contaminated with the D-isomer or requiring complex and costly resolution steps to achieve the necessary enantiomeric excess. The batch nature of these reactions also limits throughput, making it difficult to scale up production to meet the surging global demand for fluoroquinolone antibiotics without incurring prohibitive capital expenditures.
The Novel Approach
In stark contrast to these archaic methods, the technology disclosed in patent CN114950452A pioneers a direct catalytic hydrogenation route that transforms L-2-alanine into L-2-aminopropanol in a single, continuous flow process. This novel approach eliminates the need for pre-esterification and hazardous hydride reagents, utilizing molecular hydrogen as the sole reducing agent in an aqueous medium. The core of this innovation lies in the unique catalyst architecture, which combines a boric acid-modified carbon support with a ternary metal oxide system (CoO-Y2O3-CeO2). This specific formulation creates a highly active surface that facilitates the sequential hydrogenation-dehydration-hydrogenation mechanism required to convert the amino acid to the amino alcohol. By operating in a continuous fixed-bed reactor, the process achieves remarkable stability and consistency, allowing for uninterrupted production runs that dramatically enhance plant efficiency. The shift from batch to continuous processing not only simplifies the operational workflow but also inherently improves safety profiles by containing high-pressure hydrogen within a closed, automated system, thereby representing a true evolution in cost reduction in pharmaceutical intermediate manufacturing.
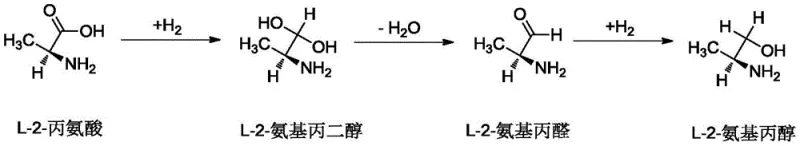
Mechanistic Insights into Co-Y-Ce Catalyzed Hydrogenation
The exceptional performance of this catalytic system is rooted in a complex interplay of electronic and structural effects orchestrated by the multi-component active phase and the functionalized support. The reaction mechanism proceeds through a delicate three-step sequence: initially, the carboxyl group of L-2-alanine is hydrogenated to form L-2-aminopropanediol; this intermediate then undergoes dehydration to yield L-2-aminopropionaldehyde; finally, the aldehyde group is hydrogenated to produce the target L-2-aminopropanol. The boric acid immobilized on the activated carbon plays a pivotal role in the initial activation step. As an electron-deficient species, the boron center interacts with the hydroxide ions of water molecules to release protons, which effectively protonate the zwitterionic structure of L-2-alanine. This protonation disrupts the internal salt formation, rendering the carboxyl group more susceptible to nucleophilic attack by activated hydrogen species on the metal surface. Simultaneously, the acidic sites introduced by the boric acid accelerate the dehydration step, preventing the accumulation of the diol intermediate and driving the equilibrium towards the aldehyde, which is the precursor to the final product.
Beyond the support effects, the synergistic interaction between Cobalt, Yttrium, and Cerium oxides is critical for controlling selectivity and suppressing deleterious side reactions. The primary challenge in amino acid hydrogenation is the tendency for racemization, where the chiral center is lost, leading to the formation of the unwanted D-isomer. The specific combination of CoO, Y2O3, and CeO2 creates a surface environment that stabilizes the chiral configuration of the substrate during the adsorption and reaction phases, effectively inhibiting the racemization pathway. Furthermore, the intermediate L-2-aminopropionaldehyde is highly reactive and prone to polymerization or oligomerization, which can foul the catalyst and reduce yield. The presence of Yttrium and Cerium oxides enhances the dispersion of the Cobalt active sites and modifies their electronic state, ensuring that the hydrogenation of the aldehyde to the alcohol occurs rapidly before polymerization can take place. This precise kinetic control is what allows the process to achieve selectivity levels exceeding 99.5%, effectively eliminating the need for costly downstream purification steps to remove isomers or polymeric byproducts.
How to Synthesize L-2-Aminopropanol Efficiently
Implementing this advanced synthesis route requires careful attention to catalyst preparation and reactor engineering to fully realize the benefits of the patented technology. The process begins with the meticulous preparation of the support, where activated carbon is treated with hydrogen peroxide to optimize its surface properties before being impregnated with boric acid and cured at high temperatures under an inert atmosphere. This ensures a stable anchoring of the acidic function which is crucial for the reaction mechanism. Subsequently, the active metal precursors are loaded via equal-volume impregnation, followed by calcination and a critical reduction activation step in a hydrogen flow to generate the active metallic or oxide species. Once the catalyst is prepared, the synthesis is conducted in a fixed-bed reactor where an aqueous solution of L-2-alanine is pumped continuously over the catalyst bed under controlled temperature and pressure conditions. The detailed standardized synthesis steps see the guide below for specific parameters regarding temperature ramps, pressure settings, and space velocities that ensure optimal performance.
- Prepare the catalyst support by treating activated carbon with hydrogen peroxide, followed by immobilization of boric acid and high-temperature curing in a nitrogen atmosphere.
- Load the active metal components (CoO, Y2O3, CeO2) onto the carrier via equal-volume impregnation using soluble metal salts, followed by drying and calcination.
- Conduct the hydrogenation reaction in a continuous fixed-bed reactor at 50-100°C and 2-10 MPa pressure using an aqueous L-2-alanine feed solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic hydrogenation technology translates into tangible strategic advantages that extend far beyond simple yield improvements. The transition from stoichiometric chemical reduction to catalytic hydrogenation fundamentally alters the cost structure of L-2-aminopropanol production by eliminating the dependency on volatile and expensive reducing agents like lithium aluminum hydride. This shift not only reduces the raw material cost per kilogram but also removes the logistical complexities and safety costs associated with storing and handling hazardous pyrophoric materials. Furthermore, the continuous nature of the fixed-bed process allows for a significantly smaller physical footprint compared to large batch reactors, enabling higher production volumes within existing facility constraints. The robustness of the catalyst, demonstrated by its ability to maintain activity over hundreds of hours of continuous operation, ensures a steady and predictable output, mitigating the risks of production stoppages that can disrupt downstream antibiotic manufacturing schedules.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reducing agents and the associated waste disposal costs leads to a drastic simplification of the production economics. By utilizing molecular hydrogen and a reusable heterogeneous catalyst, the process minimizes the consumption of consumable chemicals, resulting in substantial cost savings over the lifecycle of the plant. Additionally, the high selectivity of the reaction reduces the burden on purification units, lowering energy consumption for distillation and crystallization, which are typically the most energy-intensive stages in fine chemical processing.
- Enhanced Supply Chain Reliability: The use of readily available raw materials such as L-2-alanine and the stability of the catalyst system contribute to a more resilient supply chain. Unlike processes reliant on specialized reagents that may face supply shortages, the inputs for this hydrogenation method are commodity chemicals with stable global markets. The long operational life of the catalyst means fewer changeovers and maintenance shutdowns, ensuring a consistent flow of high-purity intermediates to API manufacturers and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The fixed-bed continuous process is inherently scalable, allowing manufacturers to increase capacity simply by numbering up reactors or increasing bed volume without re-optimizing the entire chemistry. From an environmental perspective, the process aligns perfectly with green chemistry principles by generating minimal waste, avoiding toxic solvents, and producing water as the primary byproduct. This ease of compliance with increasingly stringent environmental regulations reduces the risk of regulatory fines and facilitates smoother permitting for commercial scale-up of complex amino alcohol intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic hydrogenation technology for L-2-aminopropanol production. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does the new catalyst improve optical purity compared to traditional reduction methods?
A: The novel catalyst utilizes a boric acid-immobilized support that protonates the L-2-alanine zwitterion, facilitating specific hydrogenation while the synergistic Co-Y-Ce active sites inhibit racemization. This results in an L-isomer selectivity of over 99.5%, significantly higher than the 75-80% yields often seen with chemical resolution or borohydride reduction methods.
Q: What are the operational stability metrics for this catalytic system?
A: Experimental data from the patent indicates exceptional stability, with the catalyst maintaining consistent conversion rates (100%) and selectivity (>99.5%) over continuous operation periods exceeding 500 to 700 hours without significant deactivation or sintering of the active metal components.
Q: Does this process eliminate the need for hazardous reducing agents?
A: Yes, unlike conventional methods that rely on dangerous and expensive reagents like lithium aluminum hydride (LiAlH4) or sodium borohydride (NaBH4), this technology uses molecular hydrogen (H2) in a continuous fixed-bed process, drastically improving operational safety and reducing hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-2-Aminopropanol Supplier
The technological breakthroughs presented in patent CN114950452A underscore the immense potential of modern catalytic hydrogenation in reshaping the landscape of chiral intermediate synthesis. At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting such advanced methodologies to meet the evolving demands of the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial realities. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards for optical purity and chemical composition.
We invite forward-thinking partners to collaborate with us to leverage this cutting-edge technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our advanced catalytic capabilities can drive efficiency and reliability in your antibiotic production programs.
